Will A Glass Of Water Evaporate . Pure or distilled water evaporates faster than saltwater and other types of impure water. When liquid water meets dry air, it is not in equilibrium; On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. This is because water ordinarily contains significant amounts of dissolved air and other impurities, which are observed as small bubbles of air in a glass of water. But why does it form bubbles when it boils? Water molecules evaporate off the surface until the amount of water in the air. Thus, it can evaporate without limit at this temperature and pressure. When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. In the system (glass of water etc.) the water molecules have a distribution of different kinetic energies. Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk.
from stock.adobe.com
Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk. When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. In the system (glass of water etc.) the water molecules have a distribution of different kinetic energies. But why does it form bubbles when it boils? Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. Pure or distilled water evaporates faster than saltwater and other types of impure water. Thus, it can evaporate without limit at this temperature and pressure. Water molecules evaporate off the surface until the amount of water in the air.
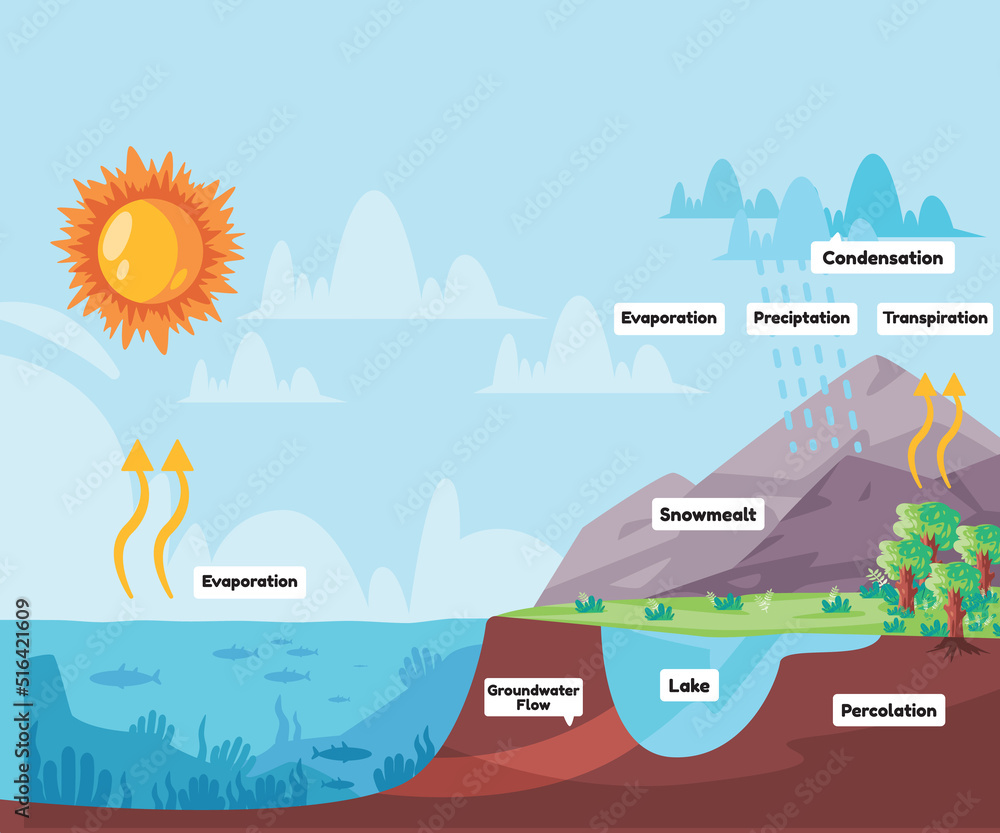
Water cycle process water evaporates to atmosphere condenses into rain
Will A Glass Of Water Evaporate Thus, it can evaporate without limit at this temperature and pressure. When liquid water meets dry air, it is not in equilibrium; This is because water ordinarily contains significant amounts of dissolved air and other impurities, which are observed as small bubbles of air in a glass of water. Water molecules evaporate off the surface until the amount of water in the air. When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk. On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. In the system (glass of water etc.) the water molecules have a distribution of different kinetic energies. Thus, it can evaporate without limit at this temperature and pressure. Pure or distilled water evaporates faster than saltwater and other types of impure water. Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. But why does it form bubbles when it boils?
From www.youtube.com
Evaporation Experiment YouTube Will A Glass Of Water Evaporate Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. But why does it form bubbles when it boils? Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the. Will A Glass Of Water Evaporate.
From chem.libretexts.org
16.2 The Liquid State Chemistry LibreTexts Will A Glass Of Water Evaporate Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. On a hot day, the water molecules in your perspiration absorb body heat and. Will A Glass Of Water Evaporate.
