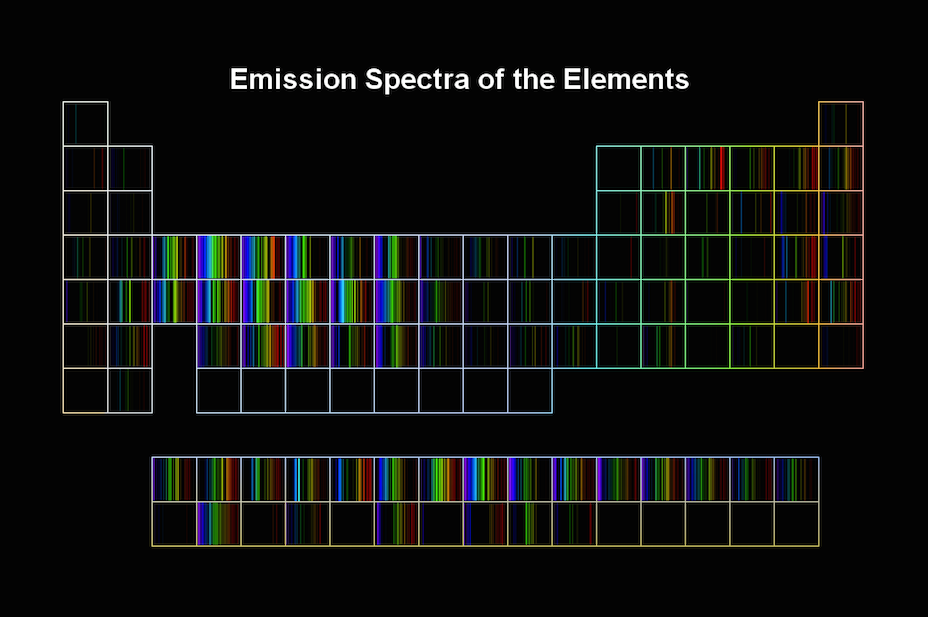
Emission Spectra Of Elements Periodic Table . The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. The emission spectra of various atoms. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. Select an element to access data. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: When hydrogen gas is placed.
from mrsmorrittscience.weebly.com
This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. The emission spectra of various atoms. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: When hydrogen gas is placed. Select an element to access data.
Emission Spectra of the Elements Arranged on the Periodic Table Mrs
Emission Spectra Of Elements Periodic Table In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. When hydrogen gas is placed. The emission spectra of various atoms. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: Select an element to access data. The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity.
From www.reddit.com
Emission Spectra of the Elements in Periodic Table Format r/chemistry Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in. Emission Spectra Of Elements Periodic Table.
From umop.net
Visible Spectra of the Elements Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: The emission spectra of various atoms. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. When hydrogen gas is placed. In the early. Emission Spectra Of Elements Periodic Table.
From brokeasshome.com
periodic table emission spectra Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the. Emission Spectra Of Elements Periodic Table.
From www.dreamstime.com
Elements Emission Spectrum List Lines Visible Light Spectra Stock Emission Spectra Of Elements Periodic Table In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. Select an element to access data. When hydrogen gas is placed. The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. The emission spectra of. Emission Spectra Of Elements Periodic Table.
From brokeasshome.com
periodic table emission spectra Emission Spectra Of Elements Periodic Table This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: When hydrogen gas is placed. The emission spectra of various atoms. The emission spectrum can be used to determine. Emission Spectra Of Elements Periodic Table.
From www.pinterest.com
line spectra chart If the emission lines of the chemical elements Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the. Emission Spectra Of Elements Periodic Table.
From brokeasshome.com
Periodic Table Emission Spectra Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: Select an element to access data. The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. In the early 20 th century, it was believed that. Emission Spectra Of Elements Periodic Table.
From www.reddit.com
Periodic table of each elements emission spectrum with regular periodic Emission Spectra Of Elements Periodic Table The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. When hydrogen gas is placed. Select an element to access data. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. The emission. Emission Spectra Of Elements Periodic Table.
From umop.net
Visible Spectra of the Elements Emission Spectra Of Elements Periodic Table Select an element to access data. The emission spectra of various atoms. When hydrogen gas is placed. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. This handbook. Emission Spectra Of Elements Periodic Table.
From brokeasshome.com
periodic table emission spectra Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: The emission spectra of various atoms. Select an element to access data. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. In the early 20 th century, it. Emission Spectra Of Elements Periodic Table.
From www.umop.net
Visible Spectra of the Elements Emission Spectra Of Elements Periodic Table The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. When hydrogen gas is placed. Select an element to access data. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or. Emission Spectra Of Elements Periodic Table.
From spiff.rit.edu
Spectrographs and Spectra Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. The emission spectra of various atoms. The emission spectrum (or line spectrum) of a chemical element is the unique. Emission Spectra Of Elements Periodic Table.
From www.pinterest.com
emission spectra periodic table Google Search AP Chem 5 Atomic Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. The emission spectra of various atoms. The emission spectrum can be used to determine the composition of a material,. Emission Spectra Of Elements Periodic Table.
