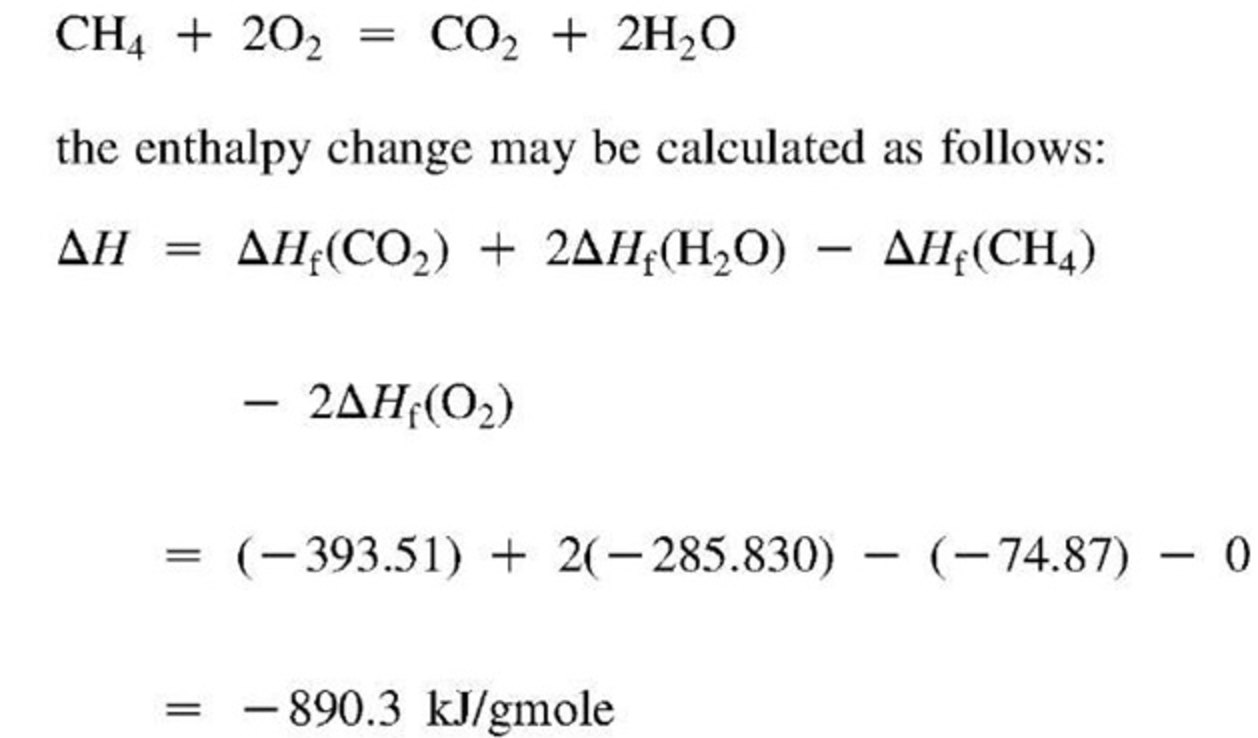Standard Enthalpy Heat Of Formation Water . The standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. The elemental form of each atom is that with the lowest enthalpy in the standard state. A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change Fortunately hess' law makes it possible to list a single value, the standard enthalpy of formation δh f, for each compound. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. The standard state heat of formation for the elemental form of each atom is zero.
from storage.googleapis.com
Fortunately hess' law makes it possible to list a single value, the standard enthalpy of formation δh f, for each compound. A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. The standard state heat of formation for the elemental form of each atom is zero. The elemental form of each atom is that with the lowest enthalpy in the standard state. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,.
Standard Enthalpy Of Formation Sulfuric Acid
Standard Enthalpy Heat Of Formation Water The standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. The elemental form of each atom is that with the lowest enthalpy in the standard state. The standard state heat of formation for the elemental form of each atom is zero. Fortunately hess' law makes it possible to list a single value, the standard enthalpy of formation δh f, for each compound. A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change The standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under.
From www.slideshare.net
Lect w10 abbrev_ thermochemistry_alg Standard Enthalpy Heat Of Formation Water 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The standard state heat of formation for the elemental form of each atom. Standard Enthalpy Heat Of Formation Water.
From pdfprof.com
enthalpies standard de formation et entropie standard Standard Enthalpy Heat Of Formation Water The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. The elemental form of each atom is that with the lowest enthalpy in the standard state. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of. Standard Enthalpy Heat Of Formation Water.
From www.slideshare.net
Tang 03 enthalpy of formation and combustion Standard Enthalpy Heat Of Formation Water Fortunately hess' law makes it possible to list a single value, the standard enthalpy of formation δh f, for each compound. A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The standard state heat of formation for the elemental form of each atom is zero.. Standard Enthalpy Heat Of Formation Water.
