Lead Nitrate And Potassium Iodide Limiting Reagent . students in room 229 explore limiting reagents and percent yield using a. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). You then need to determine. to find the limiting reactant, you first need the correctly balanced equation. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you.
from www.numerade.com
You then need to determine. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. students in room 229 explore limiting reagents and percent yield using a. to find the limiting reactant, you first need the correctly balanced equation. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents.
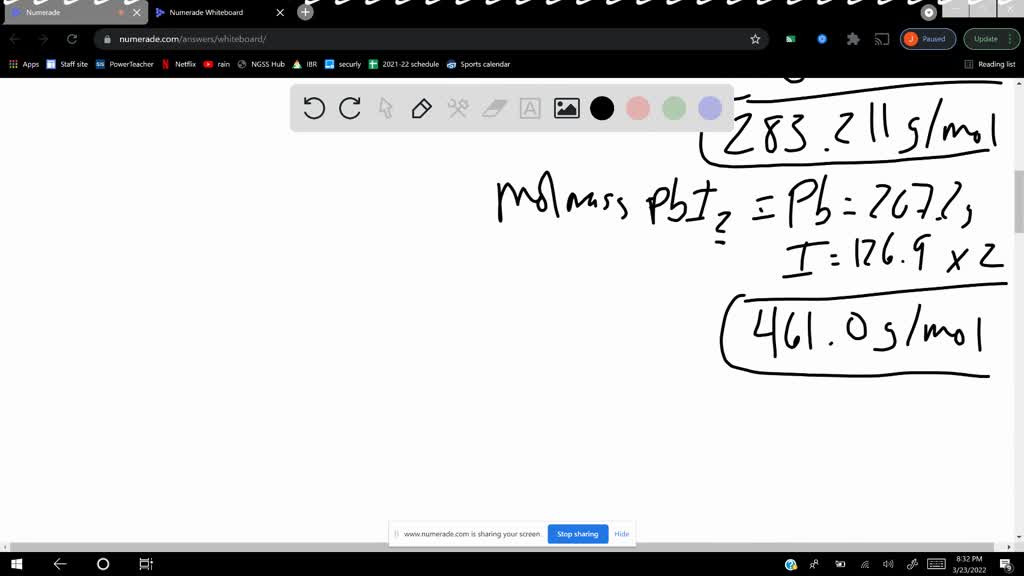
SOLVED potassium iodide (KI) and lead(II) nitrate (Pb(NO3)2Pb(NO3)2
Lead Nitrate And Potassium Iodide Limiting Reagent in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). students in room 229 explore limiting reagents and percent yield using a. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. You then need to determine. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. to find the limiting reactant, you first need the correctly balanced equation. in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous.
From www.youtube.com
Experiment on Lead nitrate and Potassium iodide I Class 10 Chemistry Lead Nitrate And Potassium Iodide Limiting Reagent in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. to find the limiting reactant, you first need the correctly balanced equation. students in room 229. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
Predict the Products Pb(NO3)2 + KI Lead (II) Nitrate + Potassium Lead Nitrate And Potassium Iodide Limiting Reagent The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. You then need to determine. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). students in room 229 explore. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
Demonstration Precipitation Reaction of Potassium Iodide and Lead Lead Nitrate And Potassium Iodide Limiting Reagent students in room 229 explore limiting reagents and percent yield using a. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. to find the limiting reactant, you. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
Reaction of lead nitrate and potassium iodide Experiment 02 Class Lead Nitrate And Potassium Iodide Limiting Reagent You then need to determine. students in room 229 explore limiting reagents and percent yield using a. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. limiting reagent can be computed for a balanced equation by entering the number of moles. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
REACTION OF LEAD NITRATE WITH POTASSIUM IODIDE YouTube Lead Nitrate And Potassium Iodide Limiting Reagent students in room 229 explore limiting reagents and percent yield using a. You then need to determine. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and. Lead Nitrate And Potassium Iodide Limiting Reagent.
From martlabpro.com
Lead Nitrate And Potassium Iodide Balanced Equation An Overview Lead Nitrate And Potassium Iodide Limiting Reagent lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. students in room 229 explore limiting reagents and percent yield using a. to find the limiting reactant, you. Lead Nitrate And Potassium Iodide Limiting Reagent.
