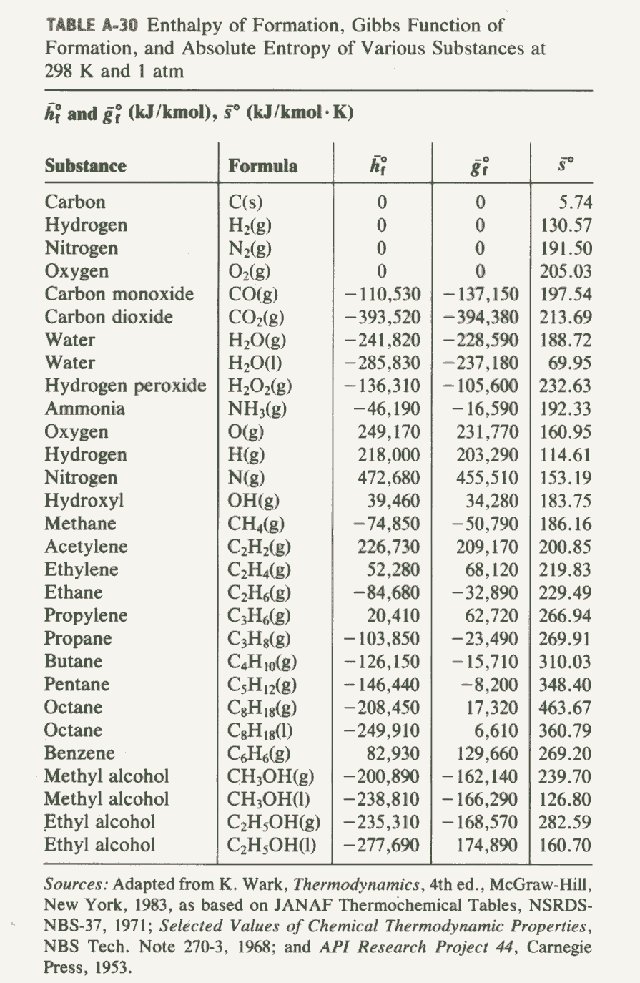
Standard Enthalpy Of Formation And Reaction . The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. standard enthalpies of formation.
from dxouglhlh.blob.core.windows.net
standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. standard enthalpies of formation. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a.
Standard Enthalpy Of Formation Mg at David Leon blog
Standard Enthalpy Of Formation And Reaction a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. standard enthalpies of formation. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is.
From mavink.com
Enthalpy Of Formation Equation Standard Enthalpy Of Formation And Reaction standard enthalpies of formation. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. The magnitude of δ h for a reaction depends on the. Standard Enthalpy Of Formation And Reaction.
From www.youtube.com
5.1 Standard enthalpy changes of formation and combustion YouTube Standard Enthalpy Of Formation And Reaction a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation. Standard Enthalpy Of Formation And Reaction.
From www.slideshare.net
Tang 03 enthalpy of formation and combustion Standard Enthalpy Of Formation And Reaction The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. standard enthalpy of formation is the enthalpy change when. Standard Enthalpy Of Formation And Reaction.
From zeviernswenson.blogspot.com
Standard Enthalpy of Formation ZeviernSwenson Standard Enthalpy Of Formation And Reaction standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. The magnitude of δ h for a reaction depends on the physical states. Standard Enthalpy Of Formation And Reaction.
From pdfprof.com
enthalpies standard de formation et entropie standard Standard Enthalpy Of Formation And Reaction The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. standard enthalpy of formation is the enthalpy change when. Standard Enthalpy Of Formation And Reaction.
From proper-cooking.info
Enthalpy Of Reaction Table Standard Enthalpy Of Formation And Reaction The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. standard enthalpies of formation. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. a standard enthalpy of formation $δh°_f$ is an. Standard Enthalpy Of Formation And Reaction.
From www.youtube.com
Enthalpies of Formation Chemsitry Tutorial YouTube Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. standard enthalpies of formation. the standard enthalpy of formation is a measure. Standard Enthalpy Of Formation And Reaction.
From www.researchgate.net
Enthalpies of formation for stable and radical species used in work Standard Enthalpy Of Formation And Reaction standard enthalpies of formation. standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. a standard enthalpy of formation $δh°_f$ is an enthalpy change for. Standard Enthalpy Of Formation And Reaction.
From studylib.net
Standard Enthalpy of Formation and Reaction Standard Enthalpy Of Formation And Reaction a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation is the enthalpy change when one mole of. Standard Enthalpy Of Formation And Reaction.
From www.youtube.com
CHEMISTRY 101 Standard Enthalpy of reaction from Standard Enthalpies Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. the standard enthalpy of formation, also known as the heat of formation, is. Standard Enthalpy Of Formation And Reaction.
From jordanhumphries.z13.web.core.windows.net
Enthalpy Of Formation Chart Standard Enthalpy Of Formation And Reaction standard enthalpies of formation. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. The magnitude of δ h for a reaction depends on the. Standard Enthalpy Of Formation And Reaction.
From classnotes.org.in
Enthalpies Of Reaction Chemistry, Class 11, Thermodynamics Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly. Standard Enthalpy Of Formation And Reaction.
From www.youtube.com
CHEM 101 Using Standard Enthalpies of Formation and Standard Enthalpy Standard Enthalpy Of Formation And Reaction standard enthalpies of formation. standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. the standard enthalpy of formation is a. Standard Enthalpy Of Formation And Reaction.
