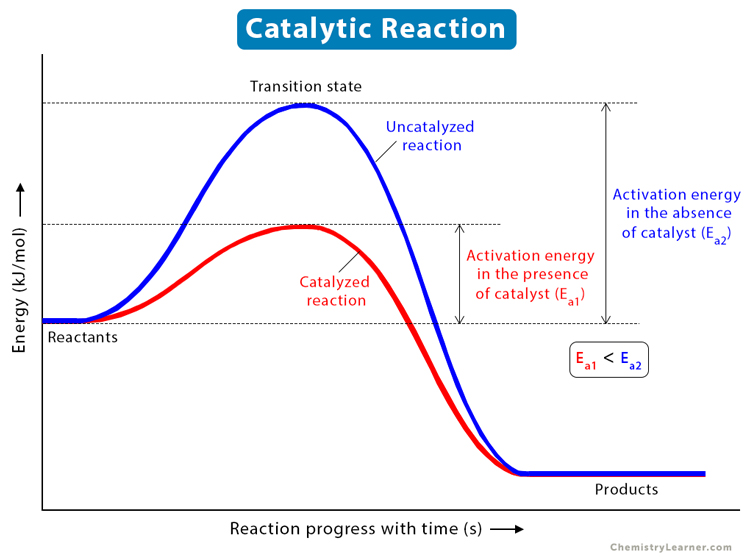
Catalyst Affect Activation Energy Of A Reaction . A catalyst lowers the activation energy of a chemical reaction. It does not lower the. When a catalyst is used at a particular concentration, the rate. effect of enzymes and catalysts. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. A catalyst provides an alternative route for the reaction. a catalyst provides an alternative route for the reaction with a lower activation energy. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. the activation energy of a reaction is 19.0 kj/mol. adding a catalyst has exactly this effect of shifting the activation energy. estimate the activation energy for each process, and identify which one involves a catalyst.
from www.chemistrylearner.com
It does not lower the. estimate the activation energy for each process, and identify which one involves a catalyst. A catalyst lowers the activation energy of a chemical reaction. adding a catalyst has exactly this effect of shifting the activation energy. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. the activation energy of a reaction is 19.0 kj/mol. effect of enzymes and catalysts. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. When a catalyst is used at a particular concentration, the rate. A catalyst provides an alternative route for the reaction.
Activation Energy Definition, Formula, and Graph
Catalyst Affect Activation Energy Of A Reaction adding a catalyst has exactly this effect of shifting the activation energy. effect of enzymes and catalysts. A catalyst lowers the activation energy of a chemical reaction. adding a catalyst has exactly this effect of shifting the activation energy. the activation energy of a reaction is 19.0 kj/mol. a catalyst provides an alternative route for the reaction with a lower activation energy. A catalyst provides an alternative route for the reaction. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. When a catalyst is used at a particular concentration, the rate. estimate the activation energy for each process, and identify which one involves a catalyst. It does not lower the. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the.
From www.youtube.com
6.2.6 / 6.2.7 Describe the effect of a catalyst on a chemical reaction / MaxwellBolztmann Catalyst Affect Activation Energy Of A Reaction A catalyst provides an alternative route for the reaction. A catalyst lowers the activation energy of a chemical reaction. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. estimate the activation energy for each process, and identify which one involves a catalyst. a catalyst provides an alternative. Catalyst Affect Activation Energy Of A Reaction.
From www.slideserve.com
PPT Reaction Rate and Equilibrium PowerPoint Presentation, free download ID5660637 Catalyst Affect Activation Energy Of A Reaction the activation energy of a reaction is 19.0 kj/mol. adding a catalyst has exactly this effect of shifting the activation energy. It does not lower the. a catalyst provides an alternative route for the reaction with a lower activation energy. A catalyst provides an alternative route for the reaction. When a catalyst is used at a particular. Catalyst Affect Activation Energy Of A Reaction.
From www.chemistrylearner.com
Activation Energy Definition, Formula, and Graph Catalyst Affect Activation Energy Of A Reaction in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. a catalyst provides an alternative route for the reaction with a lower activation energy. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. A catalyst provides. Catalyst Affect Activation Energy Of A Reaction.
