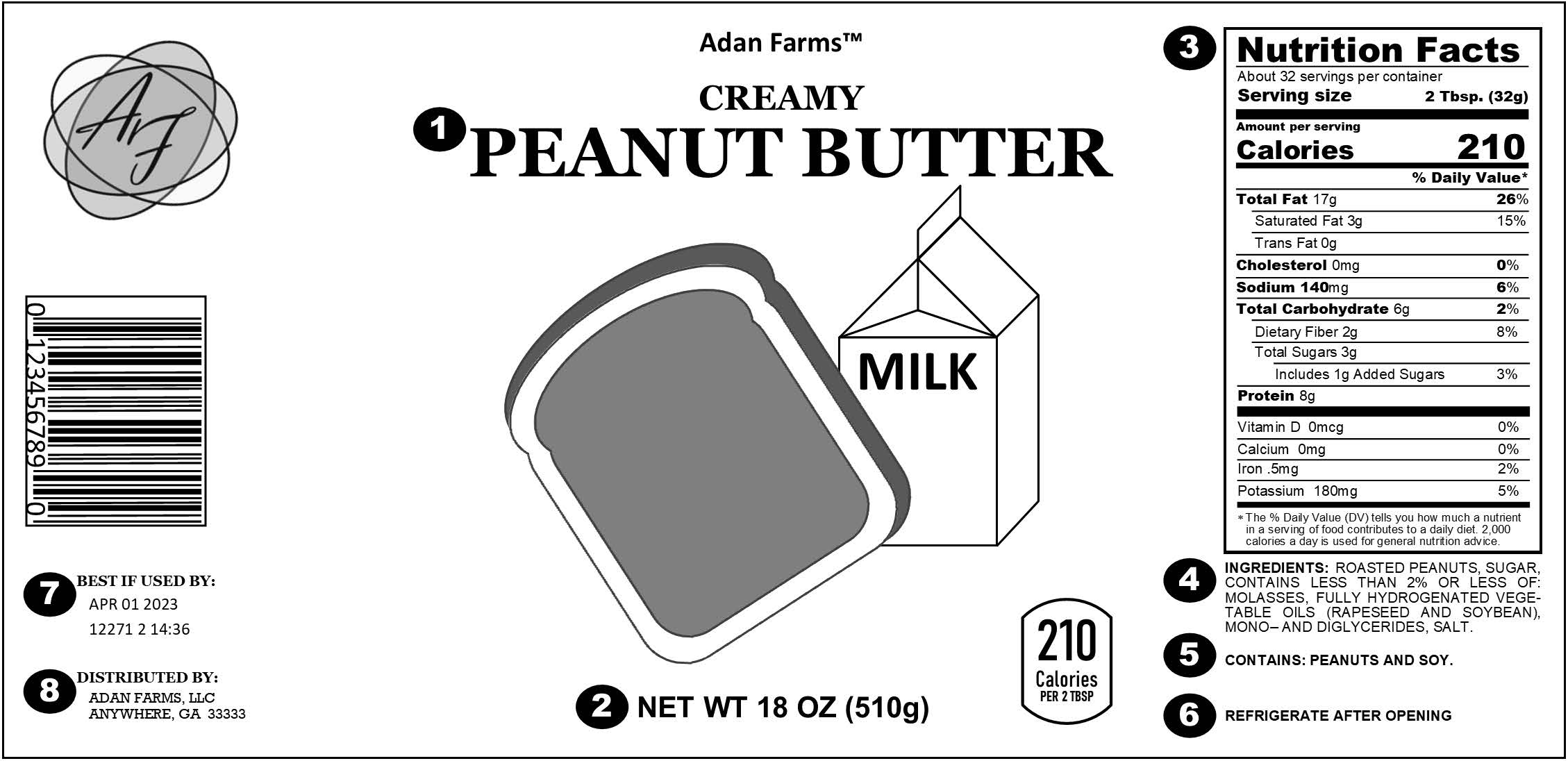
Distributor Labeling Requirements . Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. Distributor requirements (that also affect the manufacturers) october 22, 2021. Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. The information on this page is current as of mar 22, 2024. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. Some labeling requirements apply to all. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels.
from agr.ga.gov
Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). The information on this page is current as of mar 22, 2024. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. Some labeling requirements apply to all. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Distributor requirements (that also affect the manufacturers) october 22, 2021. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or.
Food Labeling Requirements Department of Agriculture
Distributor Labeling Requirements Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. The information on this page is current as of mar 22, 2024. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. Distributor requirements (that also affect the manufacturers) october 22, 2021. Some labeling requirements apply to all. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels.
