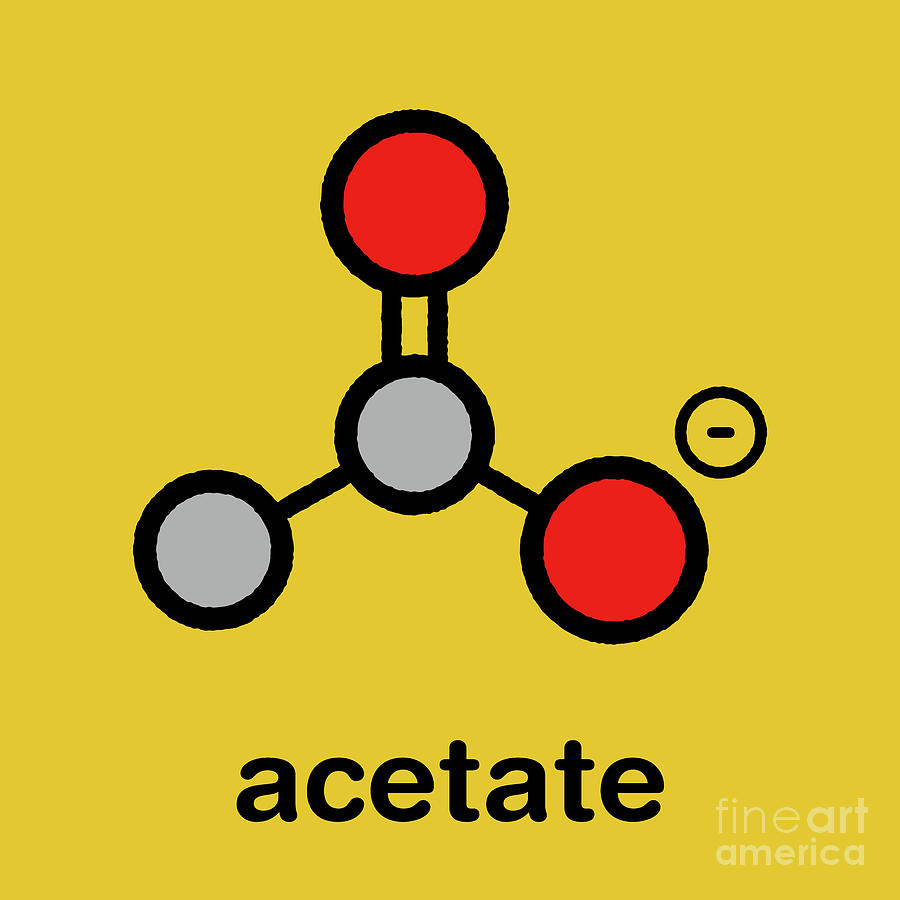
What Element Is Acetate . It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. It is formed when acetic acid is deprotonated. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Learn how acetate is formed, its properties, and its role in esters and cellulose. The rules we use depends on the type of compound we are. An acetate, or ethanoate, is a salt or ester of acetic acid. Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. Acetate is the ion of acetic acid, which is a carboxylic acid. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). In this section we will look at nomenclature of simple chemical compounds. How to quickly check pipettes? Acetate is a salt formed by acetic acid with alkaline or other bases.
from pixels.com
In this section we will look at nomenclature of simple chemical compounds. The rules we use depends on the type of compound we are. It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. Acetate is a salt formed by acetic acid with alkaline or other bases. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. An acetate, or ethanoate, is a salt or ester of acetic acid. Learn how acetate is formed, its properties, and its role in esters and cellulose. How to quickly check pipettes?
Acetate Anion Chemical Structure Photograph by Molekuul/science Photo
What Element Is Acetate The rules we use depends on the type of compound we are. Acetate is a salt formed by acetic acid with alkaline or other bases. Acetate is the ion of acetic acid, which is a carboxylic acid. How to quickly check pipettes? It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. In this section we will look at nomenclature of simple chemical compounds. The rules we use depends on the type of compound we are. An acetate, or ethanoate, is a salt or ester of acetic acid. It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. It is formed when acetic acid is deprotonated. Learn how acetate is formed, its properties, and its role in esters and cellulose. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. The acetate anion, [ch 3 coo] −, is a carboxylate and is the.
From en.wikipedia.org
Sodium acetate Wikipedia What Element Is Acetate Acetate is the ion of acetic acid, which is a carboxylic acid. Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. Learn how acetate is formed, its properties, and its role in esters. What Element Is Acetate.
From www.alamy.com
Acetate chemical structure on pink background Stock Vector Image & Art What Element Is Acetate Acetate is the ion of acetic acid, which is a carboxylic acid. It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Learn how. What Element Is Acetate.
From www.dreamstime.com
CopperII Acetate, Verdigris, Chemical Formula and Structure Stock What Element Is Acetate The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Acetate is a salt formed by acetic acid with alkaline or other bases. The rules we use depends on the type of compound we are. In this section we will look at nomenclature of simple chemical compounds. An acetate, or ethanoate, is a salt or ester of. What Element Is Acetate.
