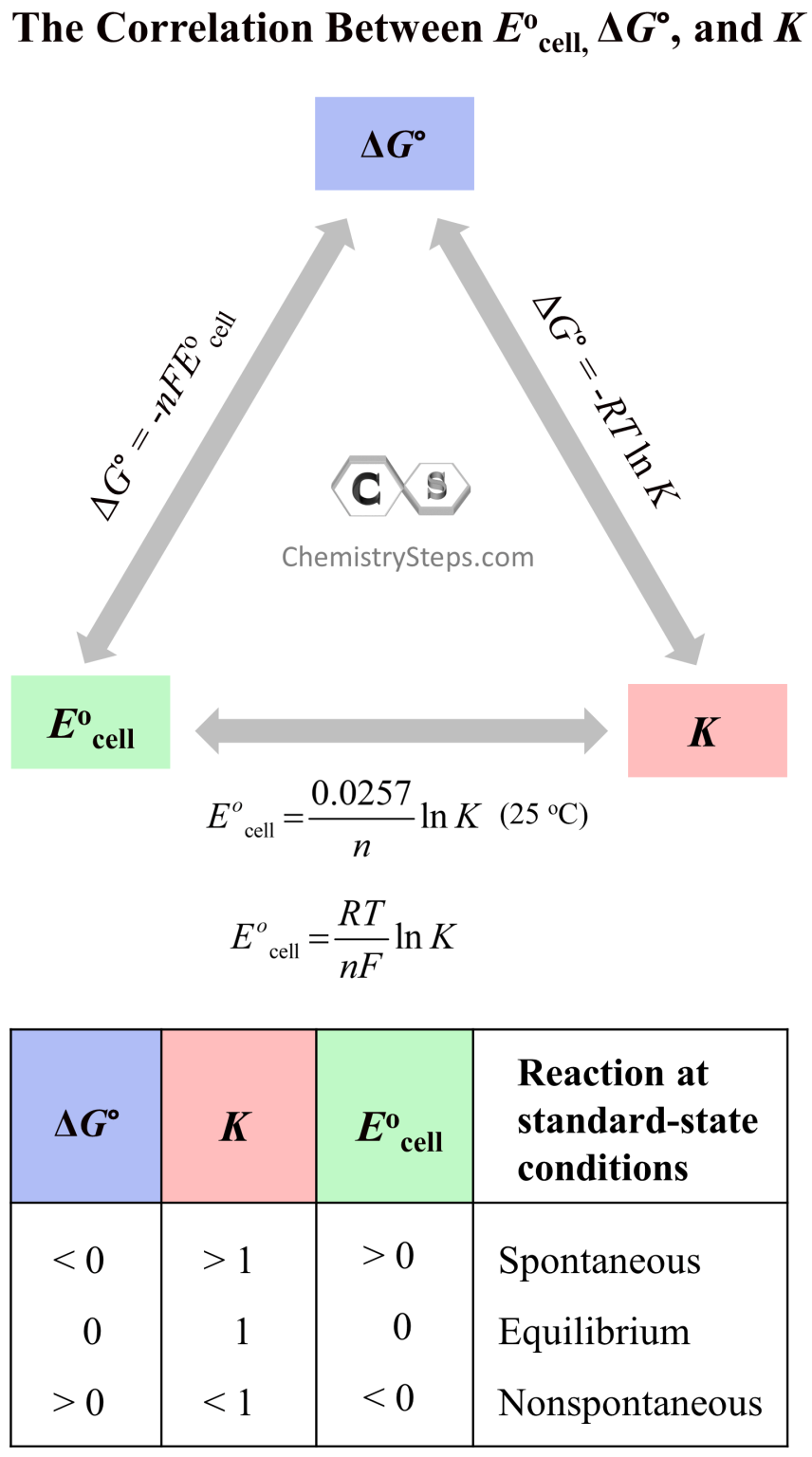
What Is Free Energy In Electrochemistry . Understand how gibbs energy pertains to electrochemical properties. Plus, examine how to calculate the electrochemical potential. Discover the cell potential and free energy calculations. Gibbs free energy, denoted \ (g\), combines enthalpy and. This connection shows there is a. Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). This is because the difference in. If e°cell > 0, then the process is. It combines enthalpy and entropy changes,. In particular, the change in free energy was defined in terms of the maximum work (w max),. In this chapter, we have learned how the cell potential will tell us whether a reaction is spontaneous. In an earlier chapter, the free energy was defined as the energy that was available to do work. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. Δ g ° cell = − nfe ° cell. The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction.