From www.chegg.com
Solved Use the standard enthalpies of formation in the table Standard Enthalpy Heat Of Formation Water The enthalpy of formation (\(δh_{f}\)) is the enthalpy change The elemental form of each atom is that with the lowest enthalpy in the standard state. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or. Standard Enthalpy Heat Of Formation Water.
From www.toppr.com
Standard Enthalpy (Heat) of formation of liquid water at 25^o C is Standard Enthalpy Heat Of Formation Water The enthalpy of formation (\(δh_{f}\)) is the enthalpy change The standard state heat of formation for the elemental form of each atom is zero. A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The elemental form of each atom is that with the lowest enthalpy. Standard Enthalpy Heat Of Formation Water.
From www.fatherskit.co
combustion of glucose enthalpy calculate the heat of combustion Standard Enthalpy Heat Of Formation Water 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard state heat of formation for the elemental form of each atom is zero. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,.. Standard Enthalpy Heat Of Formation Water.
From www.slideserve.com
PPT Standard Enthalpies of Formation PowerPoint Presentation, free Standard Enthalpy Heat Of Formation Water The standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. The standard state heat of. Standard Enthalpy Heat Of Formation Water.
From www.slideshare.net
Tang 03 enthalpy of formation and combustion Standard Enthalpy Heat Of Formation Water The enthalpy of formation (\(δh_{f}\)) is the enthalpy change The elemental form of each atom is that with the lowest enthalpy in the standard state. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. A standard enthalpy of formation $δh _f$ is an enthalpy change for. Standard Enthalpy Heat Of Formation Water.
From joizhadcr.blob.core.windows.net
Standard Enthalpy Of Formation Hcl Aq at Denise Lewis blog Standard Enthalpy Heat Of Formation Water The enthalpy of formation (\(δh_{f}\)) is the enthalpy change The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Fortunately hess' law makes it. Standard Enthalpy Heat Of Formation Water.
From www.slideshare.net
Tang 03 enthalpy of formation and combustion Standard Enthalpy Heat Of Formation Water The standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. The elemental form of each atom is that with the lowest enthalpy in the standard state. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change 136 rows. Standard Enthalpy Heat Of Formation Water.
From loetgxopm.blob.core.windows.net
Water Standard Enthalpy Of Formation at Melisa Ramirez blog Standard Enthalpy Heat Of Formation Water The standard state heat of formation for the elemental form of each atom is zero. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. The elemental form of each atom is that with the lowest enthalpy in the standard state. The standard enthalpy of formation, also known. Standard Enthalpy Heat Of Formation Water.
From www.congress-intercultural.eu
Different Types Of Heat (Enthalpy) Of Reaction Read, 42 OFF Standard Enthalpy Heat Of Formation Water Fortunately hess' law makes it possible to list a single value, the standard enthalpy of formation δh f, for each compound. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of. Standard Enthalpy Heat Of Formation Water.
From brunofuga.adv.br
Standard Enthalpy Of Formation Definition, Table, Equation, 46 OFF Standard Enthalpy Heat Of Formation Water The standard state heat of formation for the elemental form of each atom is zero. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,.. Standard Enthalpy Heat Of Formation Water.
From www.chegg.com
Solved The standard enthalpy of formation of H2O(g) at 298 K Standard Enthalpy Heat Of Formation Water The elemental form of each atom is that with the lowest enthalpy in the standard state. The standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. Fortunately hess' law makes it possible to list a single value,. Standard Enthalpy Heat Of Formation Water.
From narodnatribuna.info
Heat Of Formation And Enthalpy Data For Slag Compounds Standard Enthalpy Heat Of Formation Water The standard state heat of formation for the elemental form of each atom is zero. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The standard enthalpy of formation, also known as the heat of formation,. Standard Enthalpy Heat Of Formation Water.
From www.nagwa.com
Question Video Calculating the Standard Enthalpy of Formation for Standard Enthalpy Heat Of Formation Water 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change The elemental form of each atom is that with the lowest enthalpy in the standard state. The standard enthalpy of formation, also known as the heat of formation,. Standard Enthalpy Heat Of Formation Water.
From www.chegg.com
TABLE A286 Enthalpy of formation, Gibbs function of Standard Enthalpy Heat Of Formation Water The elemental form of each atom is that with the lowest enthalpy in the standard state. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. Fortunately hess' law makes it possible to list a single value, the standard enthalpy of formation δh f, for each compound.. Standard Enthalpy Heat Of Formation Water.
From byjus.com
The standard molar heat for formation ofethane, carbondioxide and water Standard Enthalpy Heat Of Formation Water The standard state heat of formation for the elemental form of each atom is zero. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance. Standard Enthalpy Heat Of Formation Water.
From www.toppr.com
Standard Enthalpy (Heat) of formation of liquid water at 25^o C is Standard Enthalpy Heat Of Formation Water A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard state heat. Standard Enthalpy Heat Of Formation Water.
