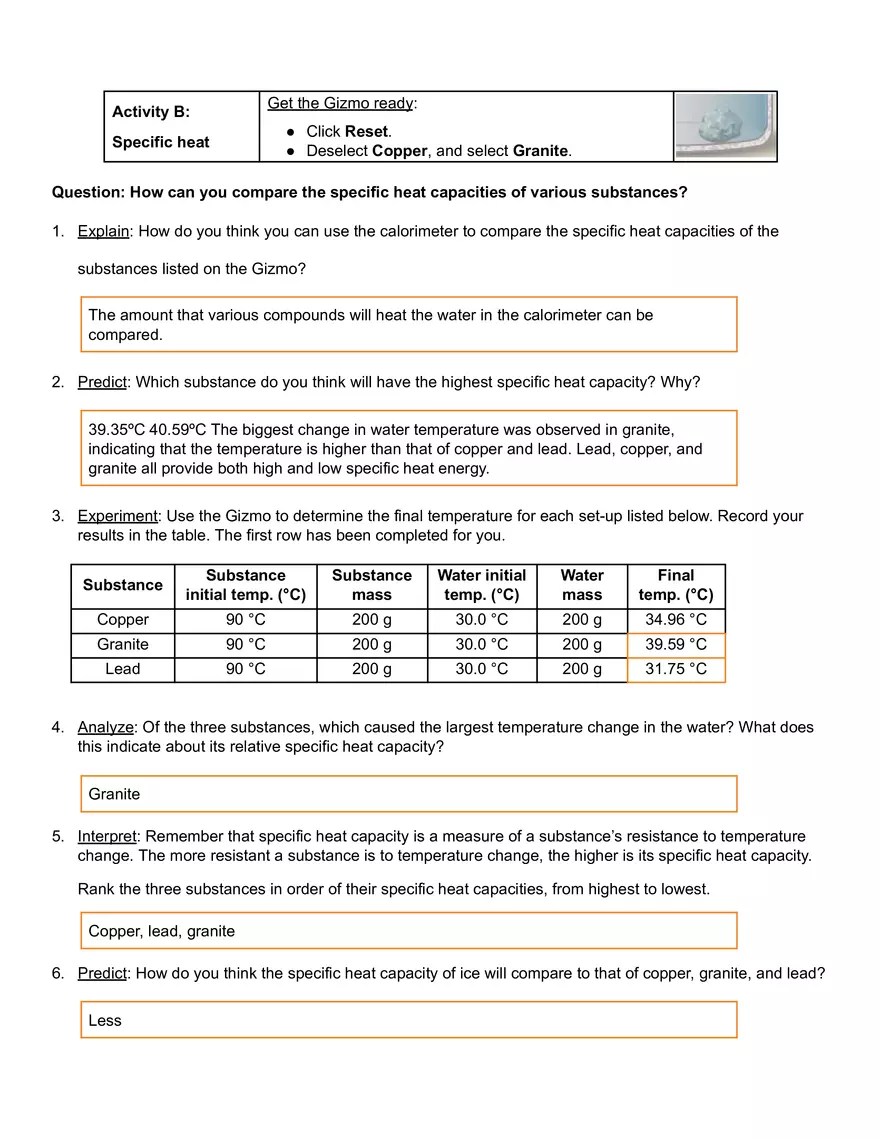
Calorimetry Lab Questions And Answers . If the final temperature of the. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. How many grams of water. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? A calorimeter is an apparatus used for calculating the heat developed during a. When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. Important calorimeter questions with answers. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar. Do this 2nd, same reason. Record mass of water and calorimeter (without thermometer). 1) what is a calorimeter? If the heat capacity of the calorimeter is 21.6. How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo?
from edubirdie.com
If the final temperature of the. Important calorimeter questions with answers. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? A calorimeter is an apparatus used for calculating the heat developed during a. If the heat capacity of the calorimeter is 21.6. When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. Do this 2nd, same reason. Record mass of water and calorimeter (without thermometer). Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar.