From www.chemistrystudent.com
Boltzmann Distribution Curves (ALevel) ChemistryStudent Catalyst Affect Activation Energy Of A Reaction When a catalyst is used at a particular concentration, the rate. effect of enzymes and catalysts. a catalyst provides an alternative route for the reaction with a lower activation energy. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. estimate the activation energy for. Catalyst Affect Activation Energy Of A Reaction.
From slideplayer.com
Involve chemical reactions ppt download Catalyst Affect Activation Energy Of A Reaction estimate the activation energy for each process, and identify which one involves a catalyst. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. When a catalyst is used at a particular concentration, the rate. It does not lower the. A catalyst lowers the activation energy of. Catalyst Affect Activation Energy Of A Reaction.
From courses.lumenlearning.com
Catalysis Chemistry Atoms First Catalyst Affect Activation Energy Of A Reaction When a catalyst is used at a particular concentration, the rate. adding a catalyst has exactly this effect of shifting the activation energy. the activation energy of a reaction is 19.0 kj/mol. effect of enzymes and catalysts. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the.. Catalyst Affect Activation Energy Of A Reaction.
From courses.lumenlearning.com
Factors Affecting Reaction Rates Chemistry Catalyst Affect Activation Energy Of A Reaction estimate the activation energy for each process, and identify which one involves a catalyst. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. A catalyst lowers the activation energy of a chemical reaction. a catalyst provides an alternative route for the reaction with a lower. Catalyst Affect Activation Energy Of A Reaction.
From dxooagcgl.blob.core.windows.net
How Does The Presence Of A Catalyst Affect The Activation Energy Of A Reaction at Debbie Catalyst Affect Activation Energy Of A Reaction It does not lower the. A catalyst provides an alternative route for the reaction. estimate the activation energy for each process, and identify which one involves a catalyst. adding a catalyst has exactly this effect of shifting the activation energy. a catalyst provides an alternative route for the reaction with a lower activation energy. effect of. Catalyst Affect Activation Energy Of A Reaction.
From www.mometrix.com
What is a Catalyst? Chemistry Review (Video) Catalyst Affect Activation Energy Of A Reaction catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. A catalyst lowers the activation energy of a chemical reaction. A catalyst provides an alternative route for the reaction. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate,. Catalyst Affect Activation Energy Of A Reaction.
From www.youtube.com
Catalyst Affects Reaction Rate Energy Diagram with a Catalyst YouTube Catalyst Affect Activation Energy Of A Reaction catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. It does not lower the. When a catalyst is used at a particular concentration, the rate. adding a catalyst has exactly this effect of shifting the activation energy. A catalyst provides an alternative route for the reaction.. Catalyst Affect Activation Energy Of A Reaction.
From www.expii.com
Catalysts (Enzymes) — Overview & Examples Expii Catalyst Affect Activation Energy Of A Reaction It does not lower the. A catalyst provides an alternative route for the reaction. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. A catalyst lowers the activation energy of a chemical reaction. effect of enzymes and catalysts. adding a catalyst has exactly this effect of shifting. Catalyst Affect Activation Energy Of A Reaction.
From www.chemengonline.com
Catalysis Fundamentals Chemical Engineering Page 1 Catalyst Affect Activation Energy Of A Reaction It does not lower the. When a catalyst is used at a particular concentration, the rate. A catalyst lowers the activation energy of a chemical reaction. A catalyst provides an alternative route for the reaction. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. estimate the. Catalyst Affect Activation Energy Of A Reaction.
From masterconceptsinchemistry.com
What’s reaction rate? How temperature, concentration, and catalyst affect it. Catalyst Affect Activation Energy Of A Reaction It does not lower the. estimate the activation energy for each process, and identify which one involves a catalyst. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. When a catalyst is used at a particular concentration, the rate. A catalyst provides an alternative route for. Catalyst Affect Activation Energy Of A Reaction.
From byjus.com
How does catalyst affect activation energy? Catalyst Affect Activation Energy Of A Reaction estimate the activation energy for each process, and identify which one involves a catalyst. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. a catalyst provides an alternative route for the reaction with a lower activation energy. the activation energy of a reaction is. Catalyst Affect Activation Energy Of A Reaction.
From dxooagcgl.blob.core.windows.net
How Does The Presence Of A Catalyst Affect The Activation Energy Of A Reaction at Debbie Catalyst Affect Activation Energy Of A Reaction It does not lower the. A catalyst provides an alternative route for the reaction. estimate the activation energy for each process, and identify which one involves a catalyst. a catalyst provides an alternative route for the reaction with a lower activation energy. When a catalyst is used at a particular concentration, the rate. adding a catalyst has. Catalyst Affect Activation Energy Of A Reaction.
