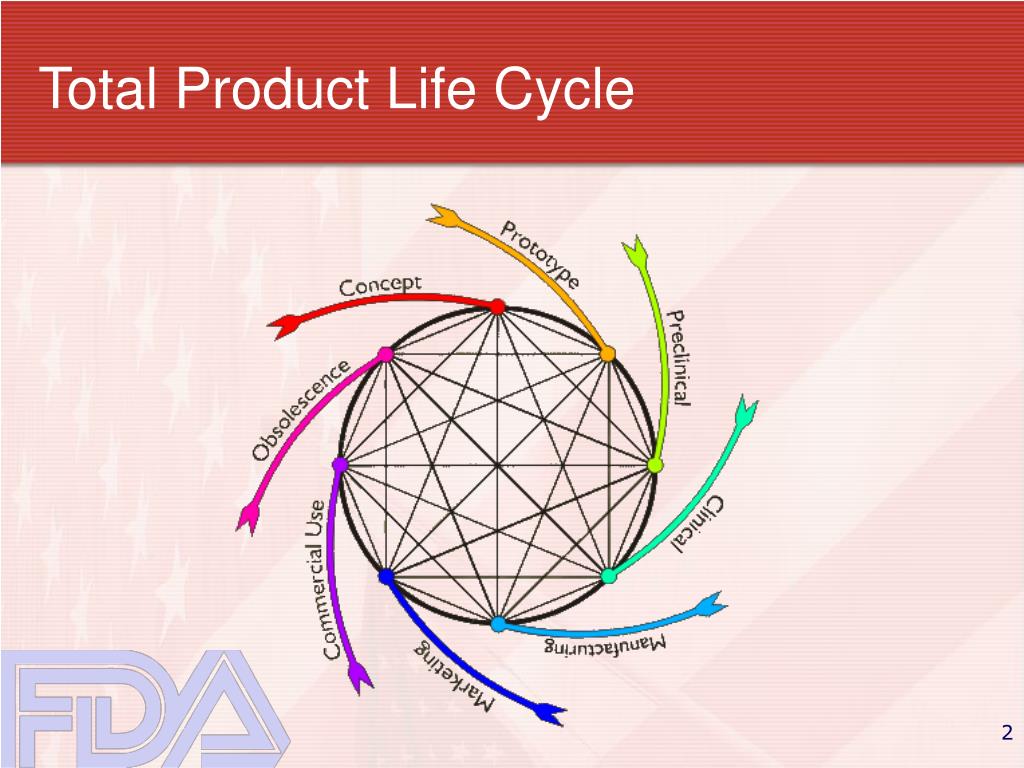
Fda Guidance Infusion Pumps Total Product Life Cycle . Entitled, ‘‘infusion pumps total product life cycle; This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Infusion pumps total product life cycle: Guidance for industry and fda staff. As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. Guidance for industry and fda staff.’’ the recommendations in this guidance. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Food and drug administration (fda) issue.
from www.slideserve.com
This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Entitled, ‘‘infusion pumps total product life cycle; As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. Guidance for industry and fda staff. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Infusion pumps total product life cycle: Guidance for industry and fda staff.’’ the recommendations in this guidance. Food and drug administration (fda) issue.
PPT Overview of Device Regulations PowerPoint Presentation, free
Fda Guidance Infusion Pumps Total Product Life Cycle Guidance for industry and fda staff. Guidance for industry and fda staff. Entitled, ‘‘infusion pumps total product life cycle; This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. Infusion pumps total product life cycle: The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Food and drug administration (fda) issue. Guidance for industry and fda staff.’’ the recommendations in this guidance.
From www.slideshare.net
Applying U.S. FDA Laws and Regulations to Each Phase of Total Product Fda Guidance Infusion Pumps Total Product Life Cycle Guidance for industry and fda staff.’’ the recommendations in this guidance. As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Guidance for industry and fda staff. Food and. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.slideshare.net
Applying U.S. FDA Laws and Regulations to Each Phase of Total Product Fda Guidance Infusion Pumps Total Product Life Cycle Guidance for industry and fda staff. Food and drug administration (fda) issue. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. The total product life cycle (tplc) approach promotes and enhances the transparency,. Fda Guidance Infusion Pumps Total Product Life Cycle.
