Evaporation Rate Of Hydrochloric Acid . How does different type of liquids affect the rate of evaporation of the liquids? Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. But we don't know how to proceed with this, as we. Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented.
from mavink.com
How does different type of liquids affect the rate of evaporation of the liquids? At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. But we don't know how to proceed with this, as we. Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented.
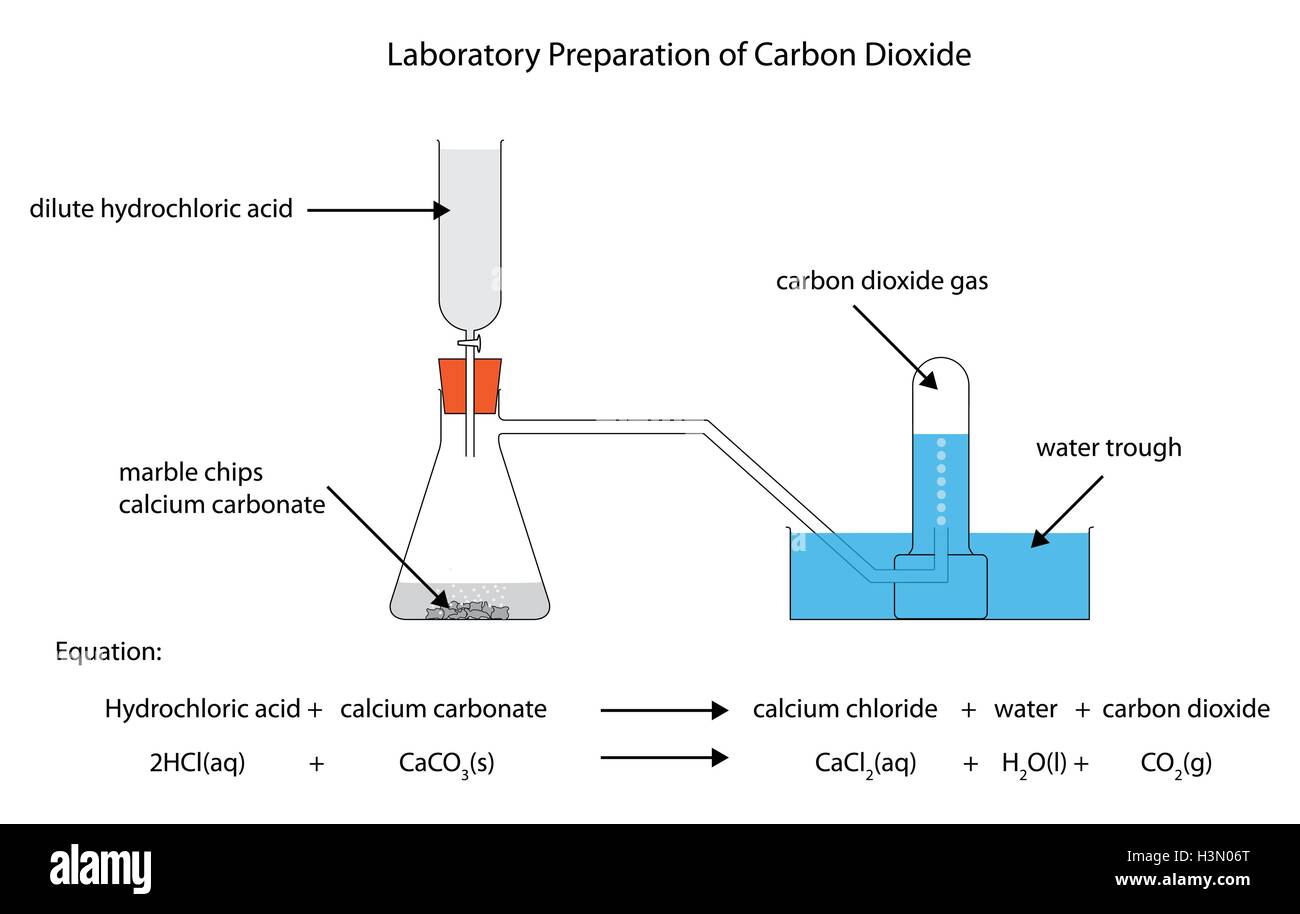
Hydrochloric Acid Diagram
Evaporation Rate Of Hydrochloric Acid How does different type of liquids affect the rate of evaporation of the liquids? But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. How does different type of liquids affect the rate of evaporation of the liquids? Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors.
