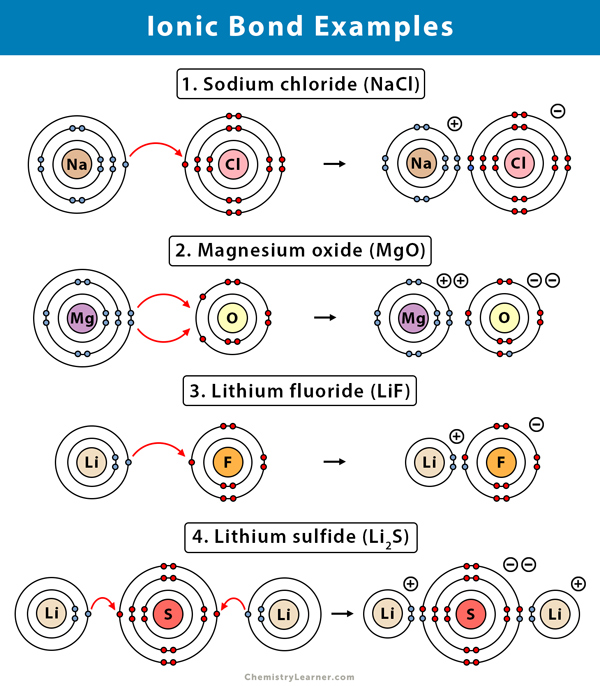
Ionic Bonds Are Typical Of Metal Compounds . The atom that loses electrons becomes a positively charged ion, known as a cation. Ionic compounds dissociate into ions when dissolved in water. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Ionic compounds conduct an electric current when melted or dissolved in water. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds consist of atoms joined together by ionic bonds. The atom that gains electrons becomes a negatively charged ion, known as an anion. Many ionic compounds are binary compounds formed by a. Solutions of ionic compounds and melted ionic compounds. Ionic compounds are hard and brittle. Covalent bonds are highly stable bonds with. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds tend to be crystalline structures with high melting points that are water soluble.
from www.chemistrylearner.com
The atom that loses electrons becomes a positively charged ion, known as a cation. The atom that gains electrons becomes a negatively charged ion, known as an anion. Many ionic compounds are binary compounds formed by a. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds consist of atoms joined together by ionic bonds. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds dissociate into ions when dissolved in water. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Ionic compounds are hard and brittle. Covalent bonds are highly stable bonds with.
Ionic Bond Facts, Definition, Properties, Examples, & Diagrams
Ionic Bonds Are Typical Of Metal Compounds Ionic compounds are hard and brittle. Ionic compounds consist of atoms joined together by ionic bonds. Covalent bonds are highly stable bonds with. Ionic compounds conduct an electric current when melted or dissolved in water. Ionic compounds tend to be crystalline structures with high melting points that are water soluble. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Ionic compounds are hard and brittle. Many ionic compounds are binary compounds formed by a. The atom that gains electrons becomes a negatively charged ion, known as an anion. The atom that loses electrons becomes a positively charged ion, known as a cation. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds dissociate into ions when dissolved in water. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Solutions of ionic compounds and melted ionic compounds.
From sciencenotes.org
Types of Chemical Bonds Ionic Bonds Are Typical Of Metal Compounds Covalent bonds are highly stable bonds with. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds dissociate into ions when dissolved in water. The atom that loses electrons becomes a positively charged ion, known as a cation.. Ionic Bonds Are Typical Of Metal Compounds.
From www.slideserve.com
PPT Chapter 4 Compounds and Their Bonds PowerPoint Presentation, free Ionic Bonds Are Typical Of Metal Compounds Covalent bonds are highly stable bonds with. Many ionic compounds are binary compounds formed by a. Ionic compounds consist of atoms joined together by ionic bonds. Solutions of ionic compounds and melted ionic compounds. Ionic compounds dissociate into ions when dissolved in water. Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Compounds composed. Ionic Bonds Are Typical Of Metal Compounds.
From www.sliderbase.com
Ionic Bonding Presentation Chemistry Ionic Bonds Are Typical Of Metal Compounds Ionic compounds are hard and brittle. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Solutions of ionic compounds and melted ionic compounds. Ionic compounds conduct an electric current when melted or dissolved in water. The atom that gains electrons becomes a negatively charged ion, known as an anion.. Ionic Bonds Are Typical Of Metal Compounds.
