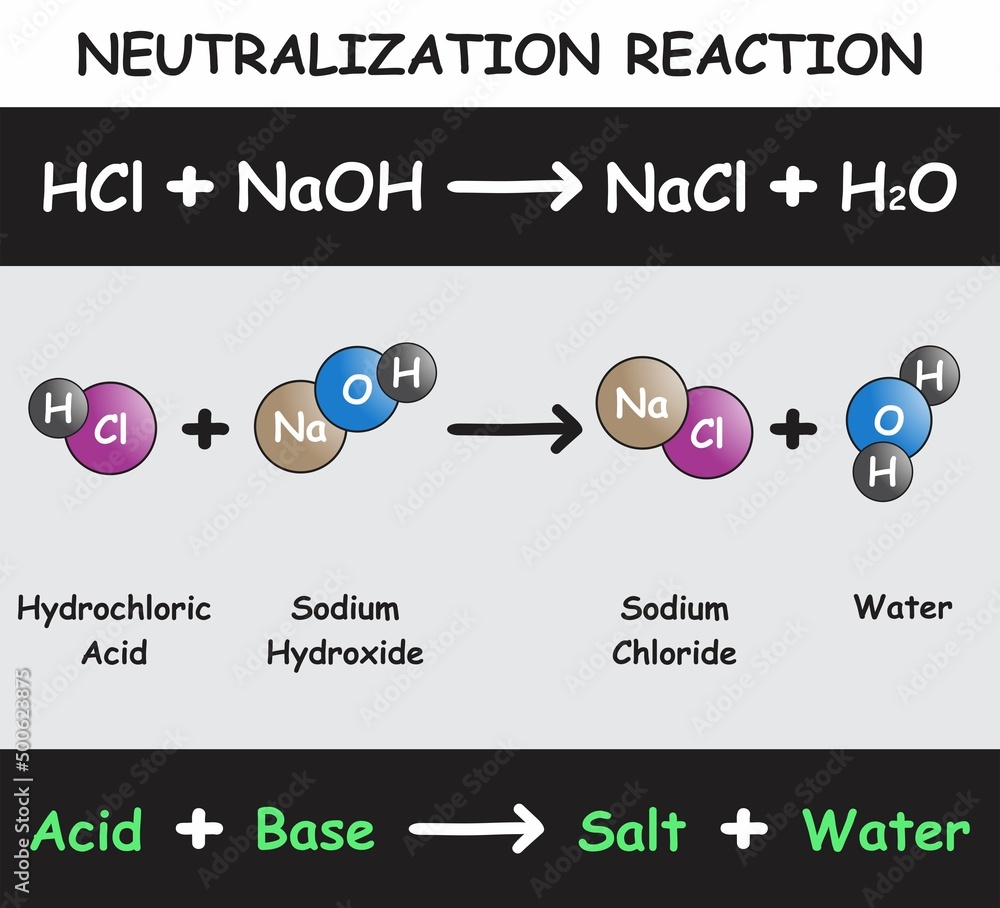
Hydrochloric Acid Produce Sodium Chloride . When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. A compound formed by neutralisation of an acid by. In this reaction, naoh reacts. They then concentrate the solution and allow it to crystallise. When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. The balanced chemical equation for this reaction is: For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. Acidity is caused by a high concentration of hydrogen ions. Ammonia forms ammonium salts when it reacts with acids. Naoh + hcl → nacl + h 2 o. A base which is soluble in water. Hydrochloric acid is an ionic. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution.
from stock.adobe.com
Acidity is caused by a high concentration of hydrogen ions. Naoh + hcl → nacl + h 2 o. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. Hydrochloric acid is an ionic. For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. A compound formed by neutralisation of an acid by. Ammonia forms ammonium salts when it reacts with acids. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides;
Neutralization Reaction Infographic Diagram with example of
Hydrochloric Acid Produce Sodium Chloride When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. A base which is soluble in water. They then concentrate the solution and allow it to crystallise. When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. The balanced chemical equation for this reaction is: For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): In this reaction, naoh reacts. A compound formed by neutralisation of an acid by. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; Acidity is caused by a high concentration of hydrogen ions. For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. Ammonia forms ammonium salts when it reacts with acids. Hydrochloric acid is an ionic. Naoh + hcl → nacl + h 2 o. When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs.
