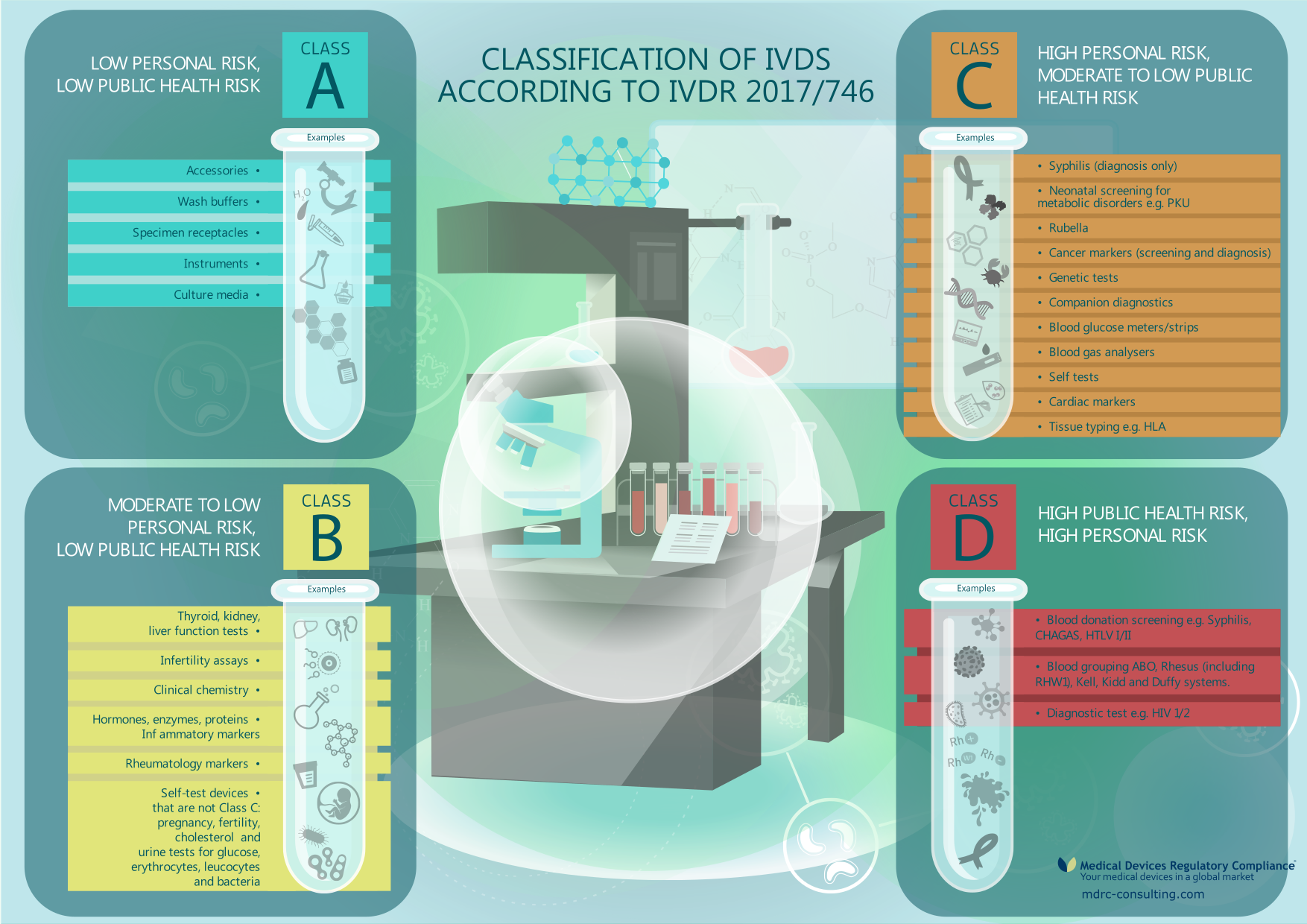
Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) . This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find out how it affects in vitro. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european.
from mdrc-consulting.com
This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Find out how it affects in vitro.
Europe's IVD regulatory approval process MDRC
Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find out how it affects in vitro.