From www.youtube.com
PROPERTIES OF IONIC COMPOUNDS AND OCCURRENCE OF METALS YouTube Ionic Bonds Are Typical Of Metal Compounds The atom that loses electrons becomes a positively charged ion, known as a cation. Ionic compounds dissociate into ions when dissolved in water. Solutions of ionic compounds and melted ionic compounds. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Many ionic compounds are binary compounds formed by a.. Ionic Bonds Are Typical Of Metal Compounds.
From www.thoughtco.com
Examples of Ionic Bonds and Compounds Ionic Bonds Are Typical Of Metal Compounds Ionic compounds consist of atoms joined together by ionic bonds. Ionic compounds dissociate into ions when dissolved in water. Solutions of ionic compounds and melted ionic compounds. Ionic compounds conduct an electric current when melted or dissolved in water. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: In. Ionic Bonds Are Typical Of Metal Compounds.
From www.britannica.com
ionic bond Definition, Properties, Examples, & Facts Britannica Ionic Bonds Are Typical Of Metal Compounds Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Ionic compounds dissociate into ions when dissolved in water. Ionic compounds conduct an electric current when melted or dissolved in water. The atom that loses. Ionic Bonds Are Typical Of Metal Compounds.
From www.slideserve.com
PPT Ionic Nomenclature PowerPoint Presentation, free download ID Ionic Bonds Are Typical Of Metal Compounds Ionic compounds dissociate into ions when dissolved in water. Many ionic compounds are binary compounds formed by a. Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Covalent bonds are highly stable bonds with.. Ionic Bonds Are Typical Of Metal Compounds.
From revisechemistry.uk
Chemical Bonds, Ionic, Covalent and Metallic AQA C2 revisechemistry.uk Ionic Bonds Are Typical Of Metal Compounds The atom that loses electrons becomes a positively charged ion, known as a cation. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Covalent bonds are highly stable bonds with. Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Compounds composed of ions. Ionic Bonds Are Typical Of Metal Compounds.
From www.chemistrylearner.com
Ionic, Covalent, and Metallic Bonds Differences and Similarities Ionic Bonds Are Typical Of Metal Compounds Ionic compounds are hard and brittle. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds dissociate into ions when dissolved in water. Many ionic compounds are binary compounds formed by a. Ionic compounds consist of atoms joined. Ionic Bonds Are Typical Of Metal Compounds.
From www.sliderbase.com
Ionic Bonding Presentation Chemistry Ionic Bonds Are Typical Of Metal Compounds Covalent bonds are highly stable bonds with. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds consist of atoms joined together by ionic bonds. The atom that gains electrons becomes a negatively charged ion, known as an anion. Ionic compounds are hard and brittle. Ionic compounds tend. Ionic Bonds Are Typical Of Metal Compounds.
From www.expii.com
Ionic Bond — Formation & Compounds Expii Ionic Bonds Are Typical Of Metal Compounds Many ionic compounds are binary compounds formed by a. Ionic compounds dissociate into ions when dissolved in water. Solutions of ionic compounds and melted ionic compounds. Ionic compounds consist of atoms joined together by ionic bonds. The atom that loses electrons becomes a positively charged ion, known as a cation. Covalent bonds are highly stable bonds with. Ionic compounds tend. Ionic Bonds Are Typical Of Metal Compounds.
From revisechemistry.uk
Chemical Bonds, Ionic, Covalent and Metallic AQA C2 revisechemistry.uk Ionic Bonds Are Typical Of Metal Compounds Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Ionic compounds conduct an electric current when melted or dissolved in water. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Covalent bonds are highly stable bonds with. Ionic compounds dissociate into ions when. Ionic Bonds Are Typical Of Metal Compounds.
From sciencenotes.org
Ionic Bond Definition and Examples Ionic Bonds Are Typical Of Metal Compounds Ionic compounds conduct an electric current when melted or dissolved in water. Ionic compounds are hard and brittle. Solutions of ionic compounds and melted ionic compounds. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Ionic compounds dissociate into ions when dissolved in water. Ionic compounds tend to be crystalline structures with high melting points that are water soluble.. Ionic Bonds Are Typical Of Metal Compounds.