From www.numerade.com
SOLVED Production of sodium chloride Change this word equation into Hydrochloric Acid Produce Sodium Chloride A compound formed by neutralisation of an acid by. For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. Ammonia forms ammonium salts when it reacts with acids. The balanced chemical equation for this reaction. Hydrochloric Acid Produce Sodium Chloride.
From www.chegg.com
Solved Aqueous hydrochloric acid HCl will react with solid Hydrochloric Acid Produce Sodium Chloride A compound formed by neutralisation of an acid by. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. The balanced chemical equation for this reaction. Hydrochloric Acid Produce Sodium Chloride.
From byjus.com
Write the neutralization reaction between Hydrochloric acid HCI and Hydrochloric Acid Produce Sodium Chloride Hydrochloric acid is an ionic. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; Naoh + hcl → nacl + h 2 o. For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. Ammonia forms ammonium salts when it reacts with acids. A base which is. Hydrochloric Acid Produce Sodium Chloride.
From www.numerade.com
SOLVED write a balance chemical equation with state symbol for the Hydrochloric Acid Produce Sodium Chloride The balanced chemical equation for this reaction is: In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. Naoh + hcl → nacl + h 2 o. Acidity is caused by a high concentration of hydrogen ions. A base which is soluble in water. For example, hydrochloric acid and sodium hydroxide. Hydrochloric Acid Produce Sodium Chloride.
From www.shalom-education.com
Acids and Alkalis GCSE Chemistry Revision Hydrochloric Acid Produce Sodium Chloride A base which is soluble in water. Ammonia forms ammonium salts when it reacts with acids. Acidity is caused by a high concentration of hydrogen ions. When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. The balanced chemical equation for this reaction is: They then. Hydrochloric Acid Produce Sodium Chloride.
From www.numerade.com
SOLVED When an aqueous solution of hydrochloric acid is mixed with Hydrochloric Acid Produce Sodium Chloride When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): A compound formed by. Hydrochloric Acid Produce Sodium Chloride.
From www.youtube.com
Sodium metal reacting with concentrated hydrochloric acid YouTube Hydrochloric Acid Produce Sodium Chloride A base which is soluble in water. When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. The balanced chemical equation for this reaction is: Naoh + hcl → nacl + h 2 o. Ammonia forms ammonium salts when it reacts with acids. For instance,. Hydrochloric Acid Produce Sodium Chloride.
From basdemax.netlify.app
28++ Hydrochloric Acid And Sodium Hydroxide Balanced Equation Basdemax Hydrochloric Acid Produce Sodium Chloride Acidity is caused by a high concentration of hydrogen ions. Ammonia forms ammonium salts when it reacts with acids. In this reaction, naoh reacts. They then concentrate the solution and allow it to crystallise. A compound formed by neutralisation of an acid by. For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium. Hydrochloric Acid Produce Sodium Chloride.
From insende.netlify.app
30+ Hydrochloric Acid And Sodium Hydroxide Equation Insende Hydrochloric Acid Produce Sodium Chloride Acidity is caused by a high concentration of hydrogen ions. The balanced chemical equation for this reaction is: For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): The identity of the salt produced depends on the acid used and the positive ions in the base. Hydrochloric Acid Produce Sodium Chloride.
From www.numerade.com
SOLVED Aqueous hydrochloric acid HCl will react with solid sodium Hydrochloric Acid Produce Sodium Chloride For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; The balanced chemical equation for this reaction is: Hydrochloric acid is an ionic. Acidity is caused by a high concentration of hydrogen ions. They then concentrate the. Hydrochloric Acid Produce Sodium Chloride.
From www.slideshare.net
5 3 and 5 4 Hydrochloric Acid Produce Sodium Chloride Naoh + hcl → nacl + h 2 o. When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. They then concentrate the solution and allow it to crystallise. A compound formed by neutralisation of an acid by. For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. A base which is soluble in water. The. Hydrochloric Acid Produce Sodium Chloride.
From www.slideserve.com
PPT Chapter 11 Chemical Reactions PowerPoint Presentation, free Hydrochloric Acid Produce Sodium Chloride The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; Acidity is caused by a high concentration of hydrogen ions. A base which is soluble in water. A compound formed by neutralisation of an acid by. Naoh + hcl → nacl + h 2 o. Ammonia forms ammonium. Hydrochloric Acid Produce Sodium Chloride.