From www.thoughtco.com
Functional Groups in Organic Chemistry What Element Is Acetate It is formed when acetic acid is deprotonated. An acetate, or ethanoate, is a salt or ester of acetic acid. Acetate is a salt formed by acetic acid with alkaline or other bases. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. How to quickly check pipettes? Acetate, also. What Element Is Acetate.
From www.slideserve.com
PPT Acetate! PowerPoint Presentation, free download ID5672409 What Element Is Acetate Acetate is a salt formed by acetic acid with alkaline or other bases. Acetate is the ion of acetic acid, which is a carboxylic acid. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). In this section we will look. What Element Is Acetate.
From www.shutterstock.com
Structural Chemical Formula Model Ethyl Acetate Stock Illustration What Element Is Acetate It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. In this section we will look at nomenclature of simple chemical compounds. It is also. What Element Is Acetate.
From www.dreamstime.com
Ethyl Acetate, Ethyl Ethanoate, C4H8O2 Molecule. it is Acetate Ester What Element Is Acetate It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. In this section we will look at nomenclature of simple chemical compounds. An acetate, or ethanoate, is a salt or ester of acetic acid. Learn how acetate is formed, its properties, and its role in esters and cellulose. The rules we use depends on the type of. What Element Is Acetate.
From www.alamy.com
Ethyl acetate, ethyl ethanoate, C4H8O2 molecule. It is acetate ester What Element Is Acetate It is formed when acetic acid is deprotonated. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). Acetate is the ion of acetic acid, which is a carboxylic acid. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. It has a role as a human metabolite. What Element Is Acetate.
From fr.wikidoc.org
Acetic acid wikidoc What Element Is Acetate Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. It is formed when acetic acid is deprotonated. It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. How to quickly check pipettes? Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the. What Element Is Acetate.
From chemistry291.blogspot.com
What is the Chemical Formula for Acetate Ion What Element Is Acetate It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. Acetate is a salt formed by acetic acid with alkaline or other bases. It is formed when acetic acid is deprotonated. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Acetate is a monocarboxylic acid anion resulting from the. What Element Is Acetate.
From utedzz.blogspot.com
Periodic Table Of Elements List With Names Periodic Table Timeline What Element Is Acetate The acetate anion, [ch 3 coo] −, is a carboxylate and is the. In this section we will look at nomenclature of simple chemical compounds. Acetate is a salt formed by acetic acid with alkaline or other bases. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). Acetate is a monocarboxylic acid anion resulting from the removal. What Element Is Acetate.
From www.slideserve.com
PPT Acetate! PowerPoint Presentation, free download ID5672409 What Element Is Acetate Learn how acetate is formed, its properties, and its role in esters and cellulose. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. An acetate, or ethanoate, is a salt or ester of acetic acid. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. Acetate is a salt. What Element Is Acetate.
From en.wikipedia.org
Ethyl acetate Wikipedia What Element Is Acetate How to quickly check pipettes? It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. It has a role as a. What Element Is Acetate.
From www.alamy.com
Acetate Stock Photos & Acetate Stock Images Alamy What Element Is Acetate The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Learn how acetate is formed, its properties, and its role in esters and cellulose. It is formed when acetic acid is deprotonated. The rules we use depends on the type of compound we are. Acetate is a monocarboxylic acid anion resulting from the removal of a proton. What Element Is Acetate.
From www.vectorstock.com
Chemical formula of acetate anion Royalty Free Vector Image What Element Is Acetate Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. Acetate is a salt formed by acetic acid with alkaline or other bases. Learn how acetate is formed, its properties, and its role in esters and cellulose. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). The acetate. What Element Is Acetate.
From atomicemporium.com
Periodic Table What Element Is Acetate An acetate, or ethanoate, is a salt or ester of acetic acid. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). The rules we use depends on the type of compound we are. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. Acetate is also a functional group that. What Element Is Acetate.
From studiousguy.com
Acetic Acid Uses in Daily Life StudiousGuy What Element Is Acetate Learn how acetate is formed, its properties, and its role in esters and cellulose. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. The acetate anion, [ch 3 coo] −, is a. What Element Is Acetate.
From www.alamy.com
Acetic Acid, Structural chemical formula on a white background Stock What Element Is Acetate It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. It is formed when acetic acid is deprotonated. Acetate is a salt formed by acetic acid with alkaline or other bases. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. The rules we use depends on the type of compound we are. Acetate. What Element Is Acetate.
From collegedunia.com
Uses of Acetic Acid Definition, Structure, and Physical and Chemical What Element Is Acetate It is formed when acetic acid is deprotonated. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. In this section we will look at nomenclature of simple chemical compounds. An acetate, or ethanoate,. What Element Is Acetate.
From www.istockphoto.com
What Is Acetate Illustrations, RoyaltyFree Vector Graphics & Clip Art What Element Is Acetate Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. An acetate, or ethanoate, is a salt or ester of acetic acid. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. It is formed. What Element Is Acetate.