From derekcarrsavvy-chemist.blogspot.com
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures Ionic Bonds Are Typical Of Metal Compounds Ionic compounds dissociate into ions when dissolved in water. Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: The atom that gains electrons becomes a negatively charged ion, known as an anion. In an. Ionic Bonds Are Typical Of Metal Compounds.
From general.chemistrysteps.com
Naming Ionic Compounds Chemistry Steps Ionic Bonds Are Typical Of Metal Compounds Many ionic compounds are binary compounds formed by a. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Solutions of ionic compounds and melted ionic compounds. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Compounds composed of ions are called ionic compounds (or salts), and their constituent. Ionic Bonds Are Typical Of Metal Compounds.
From en.wikipedia.org
Ionic bonding Wikipedia Ionic Bonds Are Typical Of Metal Compounds Ionic compounds conduct an electric current when melted or dissolved in water. Ionic compounds tend to be crystalline structures with high melting points that are water soluble. The atom that gains electrons becomes a negatively charged ion, known as an anion. Many ionic compounds are binary compounds formed by a. Compounds composed of ions are called ionic compounds (or salts),. Ionic Bonds Are Typical Of Metal Compounds.
From www.sliderbase.com
Chemical Bonds Ionic Bonds Are Typical Of Metal Compounds Ionic compounds dissociate into ions when dissolved in water. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds conduct an electric current when melted or dissolved in water. Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Many ionic compounds are. Ionic Bonds Are Typical Of Metal Compounds.
From www.slideserve.com
PPT Ionic Bonding Part I PowerPoint Presentation, free download ID Ionic Bonds Are Typical Of Metal Compounds Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds are hard and brittle. Ionic compounds consist of atoms joined together by ionic bonds. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions. Ionic Bonds Are Typical Of Metal Compounds.
From courses.lumenlearning.com
Molecular and Ionic Compounds Chemistry for Majors Ionic Bonds Are Typical Of Metal Compounds Ionic compounds are hard and brittle. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Ionic compounds dissociate into ions when dissolved in water. Many ionic compounds are binary compounds formed by a. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: The atom that gains electrons becomes. Ionic Bonds Are Typical Of Metal Compounds.
From biologydictionary.net
Ionic Bond Examples Biology Dictionary Ionic Bonds Are Typical Of Metal Compounds Solutions of ionic compounds and melted ionic compounds. The atom that gains electrons becomes a negatively charged ion, known as an anion. Ionic compounds conduct an electric current when melted or dissolved in water. Many ionic compounds are binary compounds formed by a. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together. Ionic Bonds Are Typical Of Metal Compounds.
From www.jagranjosh.com
What are Ionic Compounds and how they are formed? Ionic Bonds Are Typical Of Metal Compounds Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Covalent bonds are highly stable bonds with. Solutions of ionic compounds and melted ionic compounds. Many ionic compounds are binary compounds formed by a. Ionic compounds are hard and brittle.. Ionic Bonds Are Typical Of Metal Compounds.
From www.slideserve.com
PPT Intro to Bonding Ionic Compounds (Type 1 and 2 Binary Compounds Ionic Bonds Are Typical Of Metal Compounds In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Solutions of ionic compounds and melted ionic compounds. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds dissociate into ions when dissolved in water. Covalent bonds are highly stable bonds with. The atom that loses electrons becomes. Ionic Bonds Are Typical Of Metal Compounds.
From www.sliderbase.com
Ionic Bonding Presentation Chemistry Ionic Bonds Are Typical Of Metal Compounds Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Many ionic compounds are binary compounds formed by a. Ionic compounds are hard and brittle. Covalent bonds are highly stable bonds with. The atom that. Ionic Bonds Are Typical Of Metal Compounds.
From derekcarrsavvy-chemist.blogspot.co.uk
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures Ionic Bonds Are Typical Of Metal Compounds Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Many ionic compounds are binary compounds formed by a. Covalent bonds are highly stable bonds with. Solutions of ionic compounds and melted ionic compounds. Ionic compounds conduct an electric current when melted or dissolved in water. The atom that loses. Ionic Bonds Are Typical Of Metal Compounds.