From www.shutterstock.com
520 imágenes de Molecule of sodium chloride Imágenes, fotos y Hydrochloric Acid Produce Sodium Chloride When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. They then concentrate the solution and allow it to crystallise. The identity of the salt produced depends on the acid used and. Hydrochloric Acid Produce Sodium Chloride.
From www.numerade.com
SOLVED Aqueous hydrochloric acid (HCI) will react with solid sodium Hydrochloric Acid Produce Sodium Chloride Acidity is caused by a high concentration of hydrogen ions. For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. Naoh + hcl → nacl + h 2 o. Ammonia forms ammonium salts when it reacts with acids. A base which is soluble in water. When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. A. Hydrochloric Acid Produce Sodium Chloride.
From www.numerade.com
SOLVED The production of sodium chloride was achieved by the addition Hydrochloric Acid Produce Sodium Chloride Naoh + hcl → nacl + h 2 o. Hydrochloric acid is an ionic. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): The balanced chemical. Hydrochloric Acid Produce Sodium Chloride.
From wisc.pb.unizin.org
Acids, Bases, Neutralization, and GasForming Reactions (M3Q34) UW Hydrochloric Acid Produce Sodium Chloride When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. In this reaction, naoh reacts. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; When. Hydrochloric Acid Produce Sodium Chloride.
From www.chegg.com
Solved Aqueous hydrochloric acid (HCl) reacts with solid Hydrochloric Acid Produce Sodium Chloride When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. In this reaction, naoh reacts. The balanced chemical equation for this reaction is: Naoh + hcl → nacl + h 2 o. Hydrochloric acid is an ionic. For instance, ammonia reacts with hydrochloric acid to. Hydrochloric Acid Produce Sodium Chloride.
From www.numerade.com
SOLVEDAqueous hydrochloric acid reacts with solid sodium hydroxide to Hydrochloric Acid Produce Sodium Chloride They then concentrate the solution and allow it to crystallise. For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. In this. Hydrochloric Acid Produce Sodium Chloride.
From www.toppr.com
(1) Solutions of barium chloride and sodium sulphate in water react Hydrochloric Acid Produce Sodium Chloride The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; A base which is soluble in water. The balanced chemical equation for this reaction is: Acidity is caused by a high concentration of hydrogen ions. Naoh + hcl → nacl + h 2 o. For example, hydrochloric acid. Hydrochloric Acid Produce Sodium Chloride.
From www.dreamstime.com
Diagram of Preparation of Hydrogen Chloride Gas Stock Illustration Hydrochloric Acid Produce Sodium Chloride They then concentrate the solution and allow it to crystallise. The balanced chemical equation for this reaction is: For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. A base which is soluble in water.. Hydrochloric Acid Produce Sodium Chloride.
From brainly.com
Below is the word equation for the reaction between sodium hydroxide Hydrochloric Acid Produce Sodium Chloride Naoh + hcl → nacl + h 2 o. When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; Acidity is caused by. Hydrochloric Acid Produce Sodium Chloride.
From study.com
Hydrogen Chloride vs. Hydrochloric Acid Formula, Properties Hydrochloric Acid Produce Sodium Chloride Acidity is caused by a high concentration of hydrogen ions. For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. A base which is soluble in water. In this reaction, naoh reacts. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; Naoh + hcl → nacl. Hydrochloric Acid Produce Sodium Chloride.
From www.youtube.com
Na2CO3 + HCl Sodium Carbonate + Hydrochloric Acid YouTube Hydrochloric Acid Produce Sodium Chloride For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. Acidity is caused by a high concentration of hydrogen ions. In this reaction, naoh reacts. Hydrochloric acid is an ionic. The balanced chemical equation. Hydrochloric Acid Produce Sodium Chloride.
From stock.adobe.com
Neutralization Reaction Infographic Diagram with example of Hydrochloric Acid Produce Sodium Chloride For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. A base which is soluble in water. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. In this reaction, naoh reacts. Acidity is caused by a high concentration of hydrogen ions. For example, hydrochloric acid and sodium hydroxide react. Hydrochloric Acid Produce Sodium Chloride.
