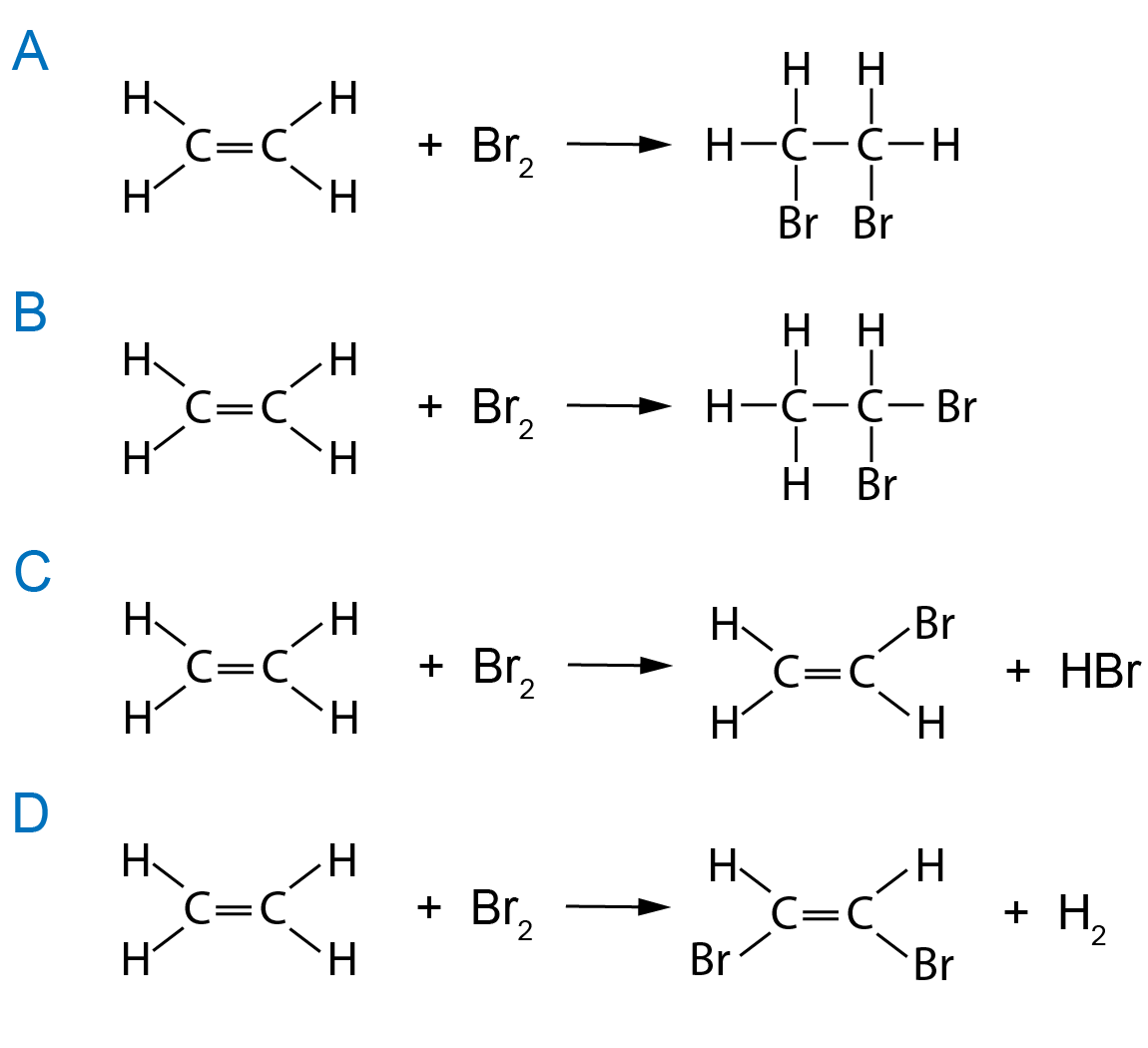Bromine Water Equation . Bromine is in group 17 (the halogens) of the periodic table. The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. Derive chemical equations from narrative descriptions of. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. This test is particularly useful in poc for distinguishing. The solution is known as bromine water. It is a deep reddish brown liquid. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. Like chlorine water, it is a good. By the end of this section, you will be able to: It is soluble in water, forming 'bromine water', a powerful.
from gradegorilla.com
Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Bromine is in group 17 (the halogens) of the periodic table. The solution is known as bromine water. The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). It is soluble in water, forming 'bromine water', a powerful. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. Like chlorine water, it is a good. It is a deep reddish brown liquid. By the end of this section, you will be able to:
GCSE Chemistry Organic Grade Gorilla
Bromine Water Equation Like chlorine water, it is a good. The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. The solution is known as bromine water. It is soluble in water, forming 'bromine water', a powerful. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. Bromine is in group 17 (the halogens) of the periodic table. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). Derive chemical equations from narrative descriptions of. By the end of this section, you will be able to: Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Like chlorine water, it is a good. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. This test is particularly useful in poc for distinguishing. It is a deep reddish brown liquid.