from general.chemistrysteps.com
In a galvanic cell, the gibbs free energy is related to the potential by: Understand how gibbs energy pertains to electrochemical properties. In this chapter, we have learned how the cell potential will tell us whether a reaction is spontaneous. In particular, the change in free energy was defined in terms of the maximum work (w max),. It combines enthalpy and entropy changes,. This is because the difference in. Gibbs free energy, denoted \ (g\), combines enthalpy and. Δ g ° cell = − nfe ° cell. This connection shows there is a. Plus, examine how to calculate the electrochemical potential.
Cell Potential, Free Energy, and Equilibrium Constant Chemistry Steps
What Is Free Energy In Electrochemistry In an earlier chapter, the free energy was defined as the energy that was available to do work. In an earlier chapter, the free energy was defined as the energy that was available to do work. This connection shows there is a. In particular, the change in free energy was defined in terms of the maximum work (w max),. Plus, examine how to calculate the electrochemical potential. If e°cell > 0, then the process is. In this chapter, we have learned how the cell potential will tell us whether a reaction is spontaneous. It combines enthalpy and entropy changes,. Discover the cell potential and free energy calculations. Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). In a galvanic cell, the gibbs free energy is related to the potential by: The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. Gibbs free energy, denoted \ (g\), combines enthalpy and. This is because the difference in. Δ g ° cell = − nfe ° cell. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity.
From www.meritnation.com
What is the relationship between Gibbs free energy of the cell reaction What Is Free Energy In Electrochemistry Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. In an earlier chapter, the free energy was defined as the energy that was available to do work. The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. In a galvanic cell, the gibbs free. What Is Free Energy In Electrochemistry.
From www.expii.com
Gibbs Free Energy — Definition & Overview Expii What Is Free Energy In Electrochemistry The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. In an earlier chapter, the free energy was defined as the energy that was available to do work. Work is expressed as a negative number because work is being done by a system (an electrochemical cell with. What Is Free Energy In Electrochemistry.
From www.slideserve.com
PPT Lecture 2. Basic Electrochemistry PowerPoint Presentation, free What Is Free Energy In Electrochemistry Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. Δ g ° cell = − nfe ° cell. Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). In a galvanic cell, the gibbs free energy is related to the potential by: Understand how gibbs. What Is Free Energy In Electrochemistry.
From www.youtube.com
Problem 3 on Gibbs Free Energy &equilibrium constant(Electrochemistry What Is Free Energy In Electrochemistry The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). Understand how gibbs energy pertains to electrochemical properties. Δ g ° cell = − nfe. What Is Free Energy In Electrochemistry.
From www.youtube.com
Problem 5 on nernst equation&Gibbs Free Energy(Electrochemistry part 40 What Is Free Energy In Electrochemistry Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. If e°cell > 0, then the process is. It combines enthalpy and entropy changes,. Δ g ° cell = − nfe ° cell. In a galvanic cell, the gibbs free energy is related to the potential by: This is because the difference in. Gibbs free energy, denoted \. What Is Free Energy In Electrochemistry.
From www.youtube.com
Problem1 on Gibbs Free Energy &nernst equation (Electrochemistry part What Is Free Energy In Electrochemistry In this chapter, we have learned how the cell potential will tell us whether a reaction is spontaneous. It combines enthalpy and entropy changes,. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. In a galvanic cell, the gibbs free energy is related to the potential by: Work is expressed as a negative number because work is. What Is Free Energy In Electrochemistry.