From dxooagcgl.blob.core.windows.net
How Does The Presence Of A Catalyst Affect The Activation Energy Of A Reaction at Debbie Catalyst Affect Activation Energy Of A Reaction A catalyst provides an alternative route for the reaction. A catalyst lowers the activation energy of a chemical reaction. a catalyst provides an alternative route for the reaction with a lower activation energy. effect of enzymes and catalysts. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the.. Catalyst Affect Activation Energy Of A Reaction.
From dxooagcgl.blob.core.windows.net
How Does The Presence Of A Catalyst Affect The Activation Energy Of A Reaction at Debbie Catalyst Affect Activation Energy Of A Reaction It does not lower the. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. effect of enzymes and catalysts. A catalyst lowers the activation energy of a chemical reaction. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to. Catalyst Affect Activation Energy Of A Reaction.
From telegra.ph
Describe effect catalyst activation energy reaction rate Telegraph Catalyst Affect Activation Energy Of A Reaction A catalyst provides an alternative route for the reaction. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. effect of enzymes and catalysts. When a catalyst is used at a particular concentration, the rate. It does not lower the. estimate the activation energy for each. Catalyst Affect Activation Energy Of A Reaction.
From byjus.com
A catalyst lowers the activation energy of the forward reaction by 20 kJ/ mol. It also changes Catalyst Affect Activation Energy Of A Reaction It does not lower the. effect of enzymes and catalysts. a catalyst provides an alternative route for the reaction with a lower activation energy. estimate the activation energy for each process, and identify which one involves a catalyst. A catalyst lowers the activation energy of a chemical reaction. catalysts increase the rates of reactions by providing. Catalyst Affect Activation Energy Of A Reaction.
From exodjtdpy.blob.core.windows.net
Enzymes Raise The Activation Energy Of A Reaction at Sean Ferguson blog Catalyst Affect Activation Energy Of A Reaction catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. a catalyst provides an alternative route for the reaction with a lower activation energy. When a catalyst. Catalyst Affect Activation Energy Of A Reaction.
From nesslabs.com
Activation energy the chemistry of getting started Ness Labs Catalyst Affect Activation Energy Of A Reaction It does not lower the. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. estimate the activation energy for each process, and identify which one involves a catalyst. effect of enzymes and catalysts. A catalyst provides an alternative route for the reaction. in the. Catalyst Affect Activation Energy Of A Reaction.
From as-bio-and-chem.blogspot.com
Bio+Chem Notes. ^^ Recapping Rates of Reaction Catalyst Affect Activation Energy Of A Reaction adding a catalyst has exactly this effect of shifting the activation energy. When a catalyst is used at a particular concentration, the rate. A catalyst lowers the activation energy of a chemical reaction. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. estimate the activation energy for. Catalyst Affect Activation Energy Of A Reaction.
From www.cheric.org
Chemical Reaction (Reaction rate) Catalyst Affect Activation Energy Of A Reaction It does not lower the. A catalyst provides an alternative route for the reaction. estimate the activation energy for each process, and identify which one involves a catalyst. the activation energy of a reaction is 19.0 kj/mol. When a catalyst is used at a particular concentration, the rate. adding a catalyst has exactly this effect of shifting. Catalyst Affect Activation Energy Of A Reaction.
From philschatz.com
Catalysis · Chemistry Catalyst Affect Activation Energy Of A Reaction catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. A catalyst provides an alternative route for the reaction. adding a catalyst has exactly this effect of shifting the activation energy. A catalyst lowers the activation energy of a chemical reaction. estimate the activation energy for. Catalyst Affect Activation Energy Of A Reaction.
From sciencenotes.org
Factors That Affect Reaction Rate Chemical Catalyst Affect Activation Energy Of A Reaction adding a catalyst has exactly this effect of shifting the activation energy. the activation energy of a reaction is 19.0 kj/mol. a catalyst provides an alternative route for the reaction with a lower activation energy. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the.. Catalyst Affect Activation Energy Of A Reaction.