Calorimetry Lab Gizmo Answer Key Edubirdie
Calorimetry Lab Questions And Answers Important calorimeter questions with answers. A calorimeter is an apparatus used for calculating the heat developed during a. How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? Important calorimeter questions with answers. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. Record mass of water and calorimeter (without thermometer). 1) what is a calorimeter? When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. If the heat capacity of the calorimeter is 21.6. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar. How many grams of water. Do this 2nd, same reason. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? If the final temperature of the.
From www.scribd.com
Calorimetry Lab SE Calorie Heat Capacity Calorimetry Lab Questions And Answers A calorimeter is an apparatus used for calculating the heat developed during a. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? Important calorimeter questions with answers. If the heat capacity of the calorimeter is 21.6. 1) what is a calorimeter? How do you think you can use the calorimeter to compare the. Calorimetry Lab Questions And Answers.
From www.studocu.com
Calorimetry Lab SE Lab General Physics 1 Studocu Calorimetry Lab Questions And Answers How many grams of water. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? Record mass of water and calorimeter (without thermometer). Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar. Do this 2nd, same reason.. Calorimetry Lab Questions And Answers.
From studyfinder.org
Unveiling the Secrets of Experiment 25 Calorimetry Prelab Answers Calorimetry Lab Questions And Answers When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. Do this 2nd, same reason. If the heat capacity of the calorimeter is 21.6. If the final temperature of the. How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? Calorimetry. Calorimetry Lab Questions And Answers.
From www.numerade.com
SOLVED Experiment 10 Calorimetry and Specifilc Heat 0f Water Answer Calorimetry Lab Questions And Answers A calorimeter is an apparatus used for calculating the heat developed during a. 1) what is a calorimeter? Record mass of water and calorimeter (without thermometer). 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in. Calorimetry Lab Questions And Answers.
From studylib.net
Calorimetry Worksheet Calorimetry Lab Questions And Answers If the heat capacity of the calorimeter is 21.6. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a. Calorimetry Lab Questions And Answers.
From www.studocu.com
Calorimetry and Specific Heat Lab Question Using a calorimeter, how Calorimetry Lab Questions And Answers If the final temperature of the. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. Important calorimeter questions with answers. If the heat capacity of the calorimeter is 21.6. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? A calorimeter is an apparatus. Calorimetry Lab Questions And Answers.
From www.chegg.com
Solved Experiment 25 Report Sheet Calorimetry / 613 Lob Sec. Calorimetry Lab Questions And Answers How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? Do this 2nd, same reason. When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. Important calorimeter questions with answers. If the final temperature of the. A calorimeter is an apparatus. Calorimetry Lab Questions And Answers.
From www.studypool.com
SOLUTION Gizmo student exploration calorimetry lab answer key Studypool Calorimetry Lab Questions And Answers When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. Do this 2nd, same reason. A calorimeter is an apparatus used for calculating the heat developed during a. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the. Calorimetry Lab Questions And Answers.
From www.learnable.education
Year 11 Chemistry Practical Investigation Calorimetry Experiment Calorimetry Lab Questions And Answers 1) what is a calorimeter? If the final temperature of the. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? Record mass of water and calorimeter (without thermometer). Important calorimeter questions with answers. A calorimeter is an apparatus used for calculating the heat developed during a. When 1.00 g of coal is burned. Calorimetry Lab Questions And Answers.
From www.chegg.com
Solved CALORIMETRY LAB REPORT DATA AND QUESTIONS Calorimetry Calorimetry Lab Questions And Answers Record mass of water and calorimeter (without thermometer). 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? 1) what is a calorimeter? Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14). Calorimetry Lab Questions And Answers.
From quizzlibraryzimmer.z13.web.core.windows.net
Calorimetry Questions And Answers Calorimetry Lab Questions And Answers 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? If the heat capacity of the calorimeter is 21.6. Do this 2nd, same reason. A calorimeter is an apparatus. Calorimetry Lab Questions And Answers.
