Standard Heat Of Formation N2 . the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. standard enthalpies of formation. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole of a substance from.
from www.numerade.com
standard enthalpies of formation. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole of a substance from. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for.
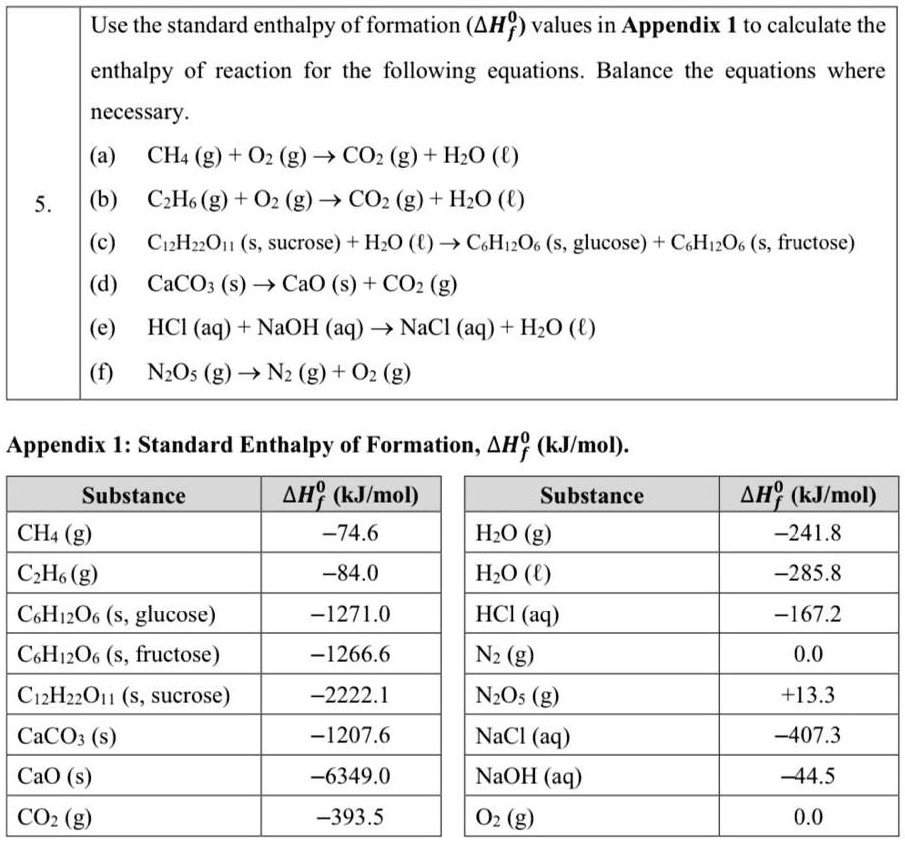
SOLVED Use the standard enthalpy of formation (ΔHf) values in Appendix I to calculate the
Standard Heat Of Formation N2 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. standard enthalpies of formation. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole of a substance from. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c.
From www.slideserve.com
PPT Energetics PowerPoint Presentation ID1204656 Standard Heat Of Formation N2 The magnitude of δ h for a reaction depends on the physical states of the reactants and the. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole of a substance from. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of. Standard Heat Of Formation N2.
From www.slideserve.com
PPT Chapter 16 Thermodynamics Entropy, Free Energy, and Equilibrium PowerPoint Presentation Standard Heat Of Formation N2 the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole. Standard Heat Of Formation N2.
From lessonluft.z19.web.core.windows.net
Heat Of Formation Chart Standard Heat Of Formation N2 the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. standard enthalpies of formation. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. . Standard Heat Of Formation N2.
From www.slideshare.net
Heat of formation by reactions Standard Heat Of Formation N2 standard enthalpies of formation. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. The magnitude of δ h for a reaction depends on the physical states of the reactants and the.. Standard Heat Of Formation N2.
From www.numerade.com
SOLVED Use the standard enthalpy of formation (ΔHf) values in Appendix I to calculate the Standard Heat Of Formation N2 definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1. Standard Heat Of Formation N2.
From ar.inspiredpencil.com
Heat Of Formation Table Standard Heat Of Formation N2 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o. Standard Heat Of Formation N2.
From www.toppr.com
N2(g) + 3H2(g) → 2NH3(g); A,Hº = 92.4 kJ mol What is the standard enthalpy of formation of NH3 Standard Heat Of Formation N2 the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. It means that 33.2 kj of. Standard Heat Of Formation N2.
From nitrogengassanroso.blogspot.com
Nitrogen Gas Nitrogen Gas Enthalpy Of Formation Standard Heat Of Formation N2 definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole of a substance from. the standard enthalpy of formation is a measure of the energy released or consumed when one mole. Standard Heat Of Formation N2.
From slideplayer.com
Chapter 17 Thermochemistry Heat and Chemical Change ppt video online download Standard Heat Of Formation N2 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole. Standard Heat Of Formation N2.
From www.numerade.com
SOLVED The standard enthalpy change for the following reaction is 164 kJ at 298 K. 2 N2(g) + O2 Standard Heat Of Formation N2 standard enthalpies of formation. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard. Standard Heat Of Formation N2.
From www.numerade.com
SOLVED All of the following have a standard heat of formation value of zero at 25°C and 1.0 Standard Heat Of Formation N2 The magnitude of δ h for a reaction depends on the physical states of the reactants and the. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. standard enthalpies of formation. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of. Standard Heat Of Formation N2.
From www.toppr.com
The standard heats of formation in kcal mol^1 of NO2(g) and N2O4(g) are 8.0 and 2.0 Standard Heat Of Formation N2 the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c.. Standard Heat Of Formation N2.