From www.slideserve.com
PPT Mechanisms of Catalytic Reactions and Characterization of Catalysts PowerPoint Catalyst Affect Activation Energy Of A Reaction the activation energy of a reaction is 19.0 kj/mol. estimate the activation energy for each process, and identify which one involves a catalyst. It does not lower the. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. When a catalyst is used at a particular. Catalyst Affect Activation Energy Of A Reaction.
From chem.libretexts.org
Catalytic Hydrogenation of Alkenes Chemistry LibreTexts Catalyst Affect Activation Energy Of A Reaction It does not lower the. A catalyst lowers the activation energy of a chemical reaction. A catalyst provides an alternative route for the reaction. adding a catalyst has exactly this effect of shifting the activation energy. effect of enzymes and catalysts. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to. Catalyst Affect Activation Energy Of A Reaction.
From www.researchgate.net
Reaction coordinate diagram showing the working principle of a catalyst. Download Scientific Catalyst Affect Activation Energy Of A Reaction adding a catalyst has exactly this effect of shifting the activation energy. in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. A catalyst lowers the activation. Catalyst Affect Activation Energy Of A Reaction.
From exorsjyuc.blob.core.windows.net
Catalyst Reaction Meaning at Jarred Mikula blog Catalyst Affect Activation Energy Of A Reaction a catalyst provides an alternative route for the reaction with a lower activation energy. When a catalyst is used at a particular concentration, the rate. the activation energy of a reaction is 19.0 kj/mol. A catalyst provides an alternative route for the reaction. adding a catalyst has exactly this effect of shifting the activation energy. in. Catalyst Affect Activation Energy Of A Reaction.
From www.researchgate.net
Effect of catalyst on energy diagram profile. Download Scientific Diagram Catalyst Affect Activation Energy Of A Reaction effect of enzymes and catalysts. A catalyst provides an alternative route for the reaction. A catalyst lowers the activation energy of a chemical reaction. adding a catalyst has exactly this effect of shifting the activation energy. When a catalyst is used at a particular concentration, the rate. estimate the activation energy for each process, and identify which. Catalyst Affect Activation Energy Of A Reaction.
From slideplayer.com
Lecture 1405 Reaction Mechanism and Catalysis ppt download Catalyst Affect Activation Energy Of A Reaction the activation energy of a reaction is 19.0 kj/mol. When a catalyst is used at a particular concentration, the rate. a catalyst provides an alternative route for the reaction with a lower activation energy. A catalyst provides an alternative route for the reaction. in the case of a biological reaction, when an enzyme (a form of catalyst). Catalyst Affect Activation Energy Of A Reaction.
From saylordotorg.github.io
Catalysis Catalyst Affect Activation Energy Of A Reaction in the case of a biological reaction, when an enzyme (a form of catalyst) binds to a substrate, the. A catalyst lowers the activation energy of a chemical reaction. A catalyst provides an alternative route for the reaction. It does not lower the. adding a catalyst has exactly this effect of shifting the activation energy. a catalyst. Catalyst Affect Activation Energy Of A Reaction.
From wou.edu
Chapter 7 Catalytic Mechanisms of Enzymes Chemistry Catalyst Affect Activation Energy Of A Reaction adding a catalyst has exactly this effect of shifting the activation energy. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. It does not lower the. A catalyst lowers the activation energy of a chemical reaction. A catalyst provides an alternative route for the reaction. . Catalyst Affect Activation Energy Of A Reaction.
From blogs.glowscotland.org.uk
Activation Energy Higher Chemistry Unit 1 Catalyst Affect Activation Energy Of A Reaction When a catalyst is used at a particular concentration, the rate. effect of enzymes and catalysts. the activation energy of a reaction is 19.0 kj/mol. It does not lower the. a catalyst provides an alternative route for the reaction with a lower activation energy. A catalyst provides an alternative route for the reaction. estimate the activation. Catalyst Affect Activation Energy Of A Reaction.
From www.researchgate.net
Free energy of activation of uncatalyzed and catalyzed reactions. Download Scientific Diagram Catalyst Affect Activation Energy Of A Reaction When a catalyst is used at a particular concentration, the rate. the activation energy of a reaction is 19.0 kj/mol. catalysts increase the rates of reactions by providing a new mechanism that has a smaller activation energy, as shown in the. A catalyst lowers the activation energy of a chemical reaction. estimate the activation energy for each. Catalyst Affect Activation Energy Of A Reaction.