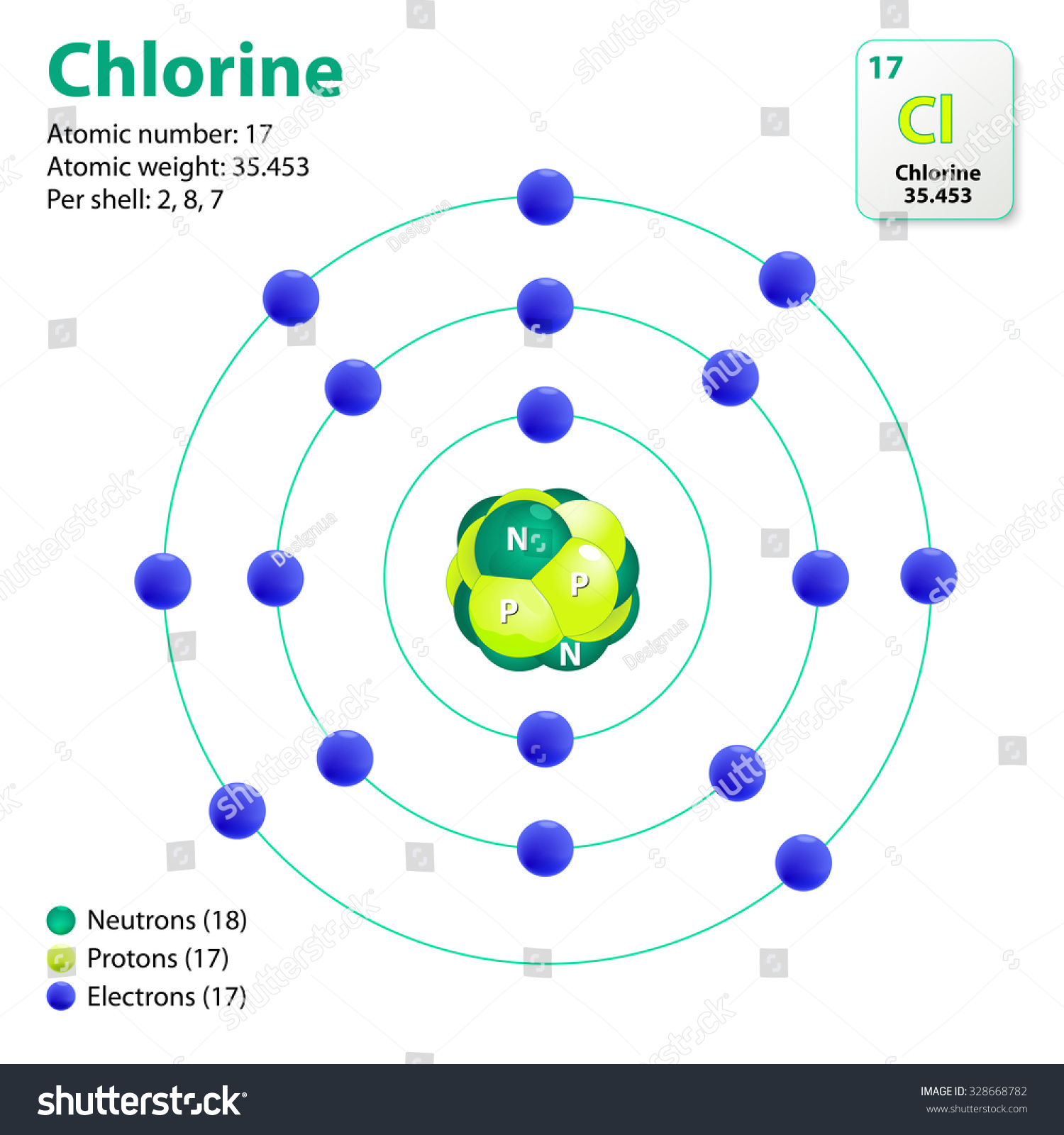
Electron Configuration Of Chlorine Free Radical . in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. a radical is a species with an unpaired electron. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. 2) the electron configuration of chlorine is: species in which an atom has an unpaired electron are called free radicals. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. Full ground state electron configuration: 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. The chlorine molecule has a covalent bond joining the 2 chlorine.
from www.shutterstock.com
a radical is a species with an unpaired electron. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. Full ground state electron configuration: The chlorine molecule has a covalent bond joining the 2 chlorine. 2) the electron configuration of chlorine is: 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. species in which an atom has an unpaired electron are called free radicals. in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open.
Atom Chlorine This Diagram Shows Electron Stock Vector 328668782
Electron Configuration Of Chlorine Free Radical species in which an atom has an unpaired electron are called free radicals. 2) the electron configuration of chlorine is: in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. Full ground state electron configuration: species in which an atom has an unpaired electron are called free radicals. The chlorine molecule has a covalent bond joining the 2 chlorine. a radical is a species with an unpaired electron. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?.