From rightmetal.weebly.com
Atomic emission spectrum chemistry definition rightmetal Emission Spectra Of Elements Periodic Table The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. If the emission lines of the chemical elements were observed through a diffraction grating, they. Emission Spectra Of Elements Periodic Table.
From www.vrogue.co
Periodic Table Atomic Emission Spectra 2023 Periodic vrogue.co Emission Spectra Of Elements Periodic Table This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. Select an element to access data. When hydrogen gas is placed. The emission spectra of. Emission Spectra Of Elements Periodic Table.
From ponhadasg8lstudyquizz.z13.web.core.windows.net
Atomic Spectra And Its Types Emission Spectra Of Elements Periodic Table The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. The emission spectrum can be used to determine the composition of a material,. Emission Spectra Of Elements Periodic Table.
From mrsmorrittscience.weebly.com
Emission Spectra of the Elements Arranged on the Periodic Table Mrs Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. The emission spectrum can be used to determine the composition of a material, since. Emission Spectra Of Elements Periodic Table.
From www.periodictableprintable.com
Periodic Table Atomic Emission Spectra 2024 Periodic Table Printable Emission Spectra Of Elements Periodic Table The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: When hydrogen gas is placed. The emission spectrum can be used to determine the. Emission Spectra Of Elements Periodic Table.
From www.resonancescience.org
What is Resonance and Why is it so Important? Emission Spectra Of Elements Periodic Table When hydrogen gas is placed. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. If the emission lines of the chemical elements were observed. Emission Spectra Of Elements Periodic Table.
From www.esa.int
ESA Absorption and emission spectra of various elements Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: The emission spectra of various atoms. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. This handbook is designed to provide a selection. Emission Spectra Of Elements Periodic Table.
From www.pinterest.com
Unique Periodic Table Jlab Pdf Periodic table, Spectrum, Emissions Emission Spectra Of Elements Periodic Table This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. In the early 20 th century, it was believed that the emission spectra. Emission Spectra Of Elements Periodic Table.
From www.visionlearning.com
Atomic Theory II Chemistry Visionlearning Emission Spectra Of Elements Periodic Table When hydrogen gas is placed. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. If the emission lines of the chemical elements were observed. Emission Spectra Of Elements Periodic Table.
From www.meta-synthesis.com
Periodic Table Database Chemogenesis Emission Spectra Of Elements Periodic Table When hydrogen gas is placed. Select an element to access data. The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or. Emission Spectra Of Elements Periodic Table.
From adawyaf.blogspot.com
Chemistry Grade 9, Atomic Emission Spectra , Introduction Emission Spectra Of Elements Periodic Table This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: The emission spectra of various atoms. When hydrogen gas is placed. The emission spectrum (or line spectrum) of a. Emission Spectra Of Elements Periodic Table.
From www.vrogue.co
Periodic Table Atomic Emission Spectrum C Electromagn vrogue.co Emission Spectra Of Elements Periodic Table Select an element to access data. In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. The emission spectra of various atoms. If the emission lines of the. Emission Spectra Of Elements Periodic Table.
From www.pinterest.com
Pin on AP Chem 5 Atomic Structure and Periodicity Emission Spectra Of Elements Periodic Table The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light. Emission Spectra Of Elements Periodic Table.
From store.fieldtestedsystems.com
Periodic Table of Spectra Poster Field Tested Systems Emission Spectra Of Elements Periodic Table Select an element to access data. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: When hydrogen gas is placed. The emission spectra. Emission Spectra Of Elements Periodic Table.
From uwaterloo.ca
Periodic Table of emission spectra Chem 13 News Magazine University Emission Spectra Of Elements Periodic Table The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: When hydrogen gas is placed. The emission spectrum (or line spectrum) of a chemical element is. Emission Spectra Of Elements Periodic Table.
From brokeasshome.com
periodic table emission spectra Emission Spectra Of Elements Periodic Table This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. The emission spectrum (or line spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. The emission spectrum can be used to determine the composition of a material,. Emission Spectra Of Elements Periodic Table.
From chemistrypuns-periodically.weebly.com
Chemistry Electron Emission Spectrum Emission Spectra Of Elements Periodic Table The emission spectra of various atoms. When hydrogen gas is placed. Select an element to access data. In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. This handbook is designed to provide a selection of the most important and frequently used atomic spectroscopic data in an easily. The. Emission Spectra Of Elements Periodic Table.
From www.periodictableprintable.com
Periodic Table Atomic Emission Spectra 2024 Periodic Table Printable Emission Spectra Of Elements Periodic Table If the emission lines of the chemical elements were observed through a diffraction grating, they would look something like this: In the early 20 th century, it was believed that the emission spectra of elements was caused by electrons within the. Select an element to access data. The emission spectra of various atoms. The emission spectrum (or line spectrum) of. Emission Spectra Of Elements Periodic Table.