From www.youtube.com
Reaction Rate Hydrochloric acid + Sodium Thiosulfate Concentration Evaporation Rate Of Hydrochloric Acid Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60%. Evaporation Rate Of Hydrochloric Acid.
From www.researchgate.net
Effect of hydrochloric acid, lactic acid, and acetic acid On rate of Evaporation Rate Of Hydrochloric Acid We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. How does different type of liquids affect the rate of evaporation of the liquids? Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. At 95% confidence, we find the evaporation coefficient for. Evaporation Rate Of Hydrochloric Acid.
From www.researchgate.net
Rate of evaporation with and without removing vapors above the liquid Evaporation Rate Of Hydrochloric Acid But we don't know how to proceed with this, as we. How does different type of liquids affect the rate of evaporation of the liquids? At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. Results of experimental study on the evaporation of hydrochloric acid in. Evaporation Rate Of Hydrochloric Acid.
From www.tessshebaylo.com
Chemical Equation For Ionization Of Hydrochloric Acid In Water Evaporation Rate Of Hydrochloric Acid Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to. Evaporation Rate Of Hydrochloric Acid.
From www.studypool.com
SOLUTION Experiment preparation and standardization of 0.1 m HCL Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by. Evaporation Rate Of Hydrochloric Acid.
From edu.rsc.org
Hydrochloric acid Magnificent molecules RSC Education Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure. Evaporation Rate Of Hydrochloric Acid.
From www.scribd.com
Standardization of Hydrochloric Acid PDF Titration Chemistry Evaporation Rate Of Hydrochloric Acid Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented. How does different type of liquids affect the rate of evaporation of the liquids? But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a. Evaporation Rate Of Hydrochloric Acid.
From www.researchgate.net
4. Hydrochloric acid and sodium hydroxide neutralisation curves, [99 Evaporation Rate Of Hydrochloric Acid But we don't know how to proceed with this, as we. Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. How does different type of liquids affect the. Evaporation Rate Of Hydrochloric Acid.
From www.peoi.org
Chapter 10 Section C Properties of Liquids Evaporation Rate Of Hydrochloric Acid We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. But we don't know how to proceed with this, as we. Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented. At 95% confidence, we find the evaporation coefficient for 1.0 m. Evaporation Rate Of Hydrochloric Acid.
From www.researchgate.net
a Schematic illustration of water evaporation process and mechanism. b Evaporation Rate Of Hydrochloric Acid How does different type of liquids affect the rate of evaporation of the liquids? We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be. Evaporation Rate Of Hydrochloric Acid.
From www.scribd.com
Us8163092 PDF PDF Hydrochloric Acid Evaporation Evaporation Rate Of Hydrochloric Acid Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. How does different type of liquids affect the rate of evaporation of the liquids? Results of experimental study on the. Evaporation Rate Of Hydrochloric Acid.
From www.researchgate.net
3 Effect of concentration on the rate of reaction between sodium Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and. Evaporation Rate Of Hydrochloric Acid.
From chemistry.stackexchange.com
surface chemistry Evaporation Rate of Hydrochloric acid Chemistry Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation coefficient for. Evaporation Rate Of Hydrochloric Acid.
From astonlayaan.blogspot.com
hydrochloric acid water phase diagram AstonLayaan Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. But we don't know how to proceed with this, as. Evaporation Rate Of Hydrochloric Acid.
From mavink.com
Hydrochloric Acid Diagram Evaporation Rate Of Hydrochloric Acid How does different type of liquids affect the rate of evaporation of the liquids? At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow. Evaporation Rate Of Hydrochloric Acid.
From www.nagwa.com
Question Video Calculating the Average Rate of Reaction of Evaporation Rate Of Hydrochloric Acid How does different type of liquids affect the rate of evaporation of the liquids? But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. Yes, hydrochloric acid (hcl) does evaporate, and the evaporation. Evaporation Rate Of Hydrochloric Acid.
From www.researchgate.net
Test step diagram of hydrochloric acid leaching of valuable elements Evaporation Rate Of Hydrochloric Acid Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation. Evaporation Rate Of Hydrochloric Acid.
From byjus.com
Write the neutralization reaction between Hydrochloric acid HCI and Evaporation Rate Of Hydrochloric Acid We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. But we don't know how to proceed with this, as we. At 95% confidence, we. Evaporation Rate Of Hydrochloric Acid.
From www.youtube.com
Rate of Reaction of Sodium Thiosulfate (Na2S2O3) and Hydrochloric Acid Evaporation Rate Of Hydrochloric Acid We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. At 95% confidence, we. Evaporation Rate Of Hydrochloric Acid.
