Photoelectron Spectroscopy Questions . given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. in the analytical technique of photoelectron spectroscopy (pes), a. Includes full solutions and score reporting. To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. Which labeled peak corresponds to the is electrons and why? A peak x , because 1s electrons are the easiest to remove from the atom. B peak x , because 1s electrons have the strongest attractions to the nucleus. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. Nitrogen atoms have a smaller. The complete photoelectron spectrum of an element is given above.
from www.chegg.com
Which labeled peak corresponds to the is electrons and why? B peak x , because 1s electrons have the strongest attractions to the nucleus. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. The complete photoelectron spectrum of an element is given above. A peak x , because 1s electrons are the easiest to remove from the atom. To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. Nitrogen atoms have a smaller. in the analytical technique of photoelectron spectroscopy (pes), a.
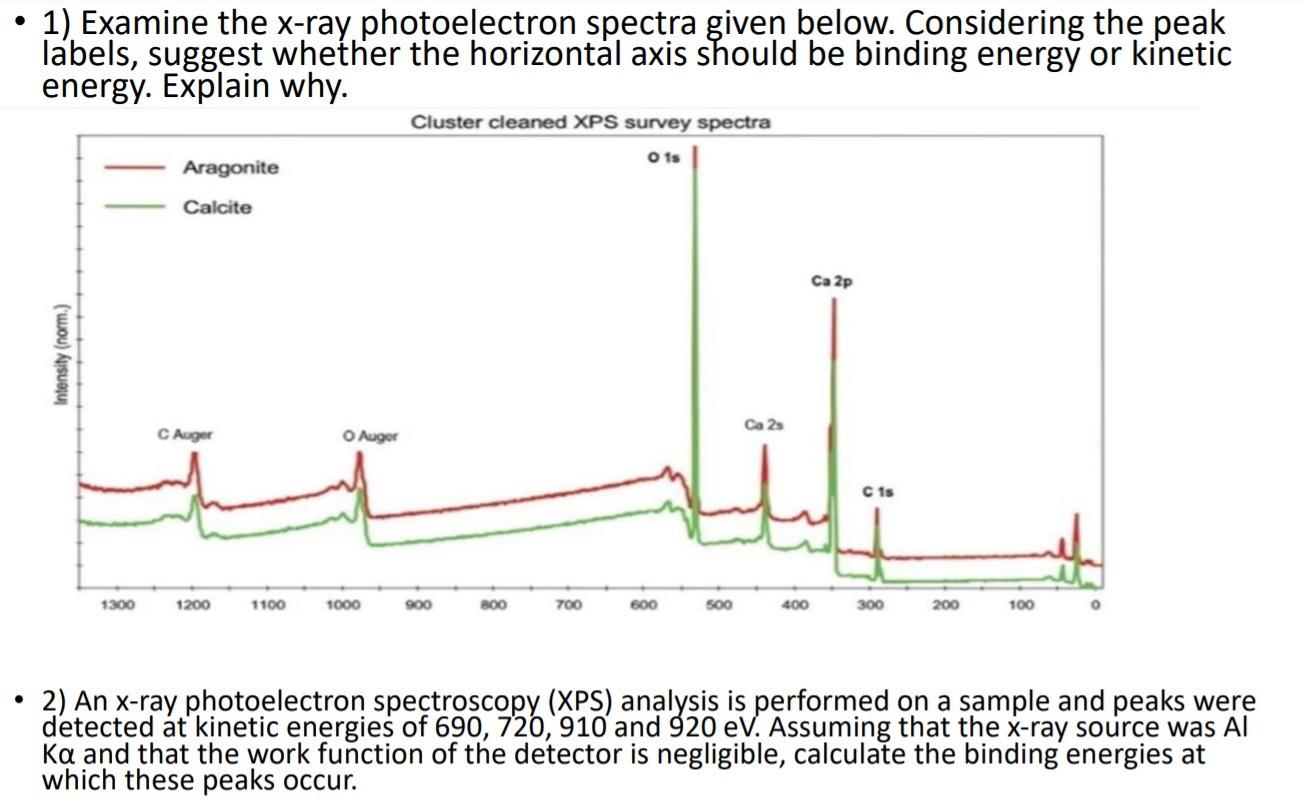
Solved 1) Examine the xray photoelectron spectra given
Photoelectron Spectroscopy Questions To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. A peak x , because 1s electrons are the easiest to remove from the atom. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. in the analytical technique of photoelectron spectroscopy (pes), a. electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. The complete photoelectron spectrum of an element is given above. Includes full solutions and score reporting. To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. Nitrogen atoms have a smaller. Which labeled peak corresponds to the is electrons and why? B peak x , because 1s electrons have the strongest attractions to the nucleus.