From www.nsta.org
Q What’s the difference between evaporation and boiling? NSTA Will A Glass Of Water Evaporate Thus, it can evaporate without limit at this temperature and pressure. This is because water ordinarily contains significant amounts of dissolved air and other impurities, which are observed as small bubbles of air in a glass of water. Pure or distilled water evaporates faster than saltwater and other types of impure water. When the surface is exposed to sunlight, some. Will A Glass Of Water Evaporate.
From www.youtube.com
Water evaporation experiment YouTube Will A Glass Of Water Evaporate But why does it form bubbles when it boils? On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. This is because water ordinarily contains significant amounts of dissolved air and other impurities, which are observed as small bubbles of air in a glass of water. When the surface. Will A Glass Of Water Evaporate.
From www.atmo.arizona.edu
Tue., Oct. 16 notes Will A Glass Of Water Evaporate When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. Pure or distilled water evaporates faster than saltwater and other types of impure water. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the.. Will A Glass Of Water Evaporate.
From www.sciencephoto.com
Evaporation of water and salt solution Stock Image C036/3503 Will A Glass Of Water Evaporate Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. Water molecules evaporate off the surface until the amount. Will A Glass Of Water Evaporate.
From www.watercubedesign.it
Evaporation Watercube Design Will A Glass Of Water Evaporate Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. But why does it form bubbles when it boils? This is because water ordinarily contains significant amounts of dissolved air and other impurities, which are observed as small bubbles of air. Will A Glass Of Water Evaporate.
From www.bbc.co.uk
Evaporation BBC Bitesize Will A Glass Of Water Evaporate On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. Pure or distilled water evaporates faster than saltwater and other types of impure water. In the system (glass of water etc.) the water molecules have a distribution of different kinetic energies. Saltwater has another substance dissolved in it (salt),. Will A Glass Of Water Evaporate.
From www.snexplores.org
Explainer What are the different states of matter? Will A Glass Of Water Evaporate Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk. Thus, it can evaporate without limit at this temperature. Will A Glass Of Water Evaporate.
From www.numerade.com
SOLVED 'please help me with questions 3, 4 5) Reading list Will A Glass Of Water Evaporate Thus, it can evaporate without limit at this temperature and pressure. On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk. This is because water ordinarily contains. Will A Glass Of Water Evaporate.
From www.slideserve.com
PPT The Water Cycle and How Humans Impact It PowerPoint Presentation Will A Glass Of Water Evaporate Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. When liquid water meets dry air, it is not. Will A Glass Of Water Evaporate.
From www.lifeschemistrypress.com
The invisibility of Water and air Life's Chemistry Press Will A Glass Of Water Evaporate When liquid water meets dry air, it is not in equilibrium; Pure or distilled water evaporates faster than saltwater and other types of impure water. In the system (glass of water etc.) the water molecules have a distribution of different kinetic energies. Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making. Will A Glass Of Water Evaporate.
From sites.google.com
Y7 Science helper Reference page Will A Glass Of Water Evaporate Water molecules evaporate off the surface until the amount of water in the air. Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. In the system (glass of water etc.) the water molecules have a distribution of different kinetic. Will A Glass Of Water Evaporate.
From www.animalia-life.club
Evaporation Water Cycle Will A Glass Of Water Evaporate On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk. Thus, it can evaporate without limit at this temperature and pressure. Saltwater has another substance dissolved in. Will A Glass Of Water Evaporate.
From www.visionlearning.com
Substances Earth Science Visionlearning Will A Glass Of Water Evaporate Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. Water molecules evaporate off the surface until the amount of water in the air. When liquid water meets dry air, it is not in equilibrium; When the surface is exposed to. Will A Glass Of Water Evaporate.
From news.mit.edu
Experiments reveal the physics of evaporation MIT News Will A Glass Of Water Evaporate When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. But why does it form bubbles when it boils? Pure or distilled water evaporates faster than saltwater and other types of impure water. When liquid water meets dry air, it is not in equilibrium; Thus, it can evaporate without limit at this temperature. Will A Glass Of Water Evaporate.
From www.vecteezy.com
Front view a glass of water isolated on transparent background 32545950 PNG Will A Glass Of Water Evaporate But why does it form bubbles when it boils? Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk. Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the. Will A Glass Of Water Evaporate.
From www.clearwaycommunitysolar.com
Exploring the Water Cycle Clearway Community Solar Will A Glass Of Water Evaporate Thus, it can evaporate without limit at this temperature and pressure. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your. Will A Glass Of Water Evaporate.
From www.teachoo.com
Why is Water liquid at room temperature? Give reasons Teachoo Will A Glass Of Water Evaporate Water molecules evaporate off the surface until the amount of water in the air. On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy. Will A Glass Of Water Evaporate.
From faithandsciencemeet.com
Just a Glass of Water Faith and Science Meet Will A Glass Of Water Evaporate On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. Water molecules evaporate off the surface until the amount of water in the air. This is because water ordinarily contains significant amounts of dissolved air and other impurities, which are observed as small bubbles of air in a glass. Will A Glass Of Water Evaporate.