From www.scribd.com
General Device Labeling Requirements PDF PDF Government Business Distributor Labeling Requirements If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. The information on this page is. Distributor Labeling Requirements.
From labelbasic.com
CBD Labeling Regulations For Packaging Products Distributor Labeling Requirements Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person. Distributor Labeling Requirements.
From www.artworkflowhq.com
Your Goto Handbook of FDA’s Labeling Requirements For Dietary Supplements Distributor Labeling Requirements Distributor requirements (that also affect the manufacturers) october 22, 2021. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Distributors should verify that the devices have been ce marked, that an eu declaration of. Distributor Labeling Requirements.
From dokumen.tips
(PDF) PV Labeling Requirements NEC 2014 RES Supply · PV Labeling Distributor Labeling Requirements Some labeling requirements apply to all. Distributor requirements (that also affect the manufacturers) october 22, 2021. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. The. Distributor Labeling Requirements.
From mahanakornpartners.com
Enhancing Consumer Safety New Labeling Requirements for LaserEnabled Distributor Labeling Requirements Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. Distributor requirements (that also affect the manufacturers) october 22, 2021. Distributors should verify that the devices. Distributor Labeling Requirements.
From ar.inspiredpencil.com
Fda Labeling Requirements For Food Distributor Labeling Requirements Some labeling requirements apply to all. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. The information on this page is current as of mar 22, 2024. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a. Distributor Labeling Requirements.
From mavig.com
New Product Labeling due to MDR MAVIG Distributor Labeling Requirements Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. The information on this page is current as of mar 22, 2024. Distributor requirements (that also affect the manufacturers) october 22, 2021. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a. Distributor Labeling Requirements.
From www.smithcorona.com
What Are the GHS Labeling Requirements? Distributor Labeling Requirements If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity. Distributor Labeling Requirements.
From www.slideserve.com
PPT ECON 337 Agricultural Marketing PowerPoint Presentation, free Distributor Labeling Requirements Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. The information on this page is current as of mar 22, 2024. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Some labeling requirements apply to. Distributor Labeling Requirements.
From www.vrogue.co
Eu Mdr Medical Device Labeling Requirements A Complet vrogue.co Distributor Labeling Requirements Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. The information on this page is current as of mar 22, 2024. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Fda regulations require that distributor labeling be identical to the labeling approved in the. Distributor Labeling Requirements.
From www.pdffiller.com
Fillable Online Guidance for Industry. Distributor Labeling for New Distributor Labeling Requirements Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. Distributor requirements (that also affect the manufacturers) october 22, 2021. Some labeling requirements apply to all. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. If. Distributor Labeling Requirements.
From www.researchandmarkets.com
US FDA Labeling Requirements for Medical Devices Distributor Labeling Requirements Some labeling requirements apply to all. Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Distributor requirements (that also affect the manufacturers) october 22, 2021. If. Distributor Labeling Requirements.
From www.slideserve.com
PPT Right to Know and Blood borne Pathogens PowerPoint Presentation Distributor Labeling Requirements Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Distributor requirements (that also affect the manufacturers) october 22, 2021. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the. Distributor Labeling Requirements.
From www.fda.gov.ph
Draft for Comments Guidelines on Labeling Requirements of Drug Distributor Labeling Requirements If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Distributor requirements (that also affect the manufacturers) october 22, 2021. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. Some labeling requirements apply to all. The information on this page is current as of mar 22, 2024. Products. Distributor Labeling Requirements.
From paragondsi.com
UDI Unique Device Identification for Single and Multiple Uses Distributor Labeling Requirements Distributor requirements (that also affect the manufacturers) october 22, 2021. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the. Distributor Labeling Requirements.
From vivafda.com
FDA Medical Device Labeling Requirements Viva FDA U.S. FDA Distributor Labeling Requirements Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Distributor requirements (that also affect the manufacturers) october 22, 2021. The information on this page is current as of mar 22, 2024. Distributors should verify that the devices have. Distributor Labeling Requirements.
From slator.com
Product Labeling Requirements What You Need To Know Distributor Labeling Requirements If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. Distributor requirements (that also affect the manufacturers) october 22, 2021. Some labeling requirements apply to all. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). The information on this page is current as of. Distributor Labeling Requirements.
From www.fda.gov.ph
Draft for Comments Guidelines on Labeling Requirements of Drug Distributor Labeling Requirements Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Distributor requirements (that also affect the manufacturers) october 22, 2021. If, for example, a packer, distributor, or seller intends an article for different uses than those. Distributor Labeling Requirements.