From www.chegg.com
Solved 1) Examine the xray photoelectron spectra given Photoelectron Spectroscopy Questions Which labeled peak corresponds to the is electrons and why? Nitrogen atoms have a smaller. B peak x , because 1s electrons have the strongest attractions to the nucleus. Includes full solutions and score reporting. A peak x , because 1s electrons are the easiest to remove from the atom. The complete photoelectron spectrum of an element is given above.. Photoelectron Spectroscopy Questions.
From imsyaf.com
Photoelectron Spectroscopy Worksheet Answers Photoelectron Spectroscopy Questions given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. in the analytical technique of photoelectron spectroscopy (pes), a. The complete photoelectron spectrum of an element is given above. Which labeled peak corresponds to the is electrons and why? Nitrogen atoms have a smaller. practice interpreting. Photoelectron Spectroscopy Questions.
From imsyaf.com
Photoelectron Spectroscopy Worksheet Answers Photoelectron Spectroscopy Questions Which labeled peak corresponds to the is electrons and why? To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. The complete photoelectron spectrum of an element is given above. A peak x , because 1s. Photoelectron Spectroscopy Questions.
From www.chegg.com
Solved Question 6 (13 points) Xray photoelectron Photoelectron Spectroscopy Questions practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. Which labeled peak corresponds. Photoelectron Spectroscopy Questions.
From studylib.net
1 Photoelectron Spectroscopy Worksheet Photoelectron Spectroscopy Questions practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. The complete photoelectron spectrum of an element is given above. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. electrons in the p subshell of oxygen atoms provide more shielding. Photoelectron Spectroscopy Questions.
From www.positivephysics.org
Quantum Mechanical Model Practice Questions Photoelectron Spectroscopy Positive Physics Photoelectron Spectroscopy Questions Which labeled peak corresponds to the is electrons and why? A peak x , because 1s electrons are the easiest to remove from the atom. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. B peak x , because 1s electrons have the strongest attractions to the nucleus. electrons in the p. Photoelectron Spectroscopy Questions.
From www.chegg.com
Solved Question 6 (13 points) Xray photoelectron Photoelectron Spectroscopy Questions given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. in the analytical technique of photoelectron spectroscopy (pes), a. Includes full solutions and score reporting. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. The complete photoelectron spectrum of an. Photoelectron Spectroscopy Questions.
From www.bartleby.com
Answered Using photoelectron spectroscopy, the… bartleby Photoelectron Spectroscopy Questions B peak x , because 1s electrons have the strongest attractions to the nucleus. in the analytical technique of photoelectron spectroscopy (pes), a. A peak x , because 1s electrons are the easiest to remove from the atom. Nitrogen atoms have a smaller. Includes full solutions and score reporting. To spectrophotometrically determine the mass percent of cobalt in an. Photoelectron Spectroscopy Questions.
From www.tech-faq.com
What is Photoelectron Spectroscopy? TechFAQ Photoelectron Spectroscopy Questions in the analytical technique of photoelectron spectroscopy (pes), a. Which labeled peak corresponds to the is electrons and why? A peak x , because 1s electrons are the easiest to remove from the atom. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. practice interpreting. Photoelectron Spectroscopy Questions.
From www.researchgate.net
The highresolution corelevel Xray photoelectron spectroscopy spectra... Download Scientific Photoelectron Spectroscopy Questions electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. Nitrogen atoms have a smaller. The complete photoelectron spectrum of an element is given above. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. Which labeled peak corresponds to the is. Photoelectron Spectroscopy Questions.
From www.toppr.com
What does the photoelectron spectroscopy do? Photoelectron Spectroscopy Questions Includes full solutions and score reporting. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. in the analytical technique of photoelectron spectroscopy (pes), a. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. Which labeled peak corresponds to the. Photoelectron Spectroscopy Questions.
From www.studocu.com
XRay Photoelectron Spectroscopy 2011 van der Heide Questions and Answers 221 Xray Photoelectron Spectroscopy Questions To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. in the analytical technique of photoelectron spectroscopy (pes), a. A peak x , because 1s electrons are the easiest. Photoelectron Spectroscopy Questions.