From slideplayer.com
Changes in State (PHASE CHANGES)—Notes ppt download Will A Glass Of Water Evaporate Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. This is because water ordinarily contains significant amounts of dissolved air and other. Will A Glass Of Water Evaporate.
From www.scienceabc.com
Why Does Water Evaporate At Room Temperature? Will A Glass Of Water Evaporate But why does it form bubbles when it boils? Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in. Will A Glass Of Water Evaporate.
From kids.britannica.com
evaporation and condensation Kids Britannica Kids Homework Help Will A Glass Of Water Evaporate When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. In the system (glass of water etc.) the water molecules have a distribution of different kinetic energies. But why does it form bubbles when it boils? Water molecules evaporate off the surface until the amount of water in the air. Water evaporates at. Will A Glass Of Water Evaporate.
From fphoto.photoshelter.com
science chemistry experiment states of matter Fundamental Photographs Will A Glass Of Water Evaporate When liquid water meets dry air, it is not in equilibrium; Water molecules evaporate off the surface until the amount of water in the air. Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk. When the surface is exposed to sunlight, some molecules gain enough energy to. Will A Glass Of Water Evaporate.
From sciencing.com
Fast Ways to Make Water Evaporate Sciencing Will A Glass Of Water Evaporate Water molecules evaporate off the surface until the amount of water in the air. When liquid water meets dry air, it is not in equilibrium; Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. Water easily evaporates at its. Will A Glass Of Water Evaporate.
From slideplayer.com
The Water Cycle By Erin Cole 11/2/ ppt download Will A Glass Of Water Evaporate Water evaporates at room temperature because the molecules at the surface of the liquid have weaker attraction than those in the bulk. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. When liquid water meets dry air, it is not. Will A Glass Of Water Evaporate.
From young6science3.weebly.com
5Th grade science The Water Cycle Will A Glass Of Water Evaporate In the system (glass of water etc.) the water molecules have a distribution of different kinetic energies. But why does it form bubbles when it boils? Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. When liquid water meets. Will A Glass Of Water Evaporate.
From stock.adobe.com
Water cycle process water evaporates to atmosphere condenses into rain Will A Glass Of Water Evaporate When liquid water meets dry air, it is not in equilibrium; Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing. Will A Glass Of Water Evaporate.
From www.ency123.com
Water Evaporation and Condensation Ency123 Will A Glass Of Water Evaporate Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. In the system (glass of water etc.) the water molecules have a distribution of different kinetic energies. When the surface is exposed to sunlight, some molecules gain enough energy to escape. Will A Glass Of Water Evaporate.
From stock.adobe.com
Phase change transition diagram. States matter schema. Evaporation Will A Glass Of Water Evaporate When liquid water meets dry air, it is not in equilibrium; Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. Pure or. Will A Glass Of Water Evaporate.
From www.pinterpandai.com
Penjelasan Evaporasi atau Penguapan dan Contohnya Will A Glass Of Water Evaporate But why does it form bubbles when it boils? Pure or distilled water evaporates faster than saltwater and other types of impure water. Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. When liquid water meets dry air, it is. Will A Glass Of Water Evaporate.
From water.usgs.gov
Evaporation, The Water Cycle, from USGS WaterScience School Will A Glass Of Water Evaporate When liquid water meets dry air, it is not in equilibrium; But why does it form bubbles when it boils? Water easily evaporates at its boiling point (212° f, 100° c) but evaporates much more slowly at its freezing point because of the heat energy required to evaporate the. Water evaporates at room temperature because the molecules at the surface. Will A Glass Of Water Evaporate.
From science.howstuffworks.com
1 Why does water evaporate at room temperature? 10 Science Questions Will A Glass Of Water Evaporate Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy to escape the surface. But why does it form bubbles when it boils? Thus, it can evaporate without limit at this temperature and pressure. In the system (glass of water etc.) the water molecules. Will A Glass Of Water Evaporate.
From www.jagranjosh.com
Photomolecular Effect Light Can Make Water Evaporate Without Heat, MIT Will A Glass Of Water Evaporate This is because water ordinarily contains significant amounts of dissolved air and other impurities, which are observed as small bubbles of air in a glass of water. Water molecules evaporate off the surface until the amount of water in the air. Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them. Will A Glass Of Water Evaporate.
From www.aplustopper.com
How can we Separate a Mixture of a Solid and a Liquid using Evaporation Will A Glass Of Water Evaporate In the system (glass of water etc.) the water molecules have a distribution of different kinetic energies. When the surface is exposed to sunlight, some molecules gain enough energy to escape into the atmosphere. Saltwater has another substance dissolved in it (salt), so its particles attach themselves to the water molecules, making them heavier and in need of more energy. Will A Glass Of Water Evaporate.