Standard Heat Of Formation Of Solid Sodium Chloride . standard enthalpies of formation. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific.
from www.numerade.com
standard enthalpies of formation. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. As mentioned on the previous page, using hess' law makes it possible to calculate many.
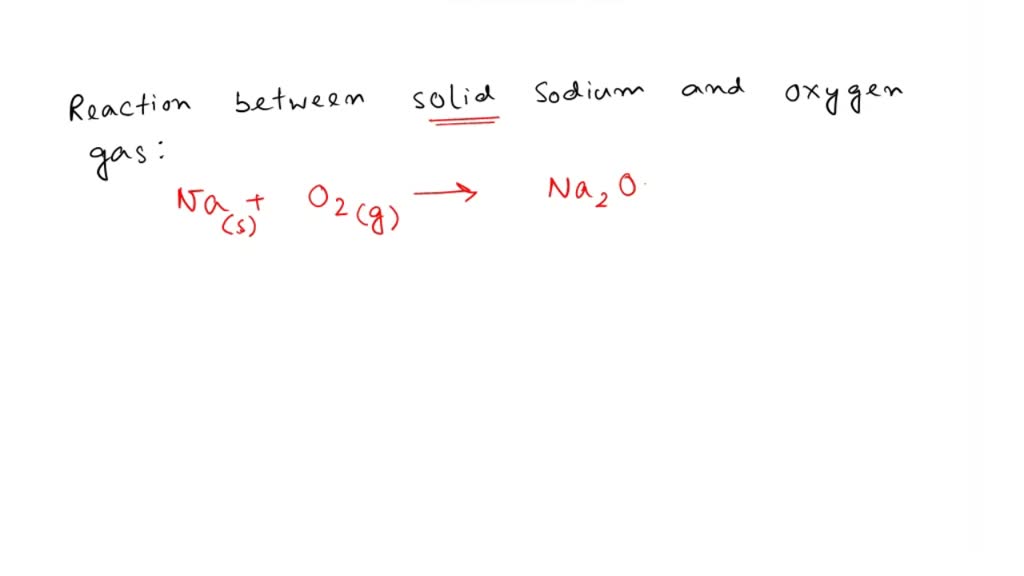
SOLVED Question 9 solid sodium chloride is produced by the reaction of chlorine gas and solid
Standard Heat Of Formation Of Solid Sodium Chloride 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. As mentioned on the previous page, using hess' law makes it possible to calculate many. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. standard enthalpies of formation. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to.
From rayb78.github.io
Heat Of Formation Chart Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. As mentioned on the previous page, using hess' law makes it possible to calculate many. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. standard enthalpies of formation. . Standard Heat Of Formation Of Solid Sodium Chloride.
From www.alamy.com
Sodium Chloride ionic bond formation. NaCl structure. Sodium and Chlorine atom chemical reaction Standard Heat Of Formation Of Solid Sodium Chloride As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. standard enthalpies of formation. learn how. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.shalom-education.com
Evaluating Molecular Models Edexcel GCSE Chemistry Revision Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. As mentioned on the previous page, using hess' law makes it possible to calculate many. standard enthalpies of formation. . Standard Heat Of Formation Of Solid Sodium Chloride.
From www.chemistrystudent.com
Enthalpy of Solution (Alevel) ChemistryStudent Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. standard enthalpies of formation. As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate the standard heat of formation of a compound from its elements, and how to use. Standard Heat Of Formation Of Solid Sodium Chloride.
From schoolworkhelper.net
Standard Enthalpies of Formation SchoolWorkHelper Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. standard enthalpies of formation. As mentioned on the previous page, using hess' law makes it possible to calculate many. . Standard Heat Of Formation Of Solid Sodium Chloride.
From stock.adobe.com
Vecteur Stock Synthesis reaction sodium chloride formation of sodium metal and chlorine gas Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.w3schools.blog
Standard enthalpy of ionization W3schools Standard Heat Of Formation Of Solid Sodium Chloride standard enthalpies of formation. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. As mentioned on the previous page, using hess' law makes it possible to calculate many. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. . Standard Heat Of Formation Of Solid Sodium Chloride.
From www.numerade.com
SOLVED Using Standard Enthalpy of Formation Enthalpy Test (all answers to three sig figs Standard Heat Of Formation Of Solid Sodium Chloride 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it. Standard Heat Of Formation Of Solid Sodium Chloride.
From chem.libretexts.org
4.3 The Reaction of Sodium with Chlorine Chemistry LibreTexts Standard Heat Of Formation Of Solid Sodium Chloride As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. 35 rows. Standard Heat Of Formation Of Solid Sodium Chloride.
From mungfali.com
Sodium Chloride Molecule Diagram Standard Heat Of Formation Of Solid Sodium Chloride 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. learn how to calculate the. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.priyamstudycentre.com
Sodium Chloride (NaCl) Uses, Crystal Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. As mentioned on the previous page, using hess' law makes it possible to calculate many. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. learn how to calculate enthalpy. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.studocu.com
Standard Enthalpy of Formation Table Standard Enthalpy of Formation* for Various Compounds Standard Heat Of Formation Of Solid Sodium Chloride 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. standard enthalpies of formation. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. As mentioned on the previous page, using hess' law makes it possible to calculate many. . Standard Heat Of Formation Of Solid Sodium Chloride.
From askfilo.com
configuration, formation of Sodium chloride from sodium and chlorine... Standard Heat Of Formation Of Solid Sodium Chloride 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. . Standard Heat Of Formation Of Solid Sodium Chloride.