From mdic-spi.org
MDIC Fda Guidance Infusion Pumps Total Product Life Cycle Guidance for industry and fda staff.’’ the recommendations in this guidance. Guidance for industry and fda staff. As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Infusion. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.regdesk.co
FDA Guidance on Infusion Pumps Performance Testing RegDesk Fda Guidance Infusion Pumps Total Product Life Cycle Guidance for industry and fda staff.’’ the recommendations in this guidance. Food and drug administration (fda) issue. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Entitled, ‘‘infusion pumps total product life cycle; Infusion pumps total product life cycle: Guidance for industry and fda staff. The total product life cycle (tplc). Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.slideserve.com
PPT FDA Perspective on Cardiovascular Device Development PowerPoint Fda Guidance Infusion Pumps Total Product Life Cycle Infusion pumps total product life cycle: Guidance for industry and fda staff. Entitled, ‘‘infusion pumps total product life cycle; Food and drug administration (fda) issue. Guidance for industry and fda staff.’’ the recommendations in this guidance. As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion.. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.regdesk.co
FDA Guidance on Infusion Pumps Hazard Analysis RegDesk Fda Guidance Infusion Pumps Total Product Life Cycle Food and drug administration (fda) issue. Entitled, ‘‘infusion pumps total product life cycle; Guidance for industry and fda staff. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.slideserve.com
PPT CDRH Vision Total Product Life Cycle PowerPoint Presentation Fda Guidance Infusion Pumps Total Product Life Cycle This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Infusion pumps total product life cycle: Food and drug administration (fda) issue. As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. The total product life cycle (tplc). Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.regdesk.co
FDA Guidance on Infusion Pumps Overview RegDesk Fda Guidance Infusion Pumps Total Product Life Cycle This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Guidance for industry and fda staff. Food and drug administration (fda) issue. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps. Fda Guidance Infusion Pumps Total Product Life Cycle.
From medhealthreview.com
FDA Starts A New TPLC, Total Product Life Cycle Advisory Pilot to Fda Guidance Infusion Pumps Total Product Life Cycle This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Entitled, ‘‘infusion pumps total product life cycle; Food and drug administration (fda) issue. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. The total product life cycle (tplc) approach promotes and enhances the. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.hoganlovells.com
Total Product Life Cycle Fda Guidance Infusion Pumps Total Product Life Cycle Guidance for industry and fda staff.’’ the recommendations in this guidance. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. As a first step, fda is issuing and requesting public comment on a new,. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.regdesk.co
FDA Guidance on Infusion Pumps Safety Assurance Cases RegDesk Fda Guidance Infusion Pumps Total Product Life Cycle The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Entitled, ‘‘infusion pumps total product life cycle; This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Guidance for industry and fda staff. Infusion pumps total product life cycle: Food and drug administration (fda) issue.. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.regdesk.co
FDA Guidance on Infusion Pumps Device Description RegDesk Fda Guidance Infusion Pumps Total Product Life Cycle This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Infusion pumps total product life cycle: Food and drug administration (fda) issue. Guidance for industry and fda staff. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. As a first step, fda is issuing. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.scribd.com
Infusion Pumps Total Product Life Cycle Guidance For Industry and FDA Fda Guidance Infusion Pumps Total Product Life Cycle Guidance for industry and fda staff. Food and drug administration (fda) issue. Infusion pumps total product life cycle: This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Guidance for industry and fda staff.’’. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.regdesk.co
FDA Guidance on Infusion Pumps Overview RegDesk Fda Guidance Infusion Pumps Total Product Life Cycle This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Guidance for industry and fda staff. Entitled, ‘‘infusion pumps total product life cycle; Guidance for industry and fda staff.’’ the recommendations in this guidance. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes.. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.regdesk.co
FDA Guidance on Infusion Pumps Hazard Analysis RegDesk Fda Guidance Infusion Pumps Total Product Life Cycle Infusion pumps total product life cycle: As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Guidance for industry and fda staff. Food and drug administration (fda) issue.. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.thepharmamarketer.com
Fda Guidance Infusion Pumps Total Product Life Cycle This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion.. Fda Guidance Infusion Pumps Total Product Life Cycle.
From ar.inspiredpencil.com
Intravenous Infusion Pump Fda Guidance Infusion Pumps Total Product Life Cycle Entitled, ‘‘infusion pumps total product life cycle; Guidance for industry and fda staff. Infusion pumps total product life cycle: As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. Guidance for industry and fda staff.’’ the recommendations in this guidance. Food and drug administration (fda) issue.. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.scribd.com
FDA Guidance On Infusion Pumps Overview RegDesk PDF Medical Fda Guidance Infusion Pumps Total Product Life Cycle This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Infusion pumps total product life cycle: As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. Food and drug administration (fda) issue. Guidance for industry and fda staff.. Fda Guidance Infusion Pumps Total Product Life Cycle.
From xplore-healthcare.com
Product Management Xplore Healthcare Fda Guidance Infusion Pumps Total Product Life Cycle Infusion pumps total product life cycle: Guidance for industry and fda staff.’’ the recommendations in this guidance. Entitled, ‘‘infusion pumps total product life cycle; Guidance for industry and fda staff. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. This document is applicable to syringe pumps, container pumps, and volumetric infusion. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.kmcsystems.com
KMC Medical Device Development Medical Product Lifecycle Management Fda Guidance Infusion Pumps Total Product Life Cycle Infusion pumps total product life cycle: Guidance for industry and fda staff.’’ the recommendations in this guidance. Food and drug administration (fda) issue. Entitled, ‘‘infusion pumps total product life cycle; The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.statonellc.com
Stat One FDA CDRH Launches the Total Product Life Cycle Fda Guidance Infusion Pumps Total Product Life Cycle As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Food. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.smartsheet.com
Ultimate Product Life Cycle Management Guide Smartsheet Fda Guidance Infusion Pumps Total Product Life Cycle This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”.. Fda Guidance Infusion Pumps Total Product Life Cycle.
