Chemistry Lab Density Of Liquids And Solids . To measure the density of a liquid, first measure the. Compare density of liquids and solids. the units of density are commonly expressed as g/cm 3 for solids, g/ml for liquids, and g/l for gases. you will be presented with actual masses and volume measurements of an unknown liquid and of a solid in order to calculate. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. the mass of a liquid or solid can be measured using a balance. Density of liquids and solids lab procedure; Explore the importance of recording data to the proper significant figures and. — santa monica college. — measure mass and volume of liquids and solids. — the s.i. the density of liquids and solids. for example, the density of liquid water is higher than that of water vapor (steam) because the water molecules are much. determine the density of solids and liquids. The density of liquids and solids, online chemistry lab manual.
from www.priyamstudycentre.com
To measure the density of a liquid, first measure the. density of solids and liquids is usually expressed in units of g/ml or g/cm3. the mass of a liquid or solid can be measured using a balance. — measure mass and volume of liquids and solids. density can be determined in the laboratory by measuring the mass and volume of a substance. the units of density are commonly expressed as g/cm3 for solids, g/ml for liquids, and g/l for gases. In order to experimentally determine the density of. The density of liquids and solids, online chemistry lab manual. — santa monica college. Explore the importance of recording data to the proper significant figures and.
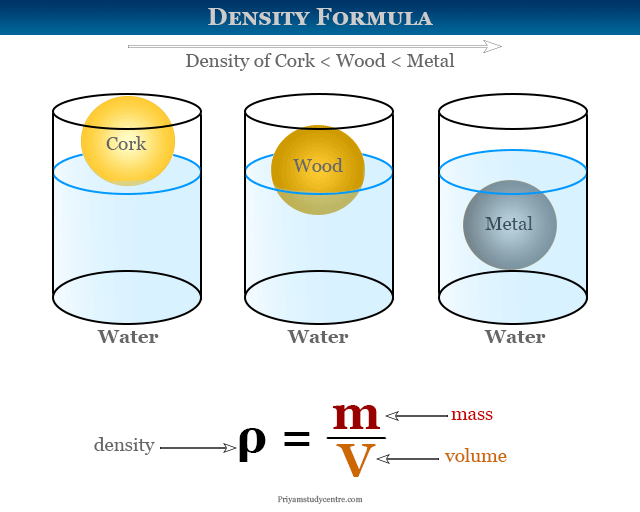
Density Formula, Definition, Measurement, Calculation
Chemistry Lab Density Of Liquids And Solids — the s.i. — put simply, solid masses have a high density as particles are very close together, liquid masses are slightly less. Compare density of liquids and solids. In order to experimentally determine the density of. — the units of density are commonly expressed as g/cm 3 for solids, g/ml for liquids, and g/l for gases. the units of density are commonly expressed as g/cm 3 for solids, g/ml for liquids, and g/l for gases. you will be presented with actual masses and volume measurements of an unknown liquid and of a solid in order to calculate. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. Defined as the ratio of mass to volume, often reported in terms of g/ml or. To measure the density of a liquid, first measure the. Explore the importance of recording data to the proper significant figures and. — santa monica college. the density of liquids and solids. the mass of a liquid or solid can be measured using a balance. Unit of density is kg/m 3, but in chemistry it is more often expressed in units of g/cm 3 for solids, and g/ml for. interact with blocks of different materials, including a custom option by modifying their mass and volume, to explore the effect on.
From www.studocu.com
Density of Solids (Lab Report) Density of Solids Lab Introduction The Chemistry Lab Density Of Liquids And Solids — determine the density of several liquids and solids. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. To measure the density of a liquid, first measure the. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. The density of liquids and solids,. Chemistry Lab Density Of Liquids And Solids.
From www.chegg.com
Solved Lab Report The Density of Liquids and Solids Part A Chemistry Lab Density Of Liquids And Solids — put simply, solid masses have a high density as particles are very close together, liquid masses are slightly less. To use standard laboratory measurement devices to measure length,. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. density can be determined in the laboratory by measuring the mass and. Chemistry Lab Density Of Liquids And Solids.
From ar.inspiredpencil.com
Density Of Liquids Chart Chemistry Lab Density Of Liquids And Solids Explore the importance of recording data to the proper significant figures and. Reinforce skills from experiment 1 (measurement of mass and temperature) understand. — prelab assignment: the mass of a liquid or solid can be measured using a balance. — determine the density of several liquids and solids. the densities of solids and liquids change slightly. Chemistry Lab Density Of Liquids And Solids.