From study.com
Sodium Thiosulfate & Hydrochloric Acid Experiment Lesson Evaporation Rate Of Hydrochloric Acid How does different type of liquids affect the rate of evaporation of the liquids? At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease. Evaporation Rate Of Hydrochloric Acid.
From www.slideserve.com
PPT Hydrochloric Acid Suppliers in UAE Hydrochloric Acid Evaporation Rate Of Hydrochloric Acid Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. But we don't know how to proceed with this, as we. We have to recommend a blower,. Evaporation Rate Of Hydrochloric Acid.
From www.flinnsci.com
Rate of Reaction of Sodium Thiosulfate and Hydrochloric Acid Evaporation Rate Of Hydrochloric Acid Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. How does different type of liquids affect the rate of evaporation of the liquids? Results of experimental study on the evaporation of hydrochloric acid in a wind tunnel have been presented. But we don't know how to proceed with this, as we. We have. Evaporation Rate Of Hydrochloric Acid.
From www.coursehero.com
[Solved] The heat of neutralization reaction between hydrochloric acid Evaporation Rate Of Hydrochloric Acid But we don't know how to proceed with this, as we. How does different type of liquids affect the rate of evaporation of the liquids? At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. Yes, hydrochloric acid (hcl) does evaporate, and the evaporation. Evaporation Rate Of Hydrochloric Acid.
From mavink.com
Hydrochloric Acid Phase Diagram Evaporation Rate Of Hydrochloric Acid Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. But we don't know how to proceed with this, as we. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. How does different type of liquids affect. Evaporation Rate Of Hydrochloric Acid.
From animalia-life.club
Common Acid Hcl Evaporation Rate Of Hydrochloric Acid We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. But we don't know how to proceed with this, as we. At 95%. Evaporation Rate Of Hydrochloric Acid.
From edu.rsc.org
Hydrochloric acid Magnificent molecules RSC Education Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24. Evaporation Rate Of Hydrochloric Acid.
From chart-studio.plotly.com
Effect of Temperature on Reaction Rate Using Magnesium and Hydrochloric Evaporation Rate Of Hydrochloric Acid But we don't know how to proceed with this, as we. How does different type of liquids affect the rate of evaporation of the liquids? Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60%. Evaporation Rate Of Hydrochloric Acid.
From www.researchgate.net
Capacity factors of actinides as a function of hydrochloric acid Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. Results of experimental study on the evaporation of hydrochloric acid. Evaporation Rate Of Hydrochloric Acid.
From www.rolledalloys.com
Hydrochloric Acid Rolled Alloys Evaporation Rate Of Hydrochloric Acid How does different type of liquids affect the rate of evaporation of the liquids? At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow. Evaporation Rate Of Hydrochloric Acid.
From www.researchgate.net
Experimental data on the evaporation of hydrochloric acid. Download Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. But we don't know how to proceed with this, as we. How does different type of liquids affect the rate. Evaporation Rate Of Hydrochloric Acid.
From www.researchgate.net
Effect of hydrochloric acid on leaching rate Download Scientific Diagram Evaporation Rate Of Hydrochloric Acid We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. How does different type of liquids affect the rate of evaporation of the. Evaporation Rate Of Hydrochloric Acid.
From stock.adobe.com
Vetor de Acidbase, neutralization reaction of hydrochloric acid and Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24. Evaporation Rate Of Hydrochloric Acid.
From chem.libretexts.org
Chapter 11.4 Vapor Pressure Chemistry LibreTexts Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. We have to recommend a blower, so that most of the generated fumes can be treated at an appropriate flow rate. Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by. Evaporation Rate Of Hydrochloric Acid.
From wisc.pb.unizin.org
Acids, Bases, Neutralization, and GasForming Reactions (M3Q34) UW Evaporation Rate Of Hydrochloric Acid At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water,. Yes, hydrochloric acid (hcl) does evaporate, and the evaporation rate is influenced by several key factors. How does different type of liquids affect the rate of evaporation of the liquids? We have to recommend a blower,. Evaporation Rate Of Hydrochloric Acid.
From mavink.com
Water Evaporation Chart Evaporation Rate Of Hydrochloric Acid But we don't know how to proceed with this, as we. How does different type of liquids affect the rate of evaporation of the liquids? At 95% confidence, we find the evaporation coefficient for 1.0 m hcl to be 0.24 ± 0.04, a ∼60% decrease relative to pure water, and for. At 95% confidence, we find the evaporation coefficient for. Evaporation Rate Of Hydrochloric Acid.