From jamanetwork.com
Characteristics of Clinical Studies Conducted Over the Total Product Fda Guidance Infusion Pumps Total Product Life Cycle Infusion pumps total product life cycle: Food and drug administration (fda) issue. Guidance for industry and fda staff.’’ the recommendations in this guidance. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Entitled, ‘‘infusion pumps total product life cycle; This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps. Fda Guidance Infusion Pumps Total Product Life Cycle.
From studylib.net
080210_iflow_infusion_pumps Fda Guidance Infusion Pumps Total Product Life Cycle Guidance for industry and fda staff. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Food and drug administration (fda) issue. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. As a first step, fda is issuing and requesting public comment on. Fda Guidance Infusion Pumps Total Product Life Cycle.
From medicaldeviceacademy.com
TPLC Total Product Life Cycle Report for MPN Medical Device Academy Fda Guidance Infusion Pumps Total Product Life Cycle The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Guidance for industry and fda staff.’’ the recommendations in this guidance. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.regdesk.co
FDA Guidance on Infusion Pumps Safety Assurance Cases RegDesk Fda Guidance Infusion Pumps Total Product Life Cycle Entitled, ‘‘infusion pumps total product life cycle; The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Food and drug administration (fda) issue. Infusion pumps total product life cycle: Guidance for industry and fda staff.’’ the recommendations in this guidance. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.slideserve.com
PPT FDA Perspective on Cardiovascular Device Development PowerPoint Fda Guidance Infusion Pumps Total Product Life Cycle As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. Entitled, ‘‘infusion pumps total product life cycle; Food and drug administration (fda) issue. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Infusion pumps total product life. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.qualitymag.com
The Rise of Total Product Life Cycle 20170901 Quality Magazine Fda Guidance Infusion Pumps Total Product Life Cycle This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Infusion pumps total product life cycle: Entitled, ‘‘infusion pumps total product life cycle; The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Food and drug administration (fda) issue. Guidance for industry and fda staff.. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.slideshare.net
Applying U.S. FDA Laws and Regulations to Each Phase of Total Product Fda Guidance Infusion Pumps Total Product Life Cycle As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. Entitled, ‘‘infusion pumps total product life cycle; Infusion pumps total product life cycle: Guidance for industry and fda staff. Food and drug administration (fda) issue. This document supersedes the “guidance on the content of premarket notification. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.youtube.com
How to use Infusion pump Explained YouTube Fda Guidance Infusion Pumps Total Product Life Cycle This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Infusion pumps total product life cycle: Entitled, ‘‘infusion pumps total product life cycle; Guidance for industry and fda staff.’’ the recommendations in this guidance. Guidance for industry and fda staff. The total product life cycle (tplc) approach promotes and enhances the transparency,. Fda Guidance Infusion Pumps Total Product Life Cycle.
From 54.221.168.112
New FDA Guidance On WiFiManaged Infusion Pumps Is Cybersafe And Fda Guidance Infusion Pumps Total Product Life Cycle This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Guidance for industry and fda staff. Food and drug administration (fda) issue. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. As a first step, fda is issuing and requesting public comment on. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.researchgate.net
The total product life cycle, known as TPLC, represents the FDA’s Fda Guidance Infusion Pumps Total Product Life Cycle Guidance for industry and fda staff. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. As a first step, fda is issuing and requesting public comment on a new, total product life cycle. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.slideserve.com
PPT Overview of Device Regulations PowerPoint Presentation, free Fda Guidance Infusion Pumps Total Product Life Cycle As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Food. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.researchgate.net
Total product life cycle (From US Food and Drug Administration. Medical Fda Guidance Infusion Pumps Total Product Life Cycle As a first step, fda is issuing and requesting public comment on a new, total product life cycle (tplc) draft guidance document for infusion. The total product life cycle (tplc) approach promotes and enhances the transparency, efficiency, and agility of the. Infusion pumps total product life cycle: Food and drug administration (fda) issue. Guidance for industry and fda staff. This. Fda Guidance Infusion Pumps Total Product Life Cycle.
From www.researchgate.net
The total product life cycle, known as TPLC, represents the FDA’s Fda Guidance Infusion Pumps Total Product Life Cycle This document is applicable to syringe pumps, container pumps, and volumetric infusion pumps for all indicated delivery modes. Entitled, ‘‘infusion pumps total product life cycle; This document supersedes the “guidance on the content of premarket notification [510(k)] submissions for external infusion pumps, ”. Guidance for industry and fda staff.’’ the recommendations in this guidance. Infusion pumps total product life cycle:. Fda Guidance Infusion Pumps Total Product Life Cycle.