From charlotteanderson.z13.web.core.windows.net
Density Chart For Liquids Chemistry Lab Density Of Liquids And Solids the units of density are commonly expressed as g/cm3 for solids, g/ml for liquids, and g/l for gases. In order to experimentally determine the density of. — put simply, solid masses have a high density as particles are very close together, liquid masses are slightly less. the densities of solids and liquids change slightly with temperature, in. Chemistry Lab Density Of Liquids And Solids.
From www.chegg.com
Solved Lab Report The Density of Liquids and Solids Part A Chemistry Lab Density Of Liquids And Solids Reinforce skills from experiment 1 (measurement of mass and temperature) understand. In order to experimentally determine the density of. Unit of density is kg/m 3, but in chemistry it is more often expressed in units of g/cm 3 for solids, and g/ml for. — prelab assignment: Density of liquids and solids lab procedure; Compare density of liquids and solids.. Chemistry Lab Density Of Liquids And Solids.
From www.studocu.com
Answer Sheet Density of Solids 1 EXPERIMENT 1A DENSITY OF A SOLID Chemistry Lab Density Of Liquids And Solids the density of liquids and solids. There are 2 kinds of balances in the general chemistry. — prelab assignment: Reinforce skills from experiment 1 (measurement of mass and temperature) understand. for example, the density of liquid water is higher than that of water vapor (steam) because the water molecules are much. — measure mass and volume. Chemistry Lab Density Of Liquids And Solids.
From studylib.net
Density lab 2012 Chemistry Lab Density Of Liquids And Solids the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. interact with blocks of different materials, including a custom option by modifying their mass and volume, to explore the effect on. Identify an unknown metal by calculating its density. Defined as the ratio of mass to volume, often reported in terms of. Chemistry Lab Density Of Liquids And Solids.
From www.youtube.com
Liquid Density Experiment Easily Conduct this Experiment at Home Chemistry Lab Density Of Liquids And Solids Unit of density is kg/m 3, but in chemistry it is more often expressed in units of g/cm 3 for solids, and g/ml for. There are 2 kinds of balances in the general chemistry. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. you will be presented with actual masses and. Chemistry Lab Density Of Liquids And Solids.
From www.chegg.com
Solved Advance Study Assignment Densities of Solids and Chemistry Lab Density Of Liquids And Solids the units of density are commonly expressed as g/cm 3 for solids, g/ml for liquids, and g/l for gases. — prelab assignment: In order to experimentally determine the density of. density can be determined in the laboratory by measuring the mass and volume of a substance. interact with blocks of different materials, including a custom option. Chemistry Lab Density Of Liquids And Solids.
From www.dreamstime.com
Density of Matter with Gas, Liquid and Solid Particle States Outline Chemistry Lab Density Of Liquids And Solids for example, the density of liquid water is higher than that of water vapor (steam) because the water molecules are much. — santa monica college. Identify an unknown metal by calculating its density. Unit of density is kg/m 3, but in chemistry it is more often expressed in units of g/cm 3 for solids, and g/ml for. To. Chemistry Lab Density Of Liquids And Solids.
From syzokigyxavusut.modellervefiyatlar.com
Solid And Liquid Density Lab Report Chemistry Lab Density Of Liquids And Solids Density of liquids and solids lab procedure; — santa monica college. To use standard laboratory measurement devices to measure length,. — determine the density of several liquids and solids. — the units of density are commonly expressed as g/cm 3 for solids, g/ml for liquids, and g/l for gases. determine the density of solids and liquids.. Chemistry Lab Density Of Liquids And Solids.
From studylib.net
Volume and Density of Solids and Liquids Chemistry Lab Density Of Liquids And Solids density can be determined in the laboratory by measuring the mass and volume of a substance. Defined as the ratio of mass to volume, often reported in terms of g/ml or. In order to experimentally determine the density of. The density of liquids and solids, online chemistry lab manual. There are 2 kinds of balances in the general chemistry.. Chemistry Lab Density Of Liquids And Solids.
From ar.inspiredpencil.com
Density Of Liquids And Solids Chemistry Lab Density Of Liquids And Solids — prelab assignment: the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. Reinforce skills from experiment 1 (measurement of mass and temperature) understand. you will be presented with actual masses and volume measurements of an unknown liquid and of a solid in order to calculate. Unit of density is kg/m. Chemistry Lab Density Of Liquids And Solids.
From www.chegg.com
Solved Lab Report The Density of Liquids and Solids Part A Chemistry Lab Density Of Liquids And Solids density of solids and liquids is usually expressed in units of g/ml or g/cm3. you will be presented with actual masses and volume measurements of an unknown liquid and of a solid in order to calculate. interact with blocks of different materials, including a custom option by modifying their mass and volume, to explore the effect on.. Chemistry Lab Density Of Liquids And Solids.