From www.pinterest.com
Distance Learning Lab Calorimetry and Temperature Distance learning Calorimetry Lab Questions And Answers A calorimeter is an apparatus used for calculating the heat developed during a. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. Important calorimeter questions with answers. If the heat capacity of the calorimeter is 21.6. How do you think you can use the calorimeter to compare the specific heat. Calorimetry Lab Questions And Answers.
From exysaymwb.blob.core.windows.net
Chemistry Simulation Calorimetry Answers at Camille Wright blog Calorimetry Lab Questions And Answers How many grams of water. If the final temperature of the. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar. When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. 1) what is a calorimeter? Record. Calorimetry Lab Questions And Answers.
From www.chegg.com
Solved Calorimetry Lab BEFORE YOU BEGIN post your Calorimetry Lab Questions And Answers If the final temperature of the. Important calorimeter questions with answers. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar. Record mass of water and calorimeter (without thermometer). 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g. Calorimetry Lab Questions And Answers.
From www.studocu.com
CoffeeCup Calorimetry Experiment Lab Report with answers and solutions Calorimetry Lab Questions And Answers How many grams of water. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? If the final temperature of the. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted. Calorimetry Lab Questions And Answers.
From learninglibrarylinton.z21.web.core.windows.net
Calorimetry Questions And Answers Calorimetry Lab Questions And Answers When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. Record mass of water and calorimeter (without thermometer). How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing. Calorimetry Lab Questions And Answers.
From qwivy.com
Calorimetry Lab GIZMO (All answers correct) Calorimetry Lab Questions And Answers How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar. If the heat capacity of the calorimeter is 21.6. Do this 2nd,. Calorimetry Lab Questions And Answers.
From quizzlibraryzimmer.z13.web.core.windows.net
Calorimetry Questions And Answers Calorimetry Lab Questions And Answers A calorimeter is an apparatus used for calculating the heat developed during a. When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. Record mass of water and calorimeter (without thermometer). 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. How much energy. Calorimetry Lab Questions And Answers.
From www.studocu.com
Calorimetry Gizmo Student Exploration Calorimetry Lab Gizmo Warmup Calorimetry Lab Questions And Answers If the final temperature of the. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. 1) what is a calorimeter? Do this 2nd, same reason. A calorimeter is an apparatus used for calculating the heat developed during a. How much energy is needed to change the temperature of 50.0 g. Calorimetry Lab Questions And Answers.
From materialdbtracy.z13.web.core.windows.net
Calorimetry Questions And Answers Calorimetry Lab Questions And Answers Do this 2nd, same reason. If the heat capacity of the calorimeter is 21.6. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. Important calorimeter questions with answers. When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. How many grams of water.. Calorimetry Lab Questions And Answers.
From studyzonejunker.z19.web.core.windows.net
Calorimetry Questions And Answers Calorimetry Lab Questions And Answers How many grams of water. Important calorimeter questions with answers. When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb. Calorimetry Lab Questions And Answers.
From edubirdie.com
Calorimetry Lab Gizmo Answer Key Edubirdie Calorimetry Lab Questions And Answers How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? Record mass of water and calorimeter (without thermometer). How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? Do this 2nd, same reason. 1) what is a calorimeter? A calorimeter is an. Calorimetry Lab Questions And Answers.
From learningmediajacob.z5.web.core.windows.net
Calorimetry Questions And Answers Calorimetry Lab Questions And Answers If the heat capacity of the calorimeter is 21.6. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar. Record mass of water and calorimeter (without thermometer). 1) what is a calorimeter? Important calorimeter questions with answers. 76.7°c a chemist mixes 75.0 g of. Calorimetry Lab Questions And Answers.
From academguide.com
Discovering insights Experiment 25 calorimetry pre lab answers Calorimetry Lab Questions And Answers How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? Important calorimeter questions with answers. If the heat capacity of the calorimeter is 21.6. If the final temperature of the. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at. Calorimetry Lab Questions And Answers.