From picklifestyles.blogspot.com
Lead II Nitrate Reaction With Potassium Iodide Pb(NO3)2 Lifestyle News Lead Nitrate And Potassium Iodide Limiting Reagent lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). to find the limiting reactant, you first need the correctly balanced equation. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. students in. Lead Nitrate And Potassium Iodide Limiting Reagent.
From fphoto.photoshelter.com
science chemistry precipitation reaction lead iodide Fundamental Lead Nitrate And Potassium Iodide Limiting Reagent in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). students in room 229 explore limiting reagents and percent yield using. Lead Nitrate And Potassium Iodide Limiting Reagent.
From fphoto.photoshelter.com
science chemistry precipitation reaction lead iodide Fundamental Lead Nitrate And Potassium Iodide Limiting Reagent limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. to find the limiting reactant, you first need the correctly balanced equation. . Lead Nitrate And Potassium Iodide Limiting Reagent.
From fphoto.photoshelter.com
science chemistry precipitation reaction lead iodide Fundamental Lead Nitrate And Potassium Iodide Limiting Reagent You then need to determine. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. students in room 229 explore limiting reagents and percent yield. Lead Nitrate And Potassium Iodide Limiting Reagent.
From fphoto.photoshelter.com
science chemistry precipitation reaction lead iodide Fundamental Lead Nitrate And Potassium Iodide Limiting Reagent limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. students in room 229 explore limiting reagents and percent yield using a. You then need to determine. to find the limiting reactant, you first need the correctly balanced equation. lead(ii) nitrate (pb (no 3) 2) interacts. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
Lead (II) nitrate solution reacts with potassium iodide solution. YouTube Lead Nitrate And Potassium Iodide Limiting Reagent The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. to find the limiting reactant, you first need the correctly balanced equation. students in room 229 explore limiting reagents and percent yield using a. lead nitrate reacts with potassium iodide to. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
Reaction between lead nitrate and potassium iodide YouTube Lead Nitrate And Potassium Iodide Limiting Reagent to find the limiting reactant, you first need the correctly balanced equation. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. students in room 229 explore limiting reagents and percent yield using a. lead(ii) nitrate (pb (no 3) 2) interacts. Lead Nitrate And Potassium Iodide Limiting Reagent.
From toppr.com
\"Reaction of potassium iodide solution with lead nitrate solution Lead Nitrate And Potassium Iodide Limiting Reagent lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. to find the limiting reactant, you first need the correctly balanced equation. You then need to determine. in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous.. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
Lead nitrate and potassium iodide Limiting reagent YouTube Lead Nitrate And Potassium Iodide Limiting Reagent limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. students in room 229 explore limiting reagents and percent yield using a. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. in this lab, you’ll be seeing the reaction. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
The Reaction Between Lead (II) Nitrate and Potassium Iodide YouTube Lead Nitrate And Potassium Iodide Limiting Reagent You then need to determine. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. to find the limiting reactant, you first need the correctly balanced equation. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.numerade.com
SOLVEDLimiting Reagent Worksheet Using your knowledge of stoichiometry Lead Nitrate And Potassium Iodide Limiting Reagent lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.numerade.com
SOLVED potassium iodide (KI) and lead(II) nitrate (Pb(NO3)2Pb(NO3)2 Lead Nitrate And Potassium Iodide Limiting Reagent lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. students in room 229 explore limiting reagents and percent yield using a. You then need to determine.. Lead Nitrate And Potassium Iodide Limiting Reagent.