From www.youtube.com
3.8Relationship between Gibbs free energy and equilibrium constant What Is Free Energy In Electrochemistry Understand how gibbs energy pertains to electrochemical properties. If e°cell > 0, then the process is. Discover the cell potential and free energy calculations. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. It combines enthalpy and entropy changes,. In this chapter, we have learned how the cell potential will tell us whether a reaction is spontaneous.. What Is Free Energy In Electrochemistry.
From www.youtube.com
Electrochemistry Free Energy YouTube What Is Free Energy In Electrochemistry It combines enthalpy and entropy changes,. This is because the difference in. In an earlier chapter, the free energy was defined as the energy that was available to do work. Gibbs free energy, denoted \ (g\), combines enthalpy and. Understand how gibbs energy pertains to electrochemical properties. In this chapter, we have learned how the cell potential will tell us. What Is Free Energy In Electrochemistry.
From www.researchgate.net
A) The Gibbs free energy change for electrochemical synthesis of What Is Free Energy In Electrochemistry Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. If e°cell > 0, then the process is. Gibbs free energy, denoted \ (g\), combines enthalpy and. The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. This is because the difference in. In a. What Is Free Energy In Electrochemistry.
From www.slideserve.com
PPT ELECTROCHEMISTRY INVOLVES TWO MAIN TYPES OF PROCESSES PowerPoint What Is Free Energy In Electrochemistry In a galvanic cell, the gibbs free energy is related to the potential by: The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. In particular, the change in free energy was defined in terms of the maximum work (w max),. Understand how gibbs energy pertains to. What Is Free Energy In Electrochemistry.
From www.youtube.com
19.1 Cell potential and Gibbs free energy (HL) YouTube What Is Free Energy In Electrochemistry The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. In particular, the change in free energy was defined in terms of the maximum work (w max),. It combines enthalpy and entropy changes,. In an earlier chapter, the free energy was defined as the energy that was. What Is Free Energy In Electrochemistry.
From mrschimomot.blogspot.com
Gibbs Free Energy Formula In Electrochemistry Mrschimomot What Is Free Energy In Electrochemistry This is because the difference in. If e°cell > 0, then the process is. In a galvanic cell, the gibbs free energy is related to the potential by: In particular, the change in free energy was defined in terms of the maximum work (w max),. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. In an earlier. What Is Free Energy In Electrochemistry.
From www.youtube.com
Electrochemistry 05/ Gibbs Free Energy+Electrochemical Series //JEE What Is Free Energy In Electrochemistry It combines enthalpy and entropy changes,. In a galvanic cell, the gibbs free energy is related to the potential by: The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. Work is expressed as a negative number because work is being done by a system (an electrochemical. What Is Free Energy In Electrochemistry.
From mrschimomot.blogspot.com
Gibbs Free Energy Formula In Electrochemistry Mrschimomot What Is Free Energy In Electrochemistry Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). In particular, the change in free energy was defined in terms of the maximum work (w max),. In this chapter, we have learned how the cell potential will tell us whether a reaction is spontaneous. It combines enthalpy. What Is Free Energy In Electrochemistry.
From www.youtube.com
Free energy and cell potential Redox reactions and electrochemistry What Is Free Energy In Electrochemistry Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. Understand how gibbs energy pertains to electrochemical properties. This connection shows there is a. Δ g ° cell = − nfe ° cell. Plus, examine how to calculate the electrochemical potential. In a galvanic cell, the gibbs free energy is related to the potential by: If e°cell >. What Is Free Energy In Electrochemistry.
From www.slideserve.com
PPT electrochemistry PowerPoint Presentation, free download ID2683693 What Is Free Energy In Electrochemistry Δ g ° cell = − nfe ° cell. In particular, the change in free energy was defined in terms of the maximum work (w max),. Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). In an earlier chapter, the free energy was defined as the energy. What Is Free Energy In Electrochemistry.
