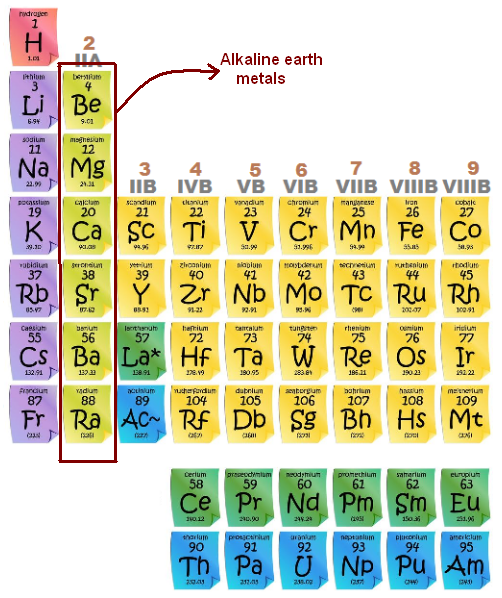
Four Properties Of Alkaline Earth Metal . The alkaline earth metals tend to. alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The name is derived from the observation that they have such high melting points (table. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). the group 2 metals have a particular name:
from www.askiitians.com
The name is derived from the observation that they have such high melting points (table. Group 2 elements are referred to as “alkaline earth” metals (tan column below). The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). the group 2 metals have a particular name: The alkaline earth metals tend to. alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state.
Alkaline Earth Metals Study Material for IIT JEE askIITians
Four Properties Of Alkaline Earth Metal The name is derived from the observation that they have such high melting points (table. The name is derived from the observation that they have such high melting points (table. Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. the group 2 metals have a particular name: The alkaline earth metals tend to. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra).
From www.sliderbase.com
Element Classes Presentation Chemistry Four Properties Of Alkaline Earth Metal the group 2 metals have a particular name: Group 2 elements are referred to as “alkaline earth” metals (tan column below). The name is derived from the observation that they have such high melting points (table. The alkaline earth metals tend to. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). . Four Properties Of Alkaline Earth Metal.
From mungfali.com
Periodic Table Of Elements Alkali Metals Four Properties Of Alkaline Earth Metal The name is derived from the observation that they have such high melting points (table. The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. the group 2 metals have a particular name: The. Four Properties Of Alkaline Earth Metal.
From www.slideserve.com
PPT Alkali Metals and Alkaline Earth Metals PowerPoint Presentation Four Properties Of Alkaline Earth Metal Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to. The name is derived from the observation that they have such high melting points (table. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium. Four Properties Of Alkaline Earth Metal.
From slidedocnow.blogspot.com
Properties Of Alkaline Earth Metals slidedocnow Four Properties Of Alkaline Earth Metal The alkaline earth metals tend to. alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. The name is derived from the observation that they have such high melting points (table. Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals are good reducing. Four Properties Of Alkaline Earth Metal.
From www.slideserve.com
PPT Alkaline Earth Metals PowerPoint Presentation ID2046855 Four Properties Of Alkaline Earth Metal The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). The alkaline earth metals tend to. the group 2 metals have a particular name: The name is derived from the observation that they have such high melting points (table. alkaline earth metals dissolve in liquid ammonia to give solutions that contain two. Four Properties Of Alkaline Earth Metal.
From www.youtube.com
Physical properties of alkaline earth metals SBlock elements bsc Four Properties Of Alkaline Earth Metal alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The name is derived from the observation that they have such high melting points (table. The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals dissolve in liquid ammonia to give. Four Properties Of Alkaline Earth Metal.
From byjus.com
Alkali Metals Properties, Electronic Configuration, Periodic Trends Four Properties Of Alkaline Earth Metal Group 2 elements are referred to as “alkaline earth” metals (tan column below). The alkaline earth metals tend to. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). The name is derived from the observation that they have such high melting points (table. the group 2 metals have a particular name: . Four Properties Of Alkaline Earth Metal.
From www.youtube.com
Identifying Alkaline earth metals by properties YouTube Four Properties Of Alkaline Earth Metal the group 2 metals have a particular name: alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Group 2 elements are referred to as “alkaline earth” metals (tan column below). The alkaline earth metals tend to. The name is derived from the observation that they have such high melting points (table. . Four Properties Of Alkaline Earth Metal.