From www.chegg.com
Solved Density Lab Getting Started Open The Beyond Labz S... Chemistry Lab Density Of Liquids And Solids The density of liquids and solids, online chemistry lab manual. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. you will be presented with actual masses and volume measurements of an unknown liquid and of a solid in order to calculate. interact with blocks of different materials, including a custom. Chemistry Lab Density Of Liquids And Solids.
From www.chegg.com
Density of Liquids and Solids Lab Overview The Chemistry Lab Density Of Liquids And Solids interact with blocks of different materials, including a custom option by modifying their mass and volume, to explore the effect on. the units of density are commonly expressed as g/cm3 for solids, g/ml for liquids, and g/l for gases. Identify an unknown metal by calculating its density. Compare density of liquids and solids. the density of liquids. Chemistry Lab Density Of Liquids And Solids.
From www.priyamstudycentre.com
Density Formula, Definition, Measurement, Calculation Chemistry Lab Density Of Liquids And Solids the mass of a liquid or solid can be measured using a balance. Reinforce skills from experiment 1 (measurement of mass and temperature) understand. Unit of density is kg/m 3, but in chemistry it is more often expressed in units of g/cm 3 for solids, and g/ml for. — the units of density are commonly expressed as g/cm. Chemistry Lab Density Of Liquids And Solids.
From www.teachoo.com
The mass per unit volume of a substance is called density. Arrange in Chemistry Lab Density Of Liquids And Solids the mass of a liquid or solid can be measured using a balance. — santa monica college. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. — the units of density are commonly expressed as g/cm 3 for solids, g/ml for liquids, and g/l for gases. To use standard. Chemistry Lab Density Of Liquids And Solids.
From www.youtube.com
Experiment 1 Density of Liquids and Solids SMU Chemistry YouTube Chemistry Lab Density Of Liquids And Solids you will be presented with actual masses and volume measurements of an unknown liquid and of a solid in order to calculate. Compare density of liquids and solids. density can be determined in the laboratory by measuring the mass and volume of a substance. density of solids and liquids is usually expressed in units of g/ml or. Chemistry Lab Density Of Liquids And Solids.
From www.scribd.com
Lab Report 2 Density of a Solid Chemistry Lab Density Of Liquids And Solids Unit of density is kg/m 3, but in chemistry it is more often expressed in units of g/cm 3 for solids, and g/ml for. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. To measure the density of a liquid, first measure the. In order to experimentally determine the density of. . Chemistry Lab Density Of Liquids And Solids.
From postonphysicalscience.weebly.com
Density Physical Science Chemistry Lab Density Of Liquids And Solids To measure the density of a liquid, first measure the. The density of liquids and solids, online chemistry lab manual. Defined as the ratio of mass to volume, often reported in terms of g/ml or. Reinforce skills from experiment 1 (measurement of mass and temperature) understand. There are 2 kinds of balances in the general chemistry. you will be. Chemistry Lab Density Of Liquids And Solids.
From www.studocu.com
Experiment 1 Density It's the lab assignment Experiment 1 Density Chemistry Lab Density Of Liquids And Solids In order to experimentally determine the density of. the units of density are commonly expressed as g/cm3 for solids, g/ml for liquids, and g/l for gases. Explore the importance of recording data to the proper significant figures and. interact with blocks of different materials, including a custom option by modifying their mass and volume, to explore the effect. Chemistry Lab Density Of Liquids And Solids.
From studymoose.com
Density of Liquids and Solids Experiment Report StudyMoose Chemistry Lab Density Of Liquids And Solids for example, the density of liquid water is higher than that of water vapor (steam) because the water molecules are much. — determine the density of several liquids and solids. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. In order to experimentally determine the density of. interact with. Chemistry Lab Density Of Liquids And Solids.
From ar.inspiredpencil.com
Density Of Solids Experiment Chemistry Lab Density Of Liquids And Solids — santa monica college. the mass of a liquid or solid can be measured using a balance. — measure mass and volume of liquids and solids. Unit of density is kg/m 3, but in chemistry it is more often expressed in units of g/cm 3 for solids, and g/ml for. — the units of density are. Chemistry Lab Density Of Liquids And Solids.
From www.animalia-life.club
Density Of Liquids Lab Chemistry Lab Density Of Liquids And Solids Reinforce skills from experiment 1 (measurement of mass and temperature) understand. To use standard laboratory measurement devices to measure length,. In order to experimentally determine the density of. Unit of density is kg/m 3, but in chemistry it is more often expressed in units of g/cm 3 for solids, and g/ml for. Defined as the ratio of mass to volume,. Chemistry Lab Density Of Liquids And Solids.