From www.youtube.com
Electrochemistry।Class 12 (Lecture 2)। Electrochemical series।Nernst What Is Free Energy In Electrochemistry This connection shows there is a. Understand how gibbs energy pertains to electrochemical properties. Plus, examine how to calculate the electrochemical potential. In particular, the change in free energy was defined in terms of the maximum work (w max),. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. In this chapter, we have learned how the cell. What Is Free Energy In Electrochemistry.
From www.youtube.com
Electrochemistry 5 Nernst Equation Equilibrium Constant Gibb’s What Is Free Energy In Electrochemistry This connection shows there is a. In a galvanic cell, the gibbs free energy is related to the potential by: If e°cell > 0, then the process is. In particular, the change in free energy was defined in terms of the maximum work (w max),. Gibbs free energy, denoted \ (g\), combines enthalpy and. It combines enthalpy and entropy changes,.. What Is Free Energy In Electrochemistry.
From www.youtube.com
ELECTROCHEMISTRY 08 GIBBS FREE ENERGY & NERNST EQUATION YouTube What Is Free Energy In Electrochemistry This connection shows there is a. In an earlier chapter, the free energy was defined as the energy that was available to do work. Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). The potential or voltage or emf or whatever you prefer to call it is. What Is Free Energy In Electrochemistry.
From mrschimomot.blogspot.com
Gibbs Free Energy Formula In Electrochemistry Mrschimomot What Is Free Energy In Electrochemistry In particular, the change in free energy was defined in terms of the maximum work (w max),. The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. This is because the difference in. Plus, examine. What Is Free Energy In Electrochemistry.
From www.researchgate.net
Free energy diagrams as a function of the electrochemical potential What Is Free Energy In Electrochemistry The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. In a galvanic cell, the gibbs free energy is related to the potential by: Gibbs free energy, denoted \ (g\), combines enthalpy and. Discover the cell potential and free energy calculations. Work is expressed as a negative. What Is Free Energy In Electrochemistry.
From study.com
Electrochemistry Free Energy and Cell Potential Energy Video What Is Free Energy In Electrochemistry If e°cell > 0, then the process is. Understand how gibbs energy pertains to electrochemical properties. Plus, examine how to calculate the electrochemical potential. Gibbs free energy, denoted \ (g\), combines enthalpy and. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. This is because the difference in. In a galvanic cell, the gibbs free energy is. What Is Free Energy In Electrochemistry.
From www.youtube.com
Gibbs free energy (Electrochemistry part 35 for CBSE class 12 JEE IIT What Is Free Energy In Electrochemistry In this chapter, we have learned how the cell potential will tell us whether a reaction is spontaneous. This is because the difference in. Understand how gibbs energy pertains to electrochemical properties. The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. Gibbs free energy is a. What Is Free Energy In Electrochemistry.
From www.slideserve.com
PPT Electrochemistry PowerPoint Presentation, free download ID2281210 What Is Free Energy In Electrochemistry Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. In a galvanic cell, the gibbs free energy is related to the potential by: It combines enthalpy and entropy changes,. This is because the difference in.. What Is Free Energy In Electrochemistry.
From mrschimomot.blogspot.com
Gibbs Free Energy Formula In Electrochemistry Mrschimomot What Is Free Energy In Electrochemistry This is because the difference in. In a galvanic cell, the gibbs free energy is related to the potential by: Plus, examine how to calculate the electrochemical potential. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. In this chapter, we have learned how the cell potential will tell us whether a reaction is spontaneous. Discover the. What Is Free Energy In Electrochemistry.
From www.youtube.com
Worked examples of Gibbs Free Energy and Electrochemistry YouTube What Is Free Energy In Electrochemistry This is because the difference in. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. Gibbs free energy, denoted \ (g\), combines enthalpy and. Plus, examine how to calculate the electrochemical potential. This connection shows there is a. In a galvanic cell, the gibbs free energy is related to the potential by: In particular, the change in. What Is Free Energy In Electrochemistry.