From studylib.net
1819Alkaline Earth Metals Four Properties Of Alkaline Earth Metal The alkaline earth metals tend to. the group 2 metals have a particular name: The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. Group 2 elements are referred to as “alkaline earth” metals. Four Properties Of Alkaline Earth Metal.
From exobgridh.blob.core.windows.net
What Are The Common Physical Properties Of Alkali Metals at Israel Four Properties Of Alkaline Earth Metal Group 2 elements are referred to as “alkaline earth” metals (tan column below). the group 2 metals have a particular name: The name is derived from the observation that they have such high melting points (table. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). The alkaline earth metals tend to. . Four Properties Of Alkaline Earth Metal.
From www.expii.com
Alkaline Earth Metals — Overview & Properties Expii Four Properties Of Alkaline Earth Metal alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. the group 2 metals have a particular name: The alkaline earth metals tend to. The name is derived from the observation that they have such high melting points (table. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and. Four Properties Of Alkaline Earth Metal.
From dxozzbgbz.blob.core.windows.net
Chemical Properties Of Alkali Metals Ppt at Crystal Gilbert blog Four Properties Of Alkaline Earth Metal The name is derived from the observation that they have such high melting points (table. the group 2 metals have a particular name: The alkaline earth metals tend to. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and. Four Properties Of Alkaline Earth Metal.
From knordslearning.com
Alkaline Earth Metals Periodic Table (With Images) Four Properties Of Alkaline Earth Metal The name is derived from the observation that they have such high melting points (table. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). Group 2 elements are referred to as “alkaline earth” metals (tan column below). the group 2 metals have a particular name: alkaline earth metals dissolve in liquid. Four Properties Of Alkaline Earth Metal.
From pediaa.com
Difference Between Alkali Metals and Alkaline Earth Metals Definition Four Properties Of Alkaline Earth Metal alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The name is derived from the observation that they have such high melting points (table. The alkaline earth metals tend to. alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. Group 2 elements are. Four Properties Of Alkaline Earth Metal.
From elchoroukhost.net
Properties Of Alkali Metals On The Periodic Table Elcho Table Four Properties Of Alkaline Earth Metal the group 2 metals have a particular name: alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals. Four Properties Of Alkaline Earth Metal.
From www.animalia-life.club
Periodic Table Of Elements Alkaline Earth Metals Four Properties Of Alkaline Earth Metal The alkaline earth metals tend to. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). The name is derived from the observation that they have such high melting points (table. alkaline earth metals dissolve in liquid. Four Properties Of Alkaline Earth Metal.
From www.youtube.com
18. Properties Of Alkaline Earth Metals YouTube Four Properties Of Alkaline Earth Metal the group 2 metals have a particular name: The alkaline earth metals tend to. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). alkaline earth metals dissolve in liquid ammonia to give solutions that contain. Four Properties Of Alkaline Earth Metal.
From byjus.com
Alkali Metals Chemical and Physical Properties of Alkali Metals Four Properties Of Alkaline Earth Metal The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. The name is derived from the observation that they have such high. Four Properties Of Alkaline Earth Metal.
From utedzz.blogspot.com
Alkaline Earth Metals Periodic Table Definition Periodic Table Timeline Four Properties Of Alkaline Earth Metal The alkaline earth metals tend to. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). the group 2 metals have a particular name: alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The name is derived from the observation that they have such high melting. Four Properties Of Alkaline Earth Metal.
From www.slideserve.com
PPT Elements and their Properties PowerPoint Presentation, free Four Properties Of Alkaline Earth Metal Group 2 elements are referred to as “alkaline earth” metals (tan column below). The alkaline earth metals tend to. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). The name is derived from the observation that they have such high melting points (table. alkaline earth metals are good reducing agents that tend. Four Properties Of Alkaline Earth Metal.
From www.youtube.com
Properties of the Alkaline Earth Metals YouTube Four Properties Of Alkaline Earth Metal The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). The name is derived from the observation that they have such high melting points (table. The alkaline earth metals tend to. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. Group 2 elements are referred to as. Four Properties Of Alkaline Earth Metal.
From www.slideserve.com
PPT Atomic Theory PowerPoint Presentation, free download ID5435746 Four Properties Of Alkaline Earth Metal Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. the group 2 metals have a particular name: The alkaline earth metals tend to. alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons. Four Properties Of Alkaline Earth Metal.