From solvedlib.com
Why is the formation of solid sodium chloride from so… SolvedLib Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. standard enthalpies of. Standard Heat Of Formation Of Solid Sodium Chloride.
From rayb78.github.io
Heat Of Formation Chart Standard Heat Of Formation Of Solid Sodium Chloride 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it. Standard Heat Of Formation Of Solid Sodium Chloride.
From learningschoolandy.z21.web.core.windows.net
Heat Of Formation List Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. As mentioned on the previous page, using hess' law makes it possible to calculate many. standard enthalpies of. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.nagwa.com
Question Video Calculating the Standard Enthalpy of Reaction for the of Ammonium Standard Heat Of Formation Of Solid Sodium Chloride 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate the standard heat. Standard Heat Of Formation Of Solid Sodium Chloride.
From mavink.com
Standard Enthalpy Chart Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. As. Standard Heat Of Formation Of Solid Sodium Chloride.
From mavink.com
Sodium Chloride Bonding Diagram Standard Heat Of Formation Of Solid Sodium Chloride standard enthalpies of formation. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point,. Standard Heat Of Formation Of Solid Sodium Chloride.
From askfilo.com
Identify the correct order of standard enthalpy of formation of sodium ha.. Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate the standard heat. Standard Heat Of Formation Of Solid Sodium Chloride.
From ar.inspiredpencil.com
Atomic Structure Of Sodium Chloride Standard Heat Of Formation Of Solid Sodium Chloride 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. learn. Standard Heat Of Formation Of Solid Sodium Chloride.
From bnospmmdwi.blogspot.com
How To Calculate Standard Enthalpy Change How to calculate standard enthalpy change of Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. As. Standard Heat Of Formation Of Solid Sodium Chloride.
From chem.libretexts.org
5.7 Enthalpies of Formation Chemistry LibreTexts Standard Heat Of Formation Of Solid Sodium Chloride standard enthalpies of formation. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. As mentioned on the previous page, using hess' law makes it possible to calculate many. . Standard Heat Of Formation Of Solid Sodium Chloride.
From quizzlistreplevies.z13.web.core.windows.net
Heat Of Formation Equations Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. standard enthalpies of formation. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. 35 rows this page provides supplementary chemical data on sodium chloride, including. Standard Heat Of Formation Of Solid Sodium Chloride.
From courses.lumenlearning.com
Water BIO103 Human Biology Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. standard enthalpies. Standard Heat Of Formation Of Solid Sodium Chloride.
From fyoqrrxjl.blob.core.windows.net
Standard Enthalpy Of Formation Reference Form at Edith Hilton blog Standard Heat Of Formation Of Solid Sodium Chloride As mentioned on the previous page, using hess' law makes it possible to calculate many. standard enthalpies of formation. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 35 rows. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.shutterstock.com
Vector Illustration Sodium Chloride Formation By Stock Vector (Royalty Free) 2269898333 Standard Heat Of Formation Of Solid Sodium Chloride 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. standard enthalpies of formation. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.youtube.com
Structure of Sodium Chloride (NaCl) Formation of NaCl How to draw NaCl Structure Ionic Standard Heat Of Formation Of Solid Sodium Chloride standard enthalpies of formation. As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.thesciencehive.co.uk
Enthalpy Changes — the science hive Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. As mentioned on the previous page, using hess' law makes it possible to calculate many. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. standard enthalpies of formation. . Standard Heat Of Formation Of Solid Sodium Chloride.
From stahonorschemistry.weebly.com
III Calculating Enthalpies STA Form IV Honors Chemistry Thermochemistry Unit Standard Heat Of Formation Of Solid Sodium Chloride standard enthalpies of formation. 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. . Standard Heat Of Formation Of Solid Sodium Chloride.
From www.slideserve.com
PPT Giant Ionic Structures PowerPoint Presentation, free download ID663487 Standard Heat Of Formation Of Solid Sodium Chloride As mentioned on the previous page, using hess' law makes it possible to calculate many. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. standard enthalpies of formation. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.numerade.com
SOLVED Question 9 solid sodium chloride is produced by the reaction of chlorine gas and solid Standard Heat Of Formation Of Solid Sodium Chloride 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. standard enthalpies of formation. As mentioned on the previous page, using hess' law makes it possible to calculate many. . Standard Heat Of Formation Of Solid Sodium Chloride.
From hxesyzijm.blob.core.windows.net
Standard Enthalpy Of Formation Database at Kevin Leblanc blog Standard Heat Of Formation Of Solid Sodium Chloride learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. As mentioned on the previous page, using hess' law makes it possible to calculate many. standard enthalpies of formation. . Standard Heat Of Formation Of Solid Sodium Chloride.
From kaden-chapter.blogspot.com
Standard Enthalpy Of Formation Table Pdf 30+ Pages Summary [1.8mb] Latest Update Kaden Books Standard Heat Of Formation Of Solid Sodium Chloride 136 rows find heat of formation data for various compounds and elements in kcal/mol or kj/mol. 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. learn how to calculate the standard heat of formation of a compound from its elements, and how to use it to. standard. Standard Heat Of Formation Of Solid Sodium Chloride.
From www.youtube.com
Balancing and Writing the Equation for Sodium + Chlorine gas YouTube Standard Heat Of Formation Of Solid Sodium Chloride 35 rows this page provides supplementary chemical data on sodium chloride, including its boiling point, density, and phase. As mentioned on the previous page, using hess' law makes it possible to calculate many. learn how to calculate enthalpy changes for chemical reactions using standard enthalpies of formation, which are measured under specific. 136 rows find heat of. Standard Heat Of Formation Of Solid Sodium Chloride.