From byjus.com
If the heat of formation of NO2 is x 1/2N2+O2=NO .heat of reaction N2+O2=2NO is y and the heat Standard Heat Of Formation N2 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole of a substance from. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. It means that 33.2 kj of. Standard Heat Of Formation N2.
From www.meritnation.com
IF THE HEAT OF FORMATION OF NO2 IS X THE HEAT OF REACTION OF N2 +O2 =2NO IS Y AND Chemistry Standard Heat Of Formation N2 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. The magnitude of δ h for a reaction depends on. Standard Heat Of Formation N2.
From www.slideserve.com
PPT CHAPTER 4 PowerPoint Presentation ID3180471 Standard Heat Of Formation N2 definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1. Standard Heat Of Formation N2.
From www.studocu.com
Standard Enthalpy of Formation Table Standard Enthalpy of Formation* for Various Compounds Standard Heat Of Formation N2 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and. Standard Heat Of Formation N2.
From www.researchgate.net
Standard Gibbs's free energy of the nitride formation reaction... Download Scientific Diagram Standard Heat Of Formation N2 the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole of a substance from. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of. Standard Heat Of Formation N2.
From www.slideserve.com
PPT CHAPTER 4 PowerPoint Presentation ID3180471 Standard Heat Of Formation N2 definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created.. Standard Heat Of Formation N2.
From slideplayer.com
Chemistry ppt download Standard Heat Of Formation N2 standard enthalpies of formation. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying. Standard Heat Of Formation N2.
From exoluenrv.blob.core.windows.net
Standard Enthalpy Of Formation Diamond at James Torres blog Standard Heat Of Formation N2 the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of. Standard Heat Of Formation N2.
From www.toppr.com
If the heat of formation of NO2 is 'x' [1/2 N2(g) + O2(g)→ NO2(g)] the heat of reaction N2(g Standard Heat Of Formation N2 standard enthalpies of formation. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. The magnitude of δ h for a reaction depends on the physical states. Standard Heat Of Formation N2.
From www.toppr.com
Standard enthalpy of combustion Calculate the enthalpy change the reaction N2(g) + 3H2(g) → Standard Heat Of Formation N2 the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole of a substance from. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of. Standard Heat Of Formation N2.
From exoyndeil.blob.core.windows.net
Standard Enthalpy Of Formation In Elements at Michael Zapien blog Standard Heat Of Formation N2 standard enthalpies of formation. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of.. Standard Heat Of Formation N2.
From exokycsnc.blob.core.windows.net
Standard Enthalpy Of Formation Table Elements at Filomena Gilbert blog Standard Heat Of Formation N2 standard enthalpies of formation. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of.. Standard Heat Of Formation N2.
From oneclass.com
OneClass Calculate the standard enthalpy of formation of N2O5 from the following data AH114.1 Standard Heat Of Formation N2 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. the standard enthalpy of formation is a measure of the. Standard Heat Of Formation N2.
From www.youtube.com
CHEMISTRY 101 Standard Enthalpy of reaction from Standard Enthalpies of Formation YouTube Standard Heat Of Formation N2 the standard enthalpy of formation, δh ∘ f, is the enthalpy change accompanying the formation of 1 mole of a substance from. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole. Standard Heat Of Formation N2.
From dxorvtvqp.blob.core.windows.net
Standard Enthalpy Of Formation Gibbsite at Samuel Speed blog Standard Heat Of Formation N2 definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created. standard enthalpies of formation. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or. Standard Heat Of Formation N2.
From mungfali.com
Enthalpies Of Formation Chart Standard Heat Of Formation N2 standard enthalpies of formation. definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. the standard enthalpy of the formation of nitrogen dioxide is +33.2 kj/mol. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2). Standard Heat Of Formation N2.
From byjus.com
Standard heat of formation of ammonia is x kJ/mol.The heat of reaction when 28 gm of N2(g Standard Heat Of Formation N2 It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created.. Standard Heat Of Formation N2.
From nitrogengassanroso.blogspot.com
Nitrogen Gas Nitrogen Gas Heat Of Formation Standard Heat Of Formation N2 definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. the standard enthalpy. Standard Heat Of Formation N2.
From exoyndeil.blob.core.windows.net
Standard Enthalpy Of Formation In Elements at Michael Zapien blog Standard Heat Of Formation N2 The magnitude of δ h for a reaction depends on the physical states of the reactants and the. standard enthalpies of formation. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c.. Standard Heat Of Formation N2.
From learningschoolgraciauwb.z4.web.core.windows.net
How To Determine The Heat Of Formation Standard Heat Of Formation N2 The magnitude of δ h for a reaction depends on the physical states of the reactants and the. It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. standard enthalpies of formation.. Standard Heat Of Formation N2.
From www.tessshebaylo.com
Balance The Following Chemical Equation And Calculate Standard Enthalpies Of Formation Standard Heat Of Formation N2 It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. the standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created.. Standard Heat Of Formation N2.
From byjus.com
22. The heat of formation (N2O5,g) in KJ/mol on the bases of the following data is 2NO(g)+O2(g Standard Heat Of Formation N2 It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. The magnitude of δ h for a reaction depends on the physical states of the reactants and the. the standard enthalpy of. Standard Heat Of Formation N2.
From schoolworkhelper.net
Standard Enthalpies of Formation Online Homework Help SchoolWorkHelper Standard Heat Of Formation N2 It means that 33.2 kj of energy is required to form one mole of no 2 from ½ mole of nitrogen (n 2) and one mole of oxygen (o 2) at 1 atmospheric pressure and 25 ˚c. standard enthalpies of formation. the standard enthalpy of formation is a measure of the energy released or consumed when one mole. Standard Heat Of Formation N2.