From thechemistrynotes.com
Comparison of properties of Alkali and Alkaline Earth Metals Four Properties Of Alkaline Earth Metal The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). Group 2 elements are referred to as “alkaline earth” metals (tan column below). The name is derived from the observation that they have such high melting points (table. alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons. Four Properties Of Alkaline Earth Metal.
From www.britannica.com
Alkalineearth metal chemical element Britannica Four Properties Of Alkaline Earth Metal The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). The name is derived from the observation that they have such high melting points (table. the group 2 metals have a particular name: The alkaline earth metals tend to. alkaline earth metals are good reducing agents that tend to form the +2. Four Properties Of Alkaline Earth Metal.
From www.tes.com
Alkali and Alkaline Earth Metals Venn Diagram Teaching Resources Four Properties Of Alkaline Earth Metal the group 2 metals have a particular name: alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). Group 2 elements are referred to as “alkaline earth” metals (tan column below).. Four Properties Of Alkaline Earth Metal.
From byjus.com
Alkaline Earth Metals Occurrence and Extraction,Physical Properties Four Properties Of Alkaline Earth Metal The name is derived from the observation that they have such high melting points (table. The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column below). The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). the group 2 metals have a particular name: . Four Properties Of Alkaline Earth Metal.
From utedzz.blogspot.com
Periodic Table Showing Alkali Metals Alkaline Earth Metals Periodic Four Properties Of Alkaline Earth Metal alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. Group 2 elements are referred to as “alkaline earth” metals (tan column below). the group 2 metals have a particular name: alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth. Four Properties Of Alkaline Earth Metal.
From www.thoughtco.com
What Are the Properties of the Alkaline Earth Metals? Four Properties Of Alkaline Earth Metal The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. the group 2 metals have a particular name:. Four Properties Of Alkaline Earth Metal.
From www.slideserve.com
PPT Alkali Metals and Alkaline Earth Metals PowerPoint Presentation Four Properties Of Alkaline Earth Metal alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. the group 2 metals have a particular name: The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). The name is derived from the observation that they have such high melting points (table. . Four Properties Of Alkaline Earth Metal.
From www.slideserve.com
PPT Families on the Periodic Table PowerPoint Presentation, free Four Properties Of Alkaline Earth Metal Group 2 elements are referred to as “alkaline earth” metals (tan column below). The name is derived from the observation that they have such high melting points (table. the group 2 metals have a particular name: alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. alkaline earth metals dissolve in liquid. Four Properties Of Alkaline Earth Metal.
From dxozzbgbz.blob.core.windows.net
Chemical Properties Of Alkali Metals Ppt at Crystal Gilbert blog Four Properties Of Alkaline Earth Metal The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). Group 2 elements are referred to as “alkaline earth” metals (tan column below). alkaline earth metals dissolve in liquid ammonia to give solutions that contain two solvated electrons per metal atom. The alkaline earth metals tend to. The name is derived from the. Four Properties Of Alkaline Earth Metal.
From www.youtube.com
ALKALINE EARTH METAL 1 physical and chemical properties alkaline Four Properties Of Alkaline Earth Metal The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column below). The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). The name is derived from the observation that they have such high melting points (table. alkaline earth metals dissolve in liquid ammonia to give. Four Properties Of Alkaline Earth Metal.
From newtondesk.com
Alkaline Earth Metals On The Periodic Table Chemistry Elements Four Properties Of Alkaline Earth Metal The name is derived from the observation that they have such high melting points (table. alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column below). the group 2 metals have a particular name: The. Four Properties Of Alkaline Earth Metal.
From byjus.com
Alkaline Earth Metals General Characteristics of Oxides, Hallides Four Properties Of Alkaline Earth Metal alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to. The name is derived from the observation that they have such high melting points (table. The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). the group 2 metals have a. Four Properties Of Alkaline Earth Metal.
From www.askiitians.com
Alkaline Earth Metals Study Material for IIT JEE askIITians Four Properties Of Alkaline Earth Metal The elements are beryllium (be), magnesium (mg), calcium (ca), strontium (sr), barium (ba), and radium (ra). the group 2 metals have a particular name: alkaline earth metals are good reducing agents that tend to form the +2 oxidation state. The alkaline earth metals tend to. Group 2 elements are referred to as “alkaline earth” metals (tan column below).. Four Properties Of Alkaline Earth Metal.