From pediaa.com
Difference Between Acetic Acid and Glacial Acetic Acid Properties What Element Is Acetate The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). It is formed when acetic acid is deprotonated. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. The rules we use depends on the type of compound. What Element Is Acetate.
From www.shutterstock.com
Chemical Structure Isoamyl Acetate C4h14o2 Stock Vector (Royalty Free What Element Is Acetate Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). An acetate, or ethanoate, is a salt or ester of acetic acid. Acetate is a salt formed by acetic acid with alkaline or other bases. It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. Acetate is the ion of acetic acid, which is a. What Element Is Acetate.
From pixels.com
Acetate Anion Chemical Structure Photograph by Molekuul/science Photo What Element Is Acetate Acetate is the ion of acetic acid, which is a carboxylic acid. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). Learn how acetate is formed, its properties, and its role in esters and cellulose. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. An acetate, or ethanoate, is a salt or ester. What Element Is Acetate.
From study.com
Acetate Definition, Formula & Structure Lesson What Element Is Acetate The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Acetate is a salt formed by acetic acid with alkaline or other bases. The rules we use depends on the type of compound we are. It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. It is formed when acetic acid is deprotonated. How. What Element Is Acetate.
From lab.honeywell.com
Sodium acetate S8750 Honeywell Research Chemicals What Element Is Acetate It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. Acetate is a salt formed by acetic acid with alkaline or other bases. Learn how acetate is formed, its properties, and its role in esters and cellulose. In this section we will look at nomenclature of simple chemical compounds. Acetate is a monocarboxylic acid anion resulting from. What Element Is Acetate.
From www.dreamstime.com
Acetic Acid, Structural Chemical Formula Stock Illustration What Element Is Acetate How to quickly check pipettes? Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. It has a role as a. What Element Is Acetate.
From www.youtube.com
How to Write the Chemical Formula for Acetate ion YouTube What Element Is Acetate Acetate is the ion of acetic acid, which is a carboxylic acid. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). It is formed when acetic acid is deprotonated. It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. An acetate, or ethanoate, is a salt or ester of acetic acid. It is also. What Element Is Acetate.
From www.alamy.com
3d render of molecular structure of Acetic acid isolated over white What Element Is Acetate Learn how acetate is formed, its properties, and its role in esters and cellulose. It is formed when acetic acid is deprotonated. Acetate is the ion of acetic acid, which is a carboxylic acid. An acetate, or ethanoate, is a salt or ester of acetic acid. Acetate is also a functional group that connects to other molecules, and a salt. What Element Is Acetate.
From www.myxxgirl.com
What Is The Molecular Formula Of Acetate Draw The Lewis Structure And What Element Is Acetate It is formed when acetic acid is deprotonated. An acetate, or ethanoate, is a salt or ester of acetic acid. Acetate is a salt formed by acetic acid with alkaline or other bases. Acetate is the ion of acetic acid, which is a carboxylic acid. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the. What Element Is Acetate.
From pixels.com
Sodium Acetate Chemical Structure Photograph by Molekuul/science Photo What Element Is Acetate Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). It is formed when acetic acid is deprotonated. Acetate is a salt formed by acetic acid with alkaline or other bases. In this section we will look at nomenclature of simple chemical compounds. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. It is. What Element Is Acetate.
From chemistry291.blogspot.com
What is the Chemical Formula for Acetate Ion What Element Is Acetate An acetate, or ethanoate, is a salt or ester of acetic acid. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). The acetate anion, [ch 3 coo] −, is a carboxylate and is the. In this section we will look at nomenclature of simple chemical compounds. Acetate is also a functional group that connects to other molecules,. What Element Is Acetate.
From chemistry291.blogspot.com
What is the Chemical Formula for Acetate Ion What Element Is Acetate It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Acetate is the ion of acetic acid, which is a carboxylic acid. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. Learn how. What Element Is Acetate.
From www.thoughtco.com
Acetate Definition What Is an Acetate? What Element Is Acetate It is formed when acetic acid is deprotonated. Acetate is the ion of acetic acid, which is a carboxylic acid. Acetate is a monocarboxylic acid anion resulting from the removal of a proton from the carboxy group of acetic acid. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. How to quickly check pipettes? Acetate, also. What Element Is Acetate.
From www.dreamstime.com
Molecule of Sodium Acetate. Stock Vector Illustration of infographic What Element Is Acetate Learn how acetate is formed, its properties, and its role in esters and cellulose. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). It is also a carboxylate anion with the formula c2h3o2(−) and a conjugate base of acetic acid. It has a role as a human metabolite and a saccharomyces cerevisiae metabolite. Acetate is a salt. What Element Is Acetate.
From fineartamerica.com
Sodium Acetate Chemical Structure Photograph by Molekuul/science Photo What Element Is Acetate How to quickly check pipettes? Acetate is also a functional group that connects to other molecules, and a salt or ester of acetic acid. The acetate anion, [ch 3 coo] −, is a carboxylate and is the. Acetate, also known as ethanoate, is the salt of acetic acid (vinegar). An acetate, or ethanoate, is a salt or ester of acetic. What Element Is Acetate.