From www.chegg.com
Solved 13. The standard enthalpies of formation for several Standard Enthalpy Heat Of Formation Water 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. The standard enthalpy of formation, also known as the heat of formation, is the enthalpy change when 1 mole of a pure substance forms from its constituent elements at standard temperature and pressure. A standard enthalpy of formation. Standard Enthalpy Heat Of Formation Water.
From www.slideserve.com
PPT Molar Enthalpy PowerPoint Presentation, free download ID6624841 Standard Enthalpy Heat Of Formation Water The enthalpy of formation (\(δh_{f}\)) is the enthalpy change 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. 136 rows standard enthalpy change. Standard Enthalpy Heat Of Formation Water.
From loetgxopm.blob.core.windows.net
Water Standard Enthalpy Of Formation at Melisa Ramirez blog Standard Enthalpy Heat Of Formation Water The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard state heat of formation for the elemental form of each atom is. Standard Enthalpy Heat Of Formation Water.
From byjus.com
21. At 300 K standard enthalpy of formation of C6H5COOH(s), CO2(g), and Standard Enthalpy Heat Of Formation Water The standard state heat of formation for the elemental form of each atom is zero. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under.. Standard Enthalpy Heat Of Formation Water.
From cewuaeqb.blob.core.windows.net
How Do You Work Out Standard Enthalpy Of Formation at Connie Stroud blog Standard Enthalpy Heat Of Formation Water 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard state heat of formation for the elemental form of each atom is zero. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,.. Standard Enthalpy Heat Of Formation Water.
From www.youtube.com
CHEMISTRY 101 Standard enthalpies of formation and reaction YouTube Standard Enthalpy Heat Of Formation Water The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. The elemental form of each atom is that with the lowest enthalpy in the standard state. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of. Standard Enthalpy Heat Of Formation Water.
From storage.googleapis.com
Standard Enthalpy Of Formation Sulfuric Acid Standard Enthalpy Heat Of Formation Water Fortunately hess' law makes it possible to list a single value, the standard enthalpy of formation δh f, for each compound. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data. Standard Enthalpy Heat Of Formation Water.
From printablevascelomgm.z13.web.core.windows.net
How To Determine The Heat Of Formation Standard Enthalpy Heat Of Formation Water 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. 136 rows standard enthalpy change of formation (data table) these tables include heat. Standard Enthalpy Heat Of Formation Water.
From rayb78.github.io
Heat Of Formation Chart Standard Enthalpy Heat Of Formation Water A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. The standard state heat of formation for the elemental form of each atom. Standard Enthalpy Heat Of Formation Water.
From www.chegg.com
Solved Use a standard enthalpies of formation table to Standard Enthalpy Heat Of Formation Water The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. The elemental form of each atom is that with the lowest enthalpy in the standard state. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change The standard state heat of formation for the elemental form of each. Standard Enthalpy Heat Of Formation Water.
From svanfitz.blogspot.com
Enthalpy Change Of Combustion PPT ENTHALPY OF FORMATION Combustion Standard Enthalpy Heat Of Formation Water The enthalpy of formation (\(δh_{f}\)) is the enthalpy change 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The elemental form of each atom is that with the lowest enthalpy in the standard state. Fortunately hess' law makes it possible to list a single value, the. Standard Enthalpy Heat Of Formation Water.
From www.researchgate.net
Heat of formation and enthalpy data for slag compounds Enthalpy of Standard Enthalpy Heat Of Formation Water 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change The standard state heat of formation. Standard Enthalpy Heat Of Formation Water.
From www.chegg.com
Solved The standard enthalpy (or heat) of reaction, delta H, Standard Enthalpy Heat Of Formation Water 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard enthalpy of formation is a measure of the energy released or consumed when one mole of a substance is created under. Fortunately hess' law makes it possible to list a single value, the standard enthalpy. Standard Enthalpy Heat Of Formation Water.
From www.youtube.com
Enthalpies of Formation Chemsitry Tutorial YouTube Standard Enthalpy Heat Of Formation Water A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat. Standard Enthalpy Heat Of Formation Water.
From byjus.com
under the same conditions heat of formation of water and CO2 are 285 Standard Enthalpy Heat Of Formation Water 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Fortunately hess' law makes it possible to list a single value, the standard enthalpy of formation δh f, for each compound. The elemental form of each atom is that with the lowest enthalpy in the standard state.. Standard Enthalpy Heat Of Formation Water.
From storage.googleapis.com
Standard Enthalpy Of Formation Sulfuric Acid Standard Enthalpy Heat Of Formation Water The standard state heat of formation for the elemental form of each atom is zero. A standard enthalpy of formation $δh _f$ is an enthalpy change for a reaction in which exactly 1 mole of a pure substance is. 136 rows standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of. Standard Enthalpy Heat Of Formation Water.