From mavink.com
Gibbs Free Energy Phase Diagram What Is Free Energy In Electrochemistry Discover the cell potential and free energy calculations. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. This is because the difference in. If e°cell > 0, then the process is. The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. In a galvanic. What Is Free Energy In Electrochemistry.
From general.chemistrysteps.com
Cell Potential, Free Energy, and Equilibrium Constant Chemistry Steps What Is Free Energy In Electrochemistry In a galvanic cell, the gibbs free energy is related to the potential by: Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). Δ g ° cell = − nfe ° cell. If e°cell > 0, then the process is. In particular, the change in free energy. What Is Free Energy In Electrochemistry.
From www.youtube.com
relation between emf and free energy electrochemistry 2 class 12 What Is Free Energy In Electrochemistry Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. If e°cell > 0, then the process is. Plus, examine how to calculate the electrochemical potential. Understand how gibbs energy pertains to electrochemical properties. In particular, the change in free energy was defined in terms of the maximum work (w max),. In a galvanic cell, the gibbs free. What Is Free Energy In Electrochemistry.
From mrschimomot.blogspot.com
Gibbs Free Energy Formula In Electrochemistry Mrschimomot What Is Free Energy In Electrochemistry In this chapter, we have learned how the cell potential will tell us whether a reaction is spontaneous. In a galvanic cell, the gibbs free energy is related to the potential by: It combines enthalpy and entropy changes,. Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. This connection shows there is a. Work is expressed as. What Is Free Energy In Electrochemistry.
From www.youtube.com
3.7Gibbs free energy of electrochemical cell, class 12th What Is Free Energy In Electrochemistry Δ g ° cell = − nfe ° cell. This is because the difference in. If e°cell > 0, then the process is. In an earlier chapter, the free energy was defined as the energy that was available to do work. Discover the cell potential and free energy calculations. It combines enthalpy and entropy changes,. Understand how gibbs energy pertains. What Is Free Energy In Electrochemistry.
From www.youtube.com
Gibbs Free Energy and Electrochemistry (Nernst Equation) YouTube What Is Free Energy In Electrochemistry Gibbs free energy is a crucial concept in electrochemistry, predicting reaction spontaneity. If e°cell > 0, then the process is. Plus, examine how to calculate the electrochemical potential. Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). It combines enthalpy and entropy changes,. In a galvanic cell,. What Is Free Energy In Electrochemistry.
From www.slideserve.com
PPT Electrochemistry MAE 212 PowerPoint Presentation ID1588026 What Is Free Energy In Electrochemistry Δ g ° cell = − nfe ° cell. Understand how gibbs energy pertains to electrochemical properties. It combines enthalpy and entropy changes,. In particular, the change in free energy was defined in terms of the maximum work (w max),. In an earlier chapter, the free energy was defined as the energy that was available to do work. If e°cell. What Is Free Energy In Electrochemistry.
From www.youtube.com
Cell Potential & Gibbs Free Energy, Standard Reduction Potentials What Is Free Energy In Electrochemistry In particular, the change in free energy was defined in terms of the maximum work (w max),. If e°cell > 0, then the process is. In an earlier chapter, the free energy was defined as the energy that was available to do work. Understand how gibbs energy pertains to electrochemical properties. This connection shows there is a. In a galvanic. What Is Free Energy In Electrochemistry.
From www.youtube.com
Electrochemistry and Free Energy, Nernst Equation, Equilibrium Constant What Is Free Energy In Electrochemistry Understand how gibbs energy pertains to electrochemical properties. Work is expressed as a negative number because work is being done by a system (an electrochemical cell with a positive potential). The potential or voltage or emf or whatever you prefer to call it is a measure of the free energy of the reaction. Plus, examine how to calculate the electrochemical. What Is Free Energy In Electrochemistry.