From www.youtube.com
Examples of Ionic Compoiunds YouTube Ionic Bonds Are Typical Of Metal Compounds Solutions of ionic compounds and melted ionic compounds. Covalent bonds are highly stable bonds with. The atom that gains electrons becomes a negatively charged ion, known as an anion. Many ionic compounds are binary compounds formed by a. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Ionic compounds are hard and brittle. Ionic compounds dissociate into ions when. Ionic Bonds Are Typical Of Metal Compounds.
From www.slideserve.com
PPT Chapter 4 Compounds and Their Bonds PowerPoint Presentation, free Ionic Bonds Are Typical Of Metal Compounds The atom that gains electrons becomes a negatively charged ion, known as an anion. Many ionic compounds are binary compounds formed by a. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds are hard and brittle. Ionic. Ionic Bonds Are Typical Of Metal Compounds.
From www.britannica.com
chemical bonding Ionic and covalent compounds Britannica Ionic Bonds Are Typical Of Metal Compounds Ionic compounds consist of atoms joined together by ionic bonds. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds tend to be crystalline structures with high melting points that are water soluble. The atom that gains electrons. Ionic Bonds Are Typical Of Metal Compounds.
From www.thoughtco.com
Examples of Ionic Bonds and Compounds Ionic Bonds Are Typical Of Metal Compounds Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Many ionic compounds are binary compounds formed by a. The atom that loses electrons becomes a positively charged ion, known as a cation. Ionic compounds are hard and brittle. Ionic compounds tend to be crystalline structures with high melting points. Ionic Bonds Are Typical Of Metal Compounds.
From chem.libretexts.org
Ionic Solids Chemistry LibreTexts Ionic Bonds Are Typical Of Metal Compounds Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Many ionic compounds are binary compounds formed by a. Covalent bonds are highly stable bonds with. The atom that gains electrons becomes a negatively charged ion, known as an anion. Ionic compounds are hard and brittle. Ionic compounds dissociate into ions when dissolved in water.. Ionic Bonds Are Typical Of Metal Compounds.
From chemistrysolution55.blogspot.com
Chemistry B.Sc Level How many types of chemical bond Ionic Bonds Are Typical Of Metal Compounds Ionic compounds tend to be crystalline structures with high melting points that are water soluble. The atom that gains electrons becomes a negatively charged ion, known as an anion. Solutions of ionic compounds and melted ionic compounds. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: The atom that. Ionic Bonds Are Typical Of Metal Compounds.
From www.slideserve.com
PPT IONIC COMPOUNDS PowerPoint Presentation, free download ID907550 Ionic Bonds Are Typical Of Metal Compounds Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Ionic compounds are hard and brittle. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Solutions of ionic compounds and melted ionic compounds. The. Ionic Bonds Are Typical Of Metal Compounds.
From www.chemistrylearner.com
Ionic Bond Facts, Definition, Properties, Examples, & Diagrams Ionic Bonds Are Typical Of Metal Compounds Ionic compounds are hard and brittle. Ionic compounds dissociate into ions when dissolved in water. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Many ionic compounds are binary. Ionic Bonds Are Typical Of Metal Compounds.
From slideplayer.com
9 Chemical Bonds Chemical Bond atoms or ions strongly attached to one Ionic Bonds Are Typical Of Metal Compounds Covalent bonds are highly stable bonds with. In an ionic solution, the \(\ce{a^+}\) ions migrate toward the. Ionic compounds are hard and brittle. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: The atom that gains electrons becomes a negatively charged ion, known as an anion. Ionic compounds tend. Ionic Bonds Are Typical Of Metal Compounds.
From www.britannica.com
Ionic bond Definition, Properties, Examples, & Facts Britannica Ionic Bonds Are Typical Of Metal Compounds Covalent bonds are highly stable bonds with. Ionic compounds dissociate into ions when dissolved in water. Many ionic compounds are binary compounds formed by a. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: Solutions of ionic compounds and melted ionic compounds. Compounds composed of ions are called ionic. Ionic Bonds Are Typical Of Metal Compounds.
From jaylenyouthmejia.blogspot.com
Ionic Bonding Between Metals and Nonmetals Ionic Bonds Are Typical Of Metal Compounds Ionic compounds conduct an electric current when melted or dissolved in water. The atom that gains electrons becomes a negatively charged ion, known as an anion. Ionic compounds dissociate into ions when dissolved in water. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: In an ionic solution, the. Ionic Bonds Are Typical Of Metal Compounds.