From materialmcgheesappier.z21.web.core.windows.net
Experiment Of Density Of Water Chemistry Lab Density Of Liquids And Solids determine the density of solids and liquids. — put simply, solid masses have a high density as particles are very close together, liquid masses are slightly less. — the s.i. the units of density are commonly expressed as g/cm3 for solids, g/ml for liquids, and g/l for gases. The density of liquids and solids, online chemistry. Chemistry Lab Density Of Liquids And Solids.
From www.chegg.com
Density of Liquids and Solids Lab Overview The Chemistry Lab Density Of Liquids And Solids — put simply, solid masses have a high density as particles are very close together, liquid masses are slightly less. Reinforce skills from experiment 1 (measurement of mass and temperature) understand. determine the density of solids and liquids. density can be determined in the laboratory by measuring the mass and volume of a substance. The density of. Chemistry Lab Density Of Liquids And Solids.
From sayngon.com
Are Liquids And Gases Exploring Density Similarities Chemistry Lab Density Of Liquids And Solids In order to experimentally determine the density of. for example, the density of liquid water is higher than that of water vapor (steam) because the water molecules are much. The density of liquids and solids, online chemistry lab manual. To use standard laboratory measurement devices to measure length,. the units of density are commonly expressed as g/cm3 for. Chemistry Lab Density Of Liquids And Solids.
From studylib.net
chemistry lab density of solutions Chemistry Lab Density Of Liquids And Solids — the s.i. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. determine the density of solids and liquids. Unit of density is kg/m 3, but in chemistry it is more often expressed. Chemistry Lab Density Of Liquids And Solids.
From studylib.net
DENSITY of SOLIDS and LIQUIDS LAB Chemistry Lab Density Of Liquids And Solids you will be presented with actual masses and volume measurements of an unknown liquid and of a solid in order to calculate. the densities of solids and liquids change slightly with temperature, in general, decreasing with increasing temperature. — the units of density are commonly expressed as g/cm 3 for solids, g/ml for liquids, and g/l for. Chemistry Lab Density Of Liquids And Solids.
From www.vecteezy.com
Liquid density scientific experiment concept. Separate fluid layers Chemistry Lab Density Of Liquids And Solids the density of liquids and solids. Compare density of liquids and solids. — the units of density are commonly expressed as g/cm 3 for solids, g/ml for liquids, and g/l for gases. the units of density are commonly expressed as g/cm3 for solids, g/ml for liquids, and g/l for gases. The density of liquids and solids, online. Chemistry Lab Density Of Liquids And Solids.
From www.dreamstime.com
Density of Liquids. Laboratory Experiment Stock Vector Illustration Chemistry Lab Density Of Liquids And Solids for example, the density of liquid water is higher than that of water vapor (steam) because the water molecules are much. you will be presented with actual masses and volume measurements of an unknown liquid and of a solid in order to calculate. density of solids and liquids is usually expressed in units of g/ml or g/cm3.. Chemistry Lab Density Of Liquids And Solids.
From chem.libretexts.org
2 The Density of Liquids and Solids (Experiment) Chemistry LibreTexts Chemistry Lab Density Of Liquids And Solids Density of liquids and solids lab procedure; Defined as the ratio of mass to volume, often reported in terms of g/ml or. — prelab assignment: density of solids and liquids is usually expressed in units of g/ml or g/cm3. the units of density are commonly expressed as g/cm 3 for solids, g/ml for liquids, and g/l for. Chemistry Lab Density Of Liquids And Solids.
From ar.inspiredpencil.com
Density Of Liquids For Kids Chemistry Lab Density Of Liquids And Solids Explore the importance of recording data to the proper significant figures and. determine the density of solids and liquids. — put simply, solid masses have a high density as particles are very close together, liquid masses are slightly less. the units of density are commonly expressed as g/cm3 for solids, g/ml for liquids, and g/l for gases.. Chemistry Lab Density Of Liquids And Solids.
From www.chegg.com
Solved Post Lab Activity Density of Solids and Liquids 1. A Chemistry Lab Density Of Liquids And Solids — santa monica college. Defined as the ratio of mass to volume, often reported in terms of g/ml or. Identify an unknown metal by calculating its density. To use standard laboratory measurement devices to measure length,. — prelab assignment: There are 2 kinds of balances in the general chemistry. The density of liquids and solids, online chemistry lab. Chemistry Lab Density Of Liquids And Solids.