From www.youtube.com
How to Balance NaOH + HCl = NaCl + H2O (Sodium Hydroxide Plus Hydrochloric Acid Produce Sodium Chloride When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; They then concentrate the solution and allow it to crystallise. For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. For example, hydrochloric acid. Hydrochloric Acid Produce Sodium Chloride.
From socratic.org
What is the pH of a .001 M solution of HCl? Socratic Hydrochloric Acid Produce Sodium Chloride A compound formed by neutralisation of an acid by. For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): Ammonia forms ammonium salts when it reacts with acids. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in. Hydrochloric Acid Produce Sodium Chloride.
From www.alamy.com
Hydrochloric Acid High Resolution Stock Photography and Images Alamy Hydrochloric Acid Produce Sodium Chloride A base which is soluble in water. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; A compound formed by neutralisation of an acid by. They then concentrate the solution and allow it to crystallise. Hydrochloric acid is an ionic. In this experiment students neutralise sodium hydroxide. Hydrochloric Acid Produce Sodium Chloride.
From www.youtube.com
Making Sodium Hydroxide and Hydrochloric Acid from salt water. YouTube Hydrochloric Acid Produce Sodium Chloride When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. A compound formed by neutralisation of an acid by. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. Naoh + hcl → nacl + h 2 o. For instance, ammonia reacts with hydrochloric acid to make ammonium. Hydrochloric Acid Produce Sodium Chloride.
From www.youtube.com
How to Write the Net Ionic Equation for Na2SO3 + HCl = NaCl + SO2 + H2O Hydrochloric Acid Produce Sodium Chloride For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. In this reaction, naoh reacts. In this experiment students neutralise sodium. Hydrochloric Acid Produce Sodium Chloride.
From www.chegg.com
Solved Aqueous hydrochloric acid (HCI) will react with solid Hydrochloric Acid Produce Sodium Chloride When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. A base which is soluble in water. The identity of the salt produced depends on the acid used and the positive ions in the base hydrochloric acid produces chlorides; In this reaction, naoh reacts. The balanced chemical equation for this reaction is: When equal amounts of a. Hydrochloric Acid Produce Sodium Chloride.
From stock.adobe.com
Laboratory preparation of hydrogen chloride. vector illustration Hydrochloric Acid Produce Sodium Chloride When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. Naoh + hcl → nacl + h 2 o. Acidity is caused by a high concentration of hydrogen ions. Ammonia forms ammonium. Hydrochloric Acid Produce Sodium Chloride.
From exyptikle.blob.core.windows.net
Chemical Reaction Between Hydrochloric Acid And Sodium Hypochlorite at Hydrochloric Acid Produce Sodium Chloride The balanced chemical equation for this reaction is: Naoh + hcl → nacl + h 2 o. Acidity is caused by a high concentration of hydrogen ions. When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. Ammonia forms ammonium salts when it reacts with acids. In this reaction, naoh reacts. A compound formed by neutralisation of. Hydrochloric Acid Produce Sodium Chloride.
From mammothmemory.net
Sodium reaction with hydrochloric acid is violent and quick Hydrochloric Acid Produce Sodium Chloride The balanced chemical equation for this reaction is: In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): They then concentrate the solution and allow it to. Hydrochloric Acid Produce Sodium Chloride.
From www.numerade.com
SOLVED When hydrochloric acid reacts with sodium sulfide, hydrogen Hydrochloric Acid Produce Sodium Chloride When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a. When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. For instance, ammonia reacts with hydrochloric acid to make ammonium chloride. Acidity is caused by a high concentration of hydrogen ions. Hydrochloric. Hydrochloric Acid Produce Sodium Chloride.
From fphoto.photoshelter.com
neutralization hydrochloric acid sodium hydroxide chemistry Hydrochloric Acid Produce Sodium Chloride When sodium hydroxide (naoh) reacts with hydrochloric acid (hcl), a chemical reaction occurs. For example, hydrochloric acid and sodium hydroxide react in a neutralisation reaction to produce the salt sodium chloride (nacl) and water (h 2 o): Acidity is caused by a high concentration of hydrogen ions. In this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the. Hydrochloric Acid Produce Sodium Chloride.