From blog.catalpha.com
Understanding FDA Labeling Requirements For Food Products Distributor Labeling Requirements Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Distributor requirements (that also affect the manufacturers) october 22, 2021. Some labeling requirements apply to all. Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. If,. Distributor Labeling Requirements.
From www.bizerba.com
Produce labeling contract Distributor Labeling Requirements Distributor requirements (that also affect the manufacturers) october 22, 2021. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). The information on this page is current as of mar 22, 2024. Some labeling requirements apply. Distributor Labeling Requirements.
From agr.ga.gov
Food Labeling Requirements Department of Agriculture Distributor Labeling Requirements Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. Products sold in the united states must generally be labeled or marked according to the requirements in. Distributor Labeling Requirements.
From slideplayer.com
Advanced Hazard Communication ppt download Distributor Labeling Requirements If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. The information on this page. Distributor Labeling Requirements.
From www.pdffiller.com
Fillable Online Guidance for Industry 231. Distributor Labeling for Distributor Labeling Requirements Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Some labeling requirements apply to all. The information on this page is current as of mar 22, 2024. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). If, for example, a packer, distributor, or seller. Distributor Labeling Requirements.
From mrlabel.com
Labeling Requirements for Retail Products in Chicago Distributor Labeling Requirements Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Distributor requirements (that also affect. Distributor Labeling Requirements.
From www.impactfirst.co
What is a distributor? Definition & 5 factors to consider Distributor Labeling Requirements Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Distributor requirements (that also affect the manufacturers) october 22, 2021. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. The information on this page is current. Distributor Labeling Requirements.
From harrisbricken.com
HempDerived Products The Law Does Not Stop at Labeling Requirements Distributor Labeling Requirements Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. The information on this page is current as of mar 22, 2024. Some labeling requirements apply to. Distributor Labeling Requirements.
From clin-r.com
Labels for Medical Devices Clin R Distributor Labeling Requirements Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. The information on this page is current as of mar 22, 2024. If multiple symbols (i.e., authorized. Distributor Labeling Requirements.
From www.canadianpackaging.com
Top Five Canadian Labelling MustHaves Canadian PackagingCanadian Distributor Labeling Requirements Some labeling requirements apply to all. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). The information on this page is current as of mar 22, 2024. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. Products sold in the united states must. Distributor Labeling Requirements.
From www.schlafenderhase.com
A Guide to Medical Device Labeling Requirements Schlafender Hase Distributor Labeling Requirements Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. The information on this page is current as of mar 22, 2024. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Products sold in the united states must generally be labeled or marked according to the requirements in. Distributor Labeling Requirements.
From www.slideserve.com
PPT Chapter 3 PowerPoint Presentation, free download ID1950891 Distributor Labeling Requirements Distributor requirements (that also affect the manufacturers) october 22, 2021. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Some labeling requirements apply to all. Fda regulations require that distributor labeling be identical to the. Distributor Labeling Requirements.
From packaginghub.com
FDA Packaging and Labeling Requirements Guide Packaging Hub Distributor Labeling Requirements Distributor requirements (that also affect the manufacturers) october 22, 2021. Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. Some labeling requirements apply to all. The information on this page is current as of mar 22, 2024. If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). If,. Distributor Labeling Requirements.
From www.vrogue.co
Canadian Device Labeling Requirements Ce Mark Package vrogue.co Distributor Labeling Requirements If multiple symbols (i.e., authorized representative, importer, distributor, translation, or repackaging). Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. Products sold in the united states must generally be labeled or marked according to the requirements in the applicable regulations. Distributors should verify that the devices have been ce. Distributor Labeling Requirements.
From info.ctmlabelingsystems.com
Guide to Organic Labeling Requirements Distributor Labeling Requirements Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. The information on this page is current as of mar 22, 2024. Distributor requirements (that also affect the manufacturers) october 22, 2021. Some labeling requirements apply to all. If, for example, a packer, distributor, or seller intends an article for. Distributor Labeling Requirements.
From gbu-taganskij.ru
EU MDR 2017/745 Medical Device Labeling Compliance, 48 OFF Distributor Labeling Requirements If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. Distributors should verify that the devices have been ce marked, that an eu declaration of conformity has been drawn up, and that labels. Some labeling requirements apply to all. If multiple symbols (i.e., authorized representative, importer,. Distributor Labeling Requirements.
From automatedmarking.com
Walmart® Pallet Shipping Label Requirements Distributor Labeling Requirements Distributor requirements (that also affect the manufacturers) october 22, 2021. If, for example, a packer, distributor, or seller intends an article for different uses than those intended by the person from whom he or. Fda regulations require that distributor labeling be identical to the labeling approved in the nada/anada, except for a different and. Products sold in the united states. Distributor Labeling Requirements.