From www.slideserve.com
PPT Alkenes PowerPoint Presentation, free download ID2372306 Bromine Water Equation The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. By the end of this section, you will be able to: Derive chemical equations from narrative descriptions of. This test is particularly. Bromine Water Equation.
From exoquglkq.blob.core.windows.net
Bromine Number Equation at Robert Landers blog Bromine Water Equation Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Derive chemical equations from narrative descriptions of. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. Bromine is in group 17 (the halogens) of the periodic table. It is soluble. Bromine Water Equation.
From www.toppr.com
When phenol is treated with bromine water, while precipitate is Bromine Water Equation Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). It is a deep reddish brown liquid. The solution is known as bromine water. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. This test is particularly useful. Bromine Water Equation.
From byjus.com
How do you test for an alkene? Bromine Water Equation Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Derive chemical equations from narrative descriptions of. Bromine is in group 17 (the halogens) of the periodic table. This test is particularly useful in poc for distinguishing. It is a deep reddish brown liquid. By the end of this. Bromine Water Equation.
From www.numerade.com
SOLVED Using condensed structure, write the balanced chemical equation Bromine Water Equation The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. Like chlorine water, it is a good. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine. Bromine Water Equation.
From exotclnfm.blob.core.windows.net
Bromine Ethyne Equation at William Spence blog Bromine Water Equation Bromine is in group 17 (the halogens) of the periodic table. Like chlorine water, it is a good. The solution is known as bromine water. It is a deep reddish brown liquid. It is soluble in water, forming 'bromine water', a powerful. Derive chemical equations from narrative descriptions of. This test is particularly useful in poc for distinguishing. The bromine. Bromine Water Equation.
From www.numerade.com
SOLVED equation for the reaction of cyclohexane and cyclohexene with Bromine Water Equation Derive chemical equations from narrative descriptions of. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving. Bromine Water Equation.
From byjus.com
What happens when ethane reacts with bromine water? Bromine Water Equation Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Bromine is in group 17 (the halogens) of the periodic table. This test is particularly useful in poc for distinguishing. The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds.. Bromine Water Equation.
From www.coursehero.com
[Solved] what happen if you combine 1 ml bromine water and few drops Bromine Water Equation Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. This test is particularly useful in poc for distinguishing. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. The solution is known as bromine water. Derive chemical equations from narrative. Bromine Water Equation.
From brainly.in
Reaction between sodium iodide and bromine water Brainly.in Bromine Water Equation Bromine is in group 17 (the halogens) of the periodic table. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. Like chlorine water, it is a good. It is a deep reddish brown liquid. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at. Bromine Water Equation.
From scienceready.com.au
Bromine Water Test Science Ready Bromine Water Equation Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The solution is known as bromine water. By the end of this section, you will be able to: The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. Bromine water. Bromine Water Equation.
From www.toppr.com
What happens when phenol reacts with bromine water?Write the chemical Bromine Water Equation It is soluble in water, forming 'bromine water', a powerful. It is a deep reddish brown liquid. The solution is known as bromine water. The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. Derive chemical equations from narrative descriptions of. This test is particularly useful in poc for distinguishing. About. Bromine Water Equation.
From www.coursehero.com
[Solved] Write a chemical equation for the reaction of bromine and Bromine Water Equation About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. Like chlorine water, it is a good. Bromine is in group 17 (the halogens) of the periodic table. It is soluble in water, forming 'bromine water', a powerful. This test is particularly useful in poc for distinguishing. By the end of this. Bromine Water Equation.
From www.slideserve.com
PPT EXTRACTION OF BROMINE FROM SEA WATER PowerPoint Presentation Bromine Water Equation The solution is known as bromine water. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Bromine is in group 17 (the halogens) of the periodic table. Derive chemical equations from narrative descriptions of. Like chlorine water, it is a good. Bromine water solution can be prepared in. Bromine Water Equation.
From www.slideserve.com
PPT EXTRACTION OF BROMINE FROM SEA WATER PowerPoint Presentation Bromine Water Equation Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). This test is particularly useful in poc for distinguishing. The solution is known as bromine water.. Bromine Water Equation.
From bazagabriellehenderson.blogspot.com
Bromine Water Test Equation Gabrielle Henderson Bromine Water Equation About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. Like chlorine water, it is a good. Derive chemical equations from narrative descriptions of. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). By the end of this. Bromine Water Equation.
From exotclnfm.blob.core.windows.net
Bromine Ethyne Equation at William Spence blog Bromine Water Equation This test is particularly useful in poc for distinguishing. Bromine is in group 17 (the halogens) of the periodic table. It is soluble in water, forming 'bromine water', a powerful. Derive chemical equations from narrative descriptions of. It is a deep reddish brown liquid. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of. Bromine Water Equation.
From www.numerade.com
The electrolysis of an aqueous solution of magnesium bromide (MgBr2 Bromine Water Equation It is soluble in water, forming 'bromine water', a powerful. Derive chemical equations from narrative descriptions of. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Bromine is in group 17 (the halogens) of the periodic table. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters. Bromine Water Equation.