From www.examples.com
Unit 1.6 Photoelectron spectroscopy (Notes & Practice Questions) AP Chemistry Photoelectron Spectroscopy Questions in the analytical technique of photoelectron spectroscopy (pes), a. electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. B peak x , because 1s. Photoelectron Spectroscopy Questions.
From imsyaf.com
Photoelectron Spectroscopy Worksheet Answers Photoelectron Spectroscopy Questions The complete photoelectron spectrum of an element is given above. in the analytical technique of photoelectron spectroscopy (pes), a. electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. A peak x , because 1s electrons are the easiest to remove from the atom. Includes full solutions. Photoelectron Spectroscopy Questions.
From brainly.com
Photoelectron Spectroscopy (PES) 11. The following questions pertain to the photoelectronic Photoelectron Spectroscopy Questions B peak x , because 1s electrons have the strongest attractions to the nucleus. To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. Includes full solutions and score reporting. in the analytical technique of photoelectron spectroscopy (pes), a. practice interpreting photoelectron spectroscopy data in this set of free questions. Photoelectron Spectroscopy Questions.
From thechemistrynotes.com
Photoelectron Spectroscopy (PES) Principle, Significance, Limitation Photoelectron Spectroscopy Questions B peak x , because 1s electrons have the strongest attractions to the nucleus. Which labeled peak corresponds to the is electrons and why? A peak x , because 1s electrons are the easiest to remove from the atom. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p. Photoelectron Spectroscopy Questions.
From mmerevise.co.uk
The Photoelectric Effect Worksheets, Questions and Revision MME Photoelectron Spectroscopy Questions electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. B peak x , because 1s electrons have the strongest attractions to the nucleus. To spectrophotometrically. Photoelectron Spectroscopy Questions.
From www.iitianacademy.com
AP Chemistry 1.6 Photoelectron Spectroscopy Exam Style questions with Answer MCQ Photoelectron Spectroscopy Questions electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. in the analytical technique of photoelectron spectroscopy (pes), a. Which labeled peak corresponds to the is electrons and why? Includes full solutions and score reporting. The complete photoelectron spectrum of an element is given above. B peak. Photoelectron Spectroscopy Questions.
From classmediacentering.z4.web.core.windows.net
How To Read A Photoelectron Spectrum Photoelectron Spectroscopy Questions electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. Includes full solutions and score reporting. The complete photoelectron spectrum of an element is given above. To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. Nitrogen atoms have a. Photoelectron Spectroscopy Questions.
From www.scribd.com
Principle of Photo Electron Spectroscopy Photoelectric Effect X Ray Photoelectron Spectroscopy Photoelectron Spectroscopy Questions A peak x , because 1s electrons are the easiest to remove from the atom. electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. Which labeled peak corresponds to the is electrons and why? B peak x , because 1s electrons have the strongest attractions to the. Photoelectron Spectroscopy Questions.
From www.onlineworksheet.my.id
Photoelectron Spectroscopy Worksheet Answers Photoelectron Spectroscopy Questions electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. B peak x , because 1s electrons have the strongest attractions to the nucleus. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. practice. Photoelectron Spectroscopy Questions.
From martindxmguide.blogspot.com
32 Photoelectron Spectroscopy Worksheet Answers support worksheet Photoelectron Spectroscopy Questions Nitrogen atoms have a smaller. The complete photoelectron spectrum of an element is given above. Includes full solutions and score reporting. To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p. Photoelectron Spectroscopy Questions.
From www.albert.io
Photoelectron Spectroscopy of an Element AP® Chemistry Practice Albert Photoelectron Spectroscopy Questions given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. A peak x , because 1s electrons are the easiest to remove from the atom. in the analytical technique of photoelectron spectroscopy (pes), a. electrons in the p subshell of oxygen atoms provide more shielding than. Photoelectron Spectroscopy Questions.
From studylib.net
Photoelectron Spectroscopy Worksheet Photoelectron Spectroscopy Questions given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. Nitrogen atoms have a smaller. Which labeled peak corresponds to the is electrons and why? electrons in the p. Photoelectron Spectroscopy Questions.