From stahonorschemistry.weebly.com
III Calculating Enthalpies STA Form IV Honors Chemistry Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpies of formation. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The magnitude of δ h for a reaction. Standard Enthalpy Of Formation And Reaction.
From mavink.com
Enthalpy Of Formation Equation Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. the standard enthalpy of formation, δh of, is the enthalpy change for. Standard Enthalpy Of Formation And Reaction.
From chemistry.stackexchange.com
thermodynamics Calculating Enthalpy of formation versus Calculating Standard Enthalpy Of Formation And Reaction a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. standard enthalpy of formation is the enthalpy change when one mole of. Standard Enthalpy Of Formation And Reaction.
From schoolworkhelper.net
StandardEnthalpiesFormation Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. standard enthalpies of formation. the standard enthalpy. Standard Enthalpy Of Formation And Reaction.
From dxorvtvqp.blob.core.windows.net
Standard Enthalpy Of Formation Gibbsite at Samuel Speed blog Standard Enthalpy Of Formation And Reaction standard enthalpies of formation. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. the standard enthalpy of formation, δh of, is the enthalpy. Standard Enthalpy Of Formation And Reaction.
From hxejgsfjx.blob.core.windows.net
Standard Enthalpy Of Formation And Reaction Hess Law at Brian Cannon blog Standard Enthalpy Of Formation And Reaction a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. the standard enthalpy of formation, also known as the heat of formation, is the. Standard Enthalpy Of Formation And Reaction.
From mungfali.com
Standard Enthalpy Of Formation Equation Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change. Standard Enthalpy Of Formation And Reaction.
From pediaa.com
What is the Difference Between Enthalpy of Formation and Enthalpy of Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. standard enthalpies of formation. The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. standard enthalpy of formation is the enthalpy change. Standard Enthalpy Of Formation And Reaction.
From ar.inspiredpencil.com
Enthalpy Of Reaction Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation. Standard Enthalpy Of Formation And Reaction.
From www.chem.fsu.edu
CHM1045 Enthalpy Lecture Standard Enthalpy Of Formation And Reaction a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpy of formation is the enthalpy change when one mole of any compound is. Standard Enthalpy Of Formation And Reaction.
From exokycsnc.blob.core.windows.net
Standard Enthalpy Of Formation Table Elements at Filomena Gilbert blog Standard Enthalpy Of Formation And Reaction a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. standard enthalpies of formation. standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. the standard enthalpy of formation, δh of, is the enthalpy change for a. Standard Enthalpy Of Formation And Reaction.
From www.youtube.com
Enthalpies of Reactions Using Average Bond Enthalpies Chemistry Standard Enthalpy Of Formation And Reaction standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. standard enthalpies of formation. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. the standard enthalpy of formation, also known as the heat. Standard Enthalpy Of Formation And Reaction.
From dxouglhlh.blob.core.windows.net
Standard Enthalpy Of Formation Mg at David Leon blog Standard Enthalpy Of Formation And Reaction standard enthalpies of formation. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. the standard enthalpy of formation is. Standard Enthalpy Of Formation And Reaction.
From general.chemistrysteps.com
Standard Enthalpies of Formation Chemistry Steps Standard Enthalpy Of Formation And Reaction standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. The magnitude of δ h for a reaction depends on the physical states of the reactants and. Standard Enthalpy Of Formation And Reaction.
From hxejgsfjx.blob.core.windows.net
Standard Enthalpy Of Formation And Reaction Hess Law at Brian Cannon blog Standard Enthalpy Of Formation And Reaction The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. standard enthalpies of formation. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. a standard enthalpy of formation $δh°_f$ is an. Standard Enthalpy Of Formation And Reaction.
From www.youtube.com
Standard Enthalpy of Formation and Formation Reactions OpenStax Standard Enthalpy Of Formation And Reaction a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. the standard enthalpy of formation, δh of, is. Standard Enthalpy Of Formation And Reaction.
From people.chem.umass.edu
to Adobe GoLive 6 Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. the standard enthalpy of formation is a measure of. Standard Enthalpy Of Formation And Reaction.
From www.chegg.com
Solved Use a standard enthalpies of formation table to Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a reaction in which exactly 1 mole of a. The magnitude of δ h for a reaction depends on the physical. Standard Enthalpy Of Formation And Reaction.
From www.slideserve.com
PPT Enthalpy of Formation PowerPoint Presentation, free download ID Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation, δh of, is the enthalpy change for a formation equation when all substances are in their standard. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a. Standard Enthalpy Of Formation And Reaction.
From www.writework.com
A comparison between the Enthalpy of formation of MgO acquired via a Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. a standard enthalpy of formation $δh°_f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. a standard enthalpy of formation \(δh^\circ_\ce{f}\) is an enthalpy change for a. Standard Enthalpy Of Formation And Reaction.
From hxejgsfjx.blob.core.windows.net
Standard Enthalpy Of Formation And Reaction Hess Law at Brian Cannon blog Standard Enthalpy Of Formation And Reaction standard enthalpy of formation is the enthalpy change when one mole of any compound is formed from its constituent. the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. the standard enthalpy of formation is a measure of the energy released. Standard Enthalpy Of Formation And Reaction.
From www.slideserve.com
PPT Standard Enthalpies of Formation PowerPoint Presentation, free Standard Enthalpy Of Formation And Reaction the standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its. The magnitude of δ h for a reaction depends on the physical states of the reactants and the products (gas, liquid, solid, or. the standard enthalpy of formation is a measure of. Standard Enthalpy Of Formation And Reaction.