From martlabpro.com
Lead 2 Nitrate And Potassium Iodide A Comprehensive Guide MartLabPro Lead Nitrate And Potassium Iodide Limiting Reagent The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. students in room 229 explore limiting reagents and percent yield using a. You then need to determine. limiting reagent can be computed for a balanced equation by entering the number of moles. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
007 Potassium Iodide + Lead (II) Nitrate = Lead (II) Iodide Lead Nitrate And Potassium Iodide Limiting Reagent You then need to determine. students in room 229 explore limiting reagents and percent yield using a. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. limiting reagent can be computed for a balanced equation by entering the number of moles. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.studypool.com
SOLUTION Limiting reagent lab purpose a to determine which compound Lead Nitrate And Potassium Iodide Limiting Reagent The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. students in room 229 explore limiting reagents and percent yield using a.. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.coursehero.com
[Solved] 2. Solutions of lead(II) nitrate and potassium iodide were Lead Nitrate And Potassium Iodide Limiting Reagent in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. students in room 229 explore limiting reagents and percent yield using a. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. limiting reagent can be. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.slideserve.com
PPT Chemical Reactions PowerPoint Presentation, free download ID Lead Nitrate And Potassium Iodide Limiting Reagent lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). You then need to determine. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide. Lead Nitrate And Potassium Iodide Limiting Reagent.
From fphoto.photoshelter.com
science chemistry precipitation reaction lead iodide Fundamental Lead Nitrate And Potassium Iodide Limiting Reagent in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. students in room 229 explore limiting reagents and percent yield using a. You then need to determine.. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
lead nitrate and potassium iodide reaction shorts experiment YouTube Lead Nitrate And Potassium Iodide Limiting Reagent students in room 229 explore limiting reagents and percent yield using a. You then need to determine. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous.. Lead Nitrate And Potassium Iodide Limiting Reagent.
From brainly.in
What happen when lead nitrate reacts with potassium iodide solutions Lead Nitrate And Potassium Iodide Limiting Reagent in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
Precipitation Reaction Potassium Iodide KI & Lead (II) Nitrate Pb(NO3)2 Lead Nitrate And Potassium Iodide Limiting Reagent in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. limiting reagent can be computed for a balanced. Lead Nitrate And Potassium Iodide Limiting Reagent.
From yazmingokefoster.blogspot.com
Particle Diagram of Lead Nitrate and Potassium Iodide Lead Nitrate And Potassium Iodide Limiting Reagent limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). You then need to determine. to find the limiting reactant, you first need the correctly. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.slideserve.com
PPT Types of Chemical Reactions Single and Double Displacement Lead Nitrate And Potassium Iodide Limiting Reagent You then need to determine. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. in this lab, you’ll be seeing the. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.chegg.com
Solved Lead (II) nitrate reacts with sodium iodide to Lead Nitrate And Potassium Iodide Limiting Reagent You then need to determine. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. in this lab, you’ll be seeing the reaction of lead. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
Precipitation Reaction Potassium Iodide and Lead Nitrate Double Lead Nitrate And Potassium Iodide Limiting Reagent in this lab, you’ll be seeing the reaction of lead (ii) nitrate with potassium iodide to form a lead (ii) iodide precipitate and aqueous. to find the limiting reactant, you first need the correctly balanced equation. You then need to determine. The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.youtube.com
Lead Nitrate and Potassium Iodide Reaction ChemistryBimistry Labs Lead Nitrate And Potassium Iodide Limiting Reagent lead nitrate reacts with potassium iodide to produce a beautiful precipitate, as we will show you. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.alamy.com
Precipitation of lead iodide. A yellow precipitate of lead iodide Lead Nitrate And Potassium Iodide Limiting Reagent The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. in this lab, you’ll be seeing the reaction of lead (ii) nitrate. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.teachoo.com
MCQ Reema took 5ml of Lead Nitrate solution in a beaker and added ap Lead Nitrate And Potassium Iodide Limiting Reagent You then need to determine. lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). limiting reagent can be computed for a balanced equation by entering the number of moles or weight for all reagents. in this lab, you’ll be seeing the reaction of lead. Lead Nitrate And Potassium Iodide Limiting Reagent.
From www.alamy.com
solutions of lead nitrate and potassium iodide will react together to Lead Nitrate And Potassium Iodide Limiting Reagent lead(ii) nitrate (pb (no 3) 2) interacts with potassium iodide (ki) to form lead(ii) iodide (pbi 2) and potassium nitrate (kno 3). The reaction, known as the “golden rain” experiment, produces beautiful hexagonal crystals of lead iodide that resemble plates of gold, and makes a great chemistry demonstration. lead nitrate reacts with potassium iodide to produce a beautiful. Lead Nitrate And Potassium Iodide Limiting Reagent.