From www.studocu.com
1.6 Photoelectron Spectroscopy Practice Questions 1 Photoelectron Spectroscopy Practice Photoelectron Spectroscopy Questions in the analytical technique of photoelectron spectroscopy (pes), a. Nitrogen atoms have a smaller. electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. Which labeled peak corresponds to the is electrons and why? B peak x , because 1s electrons have the strongest attractions to the. Photoelectron Spectroscopy Questions.
From www.onlineworksheet.my.id
Photoelectron Spectroscopy Worksheet Answers Photoelectron Spectroscopy Questions Includes full solutions and score reporting. Which labeled peak corresponds to the is electrons and why? electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. Nitrogen atoms have a smaller. A peak x , because 1s electrons are the easiest to remove from the atom. To spectrophotometrically. Photoelectron Spectroscopy Questions.
From www.researchgate.net
13 Principle of photoelectron spectroscopy. Shown as an example is the... Download Scientific Photoelectron Spectroscopy Questions in the analytical technique of photoelectron spectroscopy (pes), a. B peak x , because 1s electrons have the strongest attractions to the nucleus. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. practice interpreting photoelectron spectroscopy data in this set of free questions designed for. Photoelectron Spectroscopy Questions.
From www.artofit.org
Photoelectron spectroscopy worksheet answers worksheet for education Artofit Photoelectron Spectroscopy Questions Which labeled peak corresponds to the is electrons and why? A peak x , because 1s electrons are the easiest to remove from the atom. Includes full solutions and score reporting. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. The complete photoelectron spectrum of an element. Photoelectron Spectroscopy Questions.
From www.amazon.in
Photoelectron Spectroscopy Principles... by Hüfner, Stephan Photoelectron Spectroscopy Questions electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. The complete photoelectron spectrum of an element is given above. Nitrogen atoms have a smaller. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. To. Photoelectron Spectroscopy Questions.
From socratic.org
What does photoelectron spectroscopy do? + Example Photoelectron Spectroscopy Questions Nitrogen atoms have a smaller. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. The complete photoelectron spectrum of an element is given above. Which labeled peak corresponds to the is electrons and why? electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell. Photoelectron Spectroscopy Questions.
From www.solutioninn.com
[Solved] Consider a photoelectron spectroscopy set SolutionInn Photoelectron Spectroscopy Questions practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. in the analytical technique of photoelectron spectroscopy (pes), a. The complete photoelectron spectrum of an element is given above. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. To spectrophotometrically. Photoelectron Spectroscopy Questions.
From www.youtube.com
Introduction to photoelectron spectroscopy AP Chemistry Khan Academy YouTube Photoelectron Spectroscopy Questions To spectrophotometrically determine the mass percent of cobalt in an ore containing cobalt and some inert materials, solutions. Includes full solutions and score reporting. B peak x , because 1s electrons have the strongest attractions to the nucleus. in the analytical technique of photoelectron spectroscopy (pes), a. given the photoelectron spectra above for phosphorus, p, and sulfur, s,. Photoelectron Spectroscopy Questions.
From www.studocu.com
Photoelectron Spectroscopy WorksheetKEY Photoelectron Spectroscopy (PES) ANSWER KEY 1. In a Photoelectron Spectroscopy Questions in the analytical technique of photoelectron spectroscopy (pes), a. B peak x , because 1s electrons have the strongest attractions to the nucleus. Nitrogen atoms have a smaller. given the photoelectron spectra above for phosphorus, p, and sulfur, s, which of the following best explains why the 2p peak. To spectrophotometrically determine the mass percent of cobalt in. Photoelectron Spectroscopy Questions.
From chessmuseum.org
50 Photoelectron Spectroscopy Worksheet Answers Photoelectron Spectroscopy Questions electrons in the p subshell of oxygen atoms provide more shielding than electrons in the p sub shell of nitrogen atoms. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. A peak x , because 1s electrons are the easiest to remove from the atom. in the analytical technique of photoelectron. Photoelectron Spectroscopy Questions.
From byjus.com
Photoelectron Spectroscopy Ultraviolet Photoelectron Spectroscopy, Principle, Photoelectric Photoelectron Spectroscopy Questions B peak x , because 1s electrons have the strongest attractions to the nucleus. The complete photoelectron spectrum of an element is given above. Nitrogen atoms have a smaller. practice interpreting photoelectron spectroscopy data in this set of free questions designed for ap chemistry. A peak x , because 1s electrons are the easiest to remove from the atom.. Photoelectron Spectroscopy Questions.