From omerchemblog.blogspot.com
Omer Chemistry Blog 3.8 I can describe the reaction between alkenes Bromine Water Equation Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature.. Bromine Water Equation.
From www.youtube.com
How to write the equation for Equation for SrBr2 + H2O (Strontium Bromine Water Equation By the end of this section, you will be able to: The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. This test is particularly useful in poc for distinguishing. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane.. Bromine Water Equation.
From www.nagwa.com
Question Video Identifying the Product of the Reaction of Ethyne Gas Bromine Water Equation This test is particularly useful in poc for distinguishing. It is a deep reddish brown liquid. Derive chemical equations from narrative descriptions of. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of. Bromine Water Equation.
From www.youtube.com
Bromine Water Experiment YouTube Bromine Water Equation Like chlorine water, it is a good. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. Derive chemical equations from narrative descriptions of. The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. Bromine water is a yellow mixture solution with high. Bromine Water Equation.
From www.numerade.com
SOLVED (ii) Aniline reacts with bromine water in aqueous medium (only Bromine Water Equation Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. The solution is known as bromine water. By the end of this section, you will be able to: Bromine is in group 17 (the halogens) of the periodic table. Alkenes react in the cold with pure liquid bromine, or with a. Bromine Water Equation.
From gradegorilla.com
GCSE Chemistry Organic Grade Gorilla Bromine Water Equation Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o).. Bromine Water Equation.
From ppt-online.org
Addition reactions презентация онлайн Bromine Water Equation The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). Derive chemical equations from narrative descriptions of. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters. Bromine Water Equation.
From ar.inspiredpencil.com
Bromine Liquid Formula Bromine Water Equation About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. It is a deep reddish brown liquid. The solution is known as bromine water. It is soluble in water, forming 'bromine water', a powerful. Like chlorine water, it is a good. Derive chemical equations from narrative descriptions of. Bromine water solution can. Bromine Water Equation.
From www.youtube.com
Reactions with Bromine Water YouTube Bromine Water Equation Bromine is in group 17 (the halogens) of the periodic table. The solution is known as bromine water. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). It is soluble in water, forming 'bromine water', a powerful. The bromine water test is a chemical analysis technique. Bromine Water Equation.
From www.youtube.com
How to Write the Formula for Bromine Liquid YouTube Bromine Water Equation Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The solution is known as bromine water. This test is particularly useful in poc for distinguishing. Like chlorine water, it. Bromine Water Equation.
From exotclnfm.blob.core.windows.net
Bromine Ethyne Equation at William Spence blog Bromine Water Equation By the end of this section, you will be able to: Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). The bromine water test is a chemical analysis technique used to identify the presence of unsaturation in organic compounds. Like chlorine water, it is a good.. Bromine Water Equation.
From www.slideserve.com
PPT OCR organic reaction mechanisms PowerPoint Presentation, free Bromine Water Equation This test is particularly useful in poc for distinguishing. It is a deep reddish brown liquid. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by dissolving diatomic bromine (br 2) in water (h 2 o). Bromine is in group 17 (the halogens) of the periodic table. About 3.41 grams (0.12 ounce) of bromine dissolve in 100. Bromine Water Equation.
From exotoiijb.blob.core.windows.net
Bromine Water Gcse at Bonnie Lewis blog Bromine Water Equation Bromine is in group 17 (the halogens) of the periodic table. This test is particularly useful in poc for distinguishing. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. By the end of this section, you will be able to: The solution is known as bromine water. About 3.41 grams. Bromine Water Equation.
From byjus.com
Give a brief description of bromine water test. 5 marks Bromine Water Equation Like chlorine water, it is a good. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters (0.1 quart) of water at room temperature. By the end of this section, you will be able to: Derive chemical equations from narrative. Bromine Water Equation.
From klaehyagl.blob.core.windows.net
Bromine Molecule Formula at Antonio Godines blog Bromine Water Equation Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. It is soluble in water, forming 'bromine water', a powerful. Bromine is in group 17 (the halogens) of the periodic table. Derive chemical equations from narrative descriptions of. By the end of this section, you will be able to: This test. Bromine Water Equation.
From dawson-bogspotsheppard.blogspot.com
Bromine Water Test Equation Bromine Water Equation Bromine is in group 17 (the halogens) of the periodic table. It is soluble in water, forming 'bromine water', a powerful. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The solution is known as bromine water. About 3.41 grams (0.12 ounce) of bromine dissolve in 100 milliliters. Bromine Water Equation.
From molekula.com
Purchase Bromine [7726956] online • Catalog • Molekula Group Bromine Water Equation Like chlorine water, it is a good. Bromine water solution can be prepared in the chemistry lab by direct mixing of fumes of bromine and water. It is soluble in water, forming 'bromine water', a powerful. Bromine is in group 17 (the halogens) of the periodic table. Bromine water is a yellow mixture solution with high oxidizing properties, prepared by. Bromine Water Equation.