From www.thermofisher.com
IVDD vs. IVDR Classifications Defined and Compared OEMpowered Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.apcerls.com
In Vitro Medical Devices Regulation (IVDR) Regulatory Framework Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find out how it affects in vitro. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From connections.arabhealthonline.com
Regulation (EU) 2017/746 on In Vitro Diagnostic (IVDR) Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.slideserve.com
PPT The key differences between the MDR and IVDR in the EU PowerPoint Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.youtube.com
Navigating the In Vitro Diagnostic Regulation (IVDR) Rollout for Legacy Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Find out how it affects in vitro. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From leonresearch.com
NEW REGULATION FOR MEDICAL DEVICES IN VITRO IVDR_RG (UE) 746/2017 Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From key2compliance.com
IVDR brings stricter regulations for In Vitro Diagnostic medical Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find out how it affects in vitro. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.bsigroup.com
In Vitro Diagnostic Regulation IVDR Medical Devices BSI America Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.greenlight.guru
Transitioning to IVDR The New EU Regulation for In Vitro Diagnostic Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Find out how it affects in vitro. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.greenlight.guru
IVDR for In Vitro Diagnostic Medical Device Companies Ultimate Guide Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.universalmedica.com
The European Union In Vitro Diagnostic Medical Devices Regulation (IVDR Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.arenasolutions.com
Preparing for Europe’s New In Vitro Diagnostic Medical Device Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Find out how it affects in vitro. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.networkpartnersgroup.com
THE IVDR TOP 8 A Roadmap For Transitioning To The European in vitro Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From ehaweb.org
Implementation of the new EU Regulation for In Vitro Diagnostic Medical Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From pharmaceutical.report
Introduction to the InVitro Diagnostic Regulation (IVDR) Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.arenasolutions.com
How to Classify Your Medical Device Under the EU MDR and IVDR Arena Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Find out how it affects in vitro. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From mdrc-consulting.com
Europe's IVD regulatory approval process MDRC Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find out how it affects in vitro. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.universalmedica.com
The European Union In Vitro Diagnostic Medical Devices Regulation (IVDR Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.tuv.com
In Vitro Diagnostic Medical Device Regulation (IVDR) IN TÜV Rheinland Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From qbdgroup.com
IVDR classification of invitro diagnostic medical devices a brief guide Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.devicia.com
New EU regulation for in vitro diagnostic medical devices (IVDR 2017/ Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From management-forum.co.uk
Introduction to the InVitro Diagnostic Regulation (IVDR) Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From diagnostics.roche.com
In Vitro Diagnostics Medical Devices Regulation (IVDR) setting the Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From dicentra.com
New European Union In Vitro Diagnostics Regulation (EU IVDR) dicentra Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.presentationeze.com
IVDR 2017 746 In Vitro Diagnostics Device Regulation Implementation Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find out how it affects in vitro. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.tuvsud.com
IVDR In Vitro Diagnostic Medical Device Regulation TÜV SÜD Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Find out how it affects in vitro. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.tuvsud.com
IVDR Infographic TÜV SÜD Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From eurointervention.pcronline.com
Medical device regulation in Europe what is changing and how can I Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.thema-med.com
In Vitro Diagnostic Medical Devices proposed extension of IVDR 2017/746 Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Find out how it affects in vitro. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.jamasoftware.com
Guide to New EU In Vitro Diagnostic Regulations Jama Software Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.thermofisher.com
In Vitro Diagnostic Medical Devices Regulation IVDR EU Behind the Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Find out how it affects in vitro. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.nsmedicaldevices.com
IVDR How will new regulations impact in vitro diagnostics manufacturers? Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Find. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From clustercollaboration.eu
Tips and tools for dealing with the invitro diagnostics regulation Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the rules and requirements for placing, making available and using ivd medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.arenasolutions.com
Preparing for Europe’s New In Vitro Diagnostic Medical Device Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).
From www.youtube.com
Understanding the IN VITRO DIAGNOSTIC REGULATION (IVDR) Everything You Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr) Find out how it affects in vitro. This regulation replaces directive 98/79/ec and sets high standards of quality and safety for in vitro diagnostic medical devices in the eu. Learn about the eu regulations on medical devices (mdr) and in vitro diagnostic medical devices (ivdr) from the european. Learn about the rules and requirements for placing, making available and using. Eu Regulation On In Vitro Diagnostic Medical Devices (Ivdr).