From www.studypool.com
SOLUTION Student exploration calorimetry lab Studypool Calorimetry Lab Questions And Answers Important calorimeter questions with answers. How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of. Calorimetry Lab Questions And Answers.
From www.coursehero.com
[Solved] Please answer the following questions for a calorimetry lab Calorimetry Lab Questions And Answers How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? Important calorimeter questions with answers. If the final temperature of the. If the heat capacity of the calorimeter is 21.6. Record mass. Calorimetry Lab Questions And Answers.
From www.scribd.com
Answers TC Calorimetry Practice Questions Continuum Mechanics Calorimetry Lab Questions And Answers Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar. If the heat capacity of the calorimeter is 21.6. Do this 2nd, same reason. If the final temperature of the. 1) what is a calorimeter? When 1.00 g of coal is burned in a. Calorimetry Lab Questions And Answers.
From www.docsity.com
Calorimetry lab report Study Guides, Projects, Research Chemistry Calorimetry Lab Questions And Answers How much energy is needed to change the temperature of 50.0 g of water by 15.0oc? When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. A calorimeter is an apparatus used for calculating the heat developed during a. Important calorimeter questions with answers. Record mass of water and calorimeter (without thermometer). 76.7°c. Calorimetry Lab Questions And Answers.
From www.chegg.com
Solved Pre Lab for Calorimetry Introduction Calorimetry is Calorimetry Lab Questions And Answers Do this 2nd, same reason. How many grams of water. A calorimeter is an apparatus used for calculating the heat developed during a. If the final temperature of the. 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. Important calorimeter questions with answers. How much energy is needed to change. Calorimetry Lab Questions And Answers.
From lessonlibcuttlebone.z22.web.core.windows.net
Chemsheets Calorimetry 2 Answers Calorimetry Lab Questions And Answers 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. 1) what is a calorimeter? When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed. Calorimetry Lab Questions And Answers.
From brainly.com
Calorimetry Gizmo The answer for Calorimetry Gizmo this lab is hard to Calorimetry Lab Questions And Answers A calorimeter is an apparatus used for calculating the heat developed during a. If the heat capacity of the calorimeter is 21.6. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water, calculate the molar. How do you think you can use the calorimeter to compare the. Calorimetry Lab Questions And Answers.
From www.stuvia.com
Student Exploration Calorimetry Lab possible Questions And Answers Calorimetry Lab Questions And Answers A calorimeter is an apparatus used for calculating the heat developed during a. 1) what is a calorimeter? 76.7°c a chemist mixes 75.0 g of an unknown substance at 96.5°c with 1,150 g of water at 25.0°c. Calorimetry worksheet 1) if 0.315 moles of hexane (c 6 h 14) is combusted in a bomb calorimeter containing 5.65 liters of water,. Calorimetry Lab Questions And Answers.
From www.chegg.com
Solved The Fundamentals of Calorimetry Prelab Chemical Calorimetry Lab Questions And Answers When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. If the final temperature of the. If the heat capacity of the calorimeter is 21.6. Important calorimeter questions with answers. Do this 2nd, same reason. A calorimeter is an apparatus used for calculating the heat developed during a. How do you think you. Calorimetry Lab Questions And Answers.
From quizzlibraryzimmer.z13.web.core.windows.net
Calorimetry Questions And Answers Calorimetry Lab Questions And Answers 1) what is a calorimeter? How many grams of water. Important calorimeter questions with answers. If the heat capacity of the calorimeter is 21.6. How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? If the final temperature of the. 76.7°c a chemist mixes 75.0 g of an. Calorimetry Lab Questions And Answers.
From www.chegg.com
Solved PreLab Experiment 8; Calorimetry 1. An unknown metal Calorimetry Lab Questions And Answers When 1.00 g of coal is burned in a bomb calorimeter, the temperature increases by 1.48 °c. A calorimeter is an apparatus used for calculating the heat developed during a. How do you think you can use the calorimeter to compare the specific heat capacities of the substances listed on the gizmo? 1) what is a calorimeter? If the heat. Calorimetry Lab Questions And Answers.