From 88guru.com
Electron Configuration Rules, Example and Diagram 88Guru Electron Configuration Of Chlorine Free Radical species in which an atom has an unpaired electron are called free radicals. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. 2) the. Electron Configuration Of Chlorine Free Radical.
From 88guru.com
Electron Configuration Rules, Example and Diagram 88Guru Electron Configuration Of Chlorine Free Radical revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. 2) the electron configuration of chlorine is: Full ground state electron configuration: The chlorine molecule has a covalent bond joining the 2 chlorine. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of. Electron Configuration Of Chlorine Free Radical.
From manualfixthanedom77.z22.web.core.windows.net
Electron Arrangement Diagram Electron Configuration Of Chlorine Free Radical 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. The chlorine molecule has a covalent bond joining the 2 chlorine. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. 2) the electron configuration of chlorine is: in order to write the. Electron Configuration Of Chlorine Free Radical.
From topblogtenz.com
Chlorine Orbital diagram, Electron configuration, and Valence electrons Electron Configuration Of Chlorine Free Radical The chlorine molecule has a covalent bond joining the 2 chlorine. Full ground state electron configuration: revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. 2) the electron configuration of chlorine is: The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. . Electron Configuration Of Chlorine Free Radical.
From schematicpiscardixf.z4.web.core.windows.net
Orbital Diagram Fluorine Electron Configuration Of Chlorine Free Radical 2) the electron configuration of chlorine is: in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. The chlorine molecule has a covalent bond joining the. Electron Configuration Of Chlorine Free Radical.
From www.embibe.com
Draw the atomic structure of the Chlorine atom and chlorine ion Electron Configuration Of Chlorine Free Radical in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is. Electron Configuration Of Chlorine Free Radical.
From famousparenting.com
How Many Valence Electrons Does Chlorine Have Famous Parenting Electron Configuration Of Chlorine Free Radical The chlorine molecule has a covalent bond joining the 2 chlorine. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. a radical is a species with an unpaired electron. in order to. Electron Configuration Of Chlorine Free Radical.
From chemistry291.blogspot.com
How Many Valence Electrons Does chlorine Have?number of valence Electron Configuration Of Chlorine Free Radical The chlorine molecule has a covalent bond joining the 2 chlorine. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. species in which an atom has an unpaired electron are called free radicals.. Electron Configuration Of Chlorine Free Radical.
From www.thoughtco.com
Atoms Diagrams Electron Configurations of Elements Electron Configuration Of Chlorine Free Radical in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. Full ground state electron configuration: 1s 2 2s 2 2p 6 3s 2. Electron Configuration Of Chlorine Free Radical.
From www.chegg.com
Solved Give the full electron configuration for chlorine Electron Configuration Of Chlorine Free Radical 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. The chlorine molecule has a covalent bond joining the 2 chlorine. 2) the electron configuration of chlorine is: revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. in chemistry,. Electron Configuration Of Chlorine Free Radical.
From www.animalia-life.club
Electron Configuration For Chlorine Electron Configuration Of Chlorine Free Radical in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. 2) the electron configuration of chlorine is: in chemistry, a radical (more precisely, a free radical). Electron Configuration Of Chlorine Free Radical.
From material-properties.org
Chlorine Protons Neutrons Electrons Electron Configuration Electron Configuration Of Chlorine Free Radical 2) the electron configuration of chlorine is: in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. a radical is a species with an unpaired electron. in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired. Electron Configuration Of Chlorine Free Radical.
From basichemistry.blogspot.com
Basic Chemistry Ions, Cations, and Anions Electron Configuration Of Chlorine Free Radical in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. The chlorine molecule has a covalent bond joining the 2 chlorine. a radical is a species with an unpaired electron. in order to write the chlorine electron configuration we first need to know the. Electron Configuration Of Chlorine Free Radical.
From www.newtondesk.com
Chlorine Cl (Element 17) of Periodic Table Newton Desk Electron Configuration Of Chlorine Free Radical a radical is a species with an unpaired electron. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. 2) the electron configuration of chlorine is: Full ground state electron configuration: in order to write the chlorine electron configuration we first need to know the number of electrons. Electron Configuration Of Chlorine Free Radical.
From elchoroukhost.net
Chlorine Periodic Table Electron Configuration Elcho Table Electron Configuration Of Chlorine Free Radical The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. 2) the electron configuration of chlorine is: The chlorine molecule has a covalent bond joining the 2 chlorine. species in which an atom. Electron Configuration Of Chlorine Free Radical.
From www.animalia-life.club
Electron Configuration For Chlorine Electron Configuration Of Chlorine Free Radical in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. species in which an atom has an unpaired electron are called free radicals. 2) the electron configuration of chlorine is: The three free radicals shown in the bottom panel of figure 1 are called. Electron Configuration Of Chlorine Free Radical.
From animalia-life.club
Lewis Dot Structure For Chlorine Electron Configuration Of Chlorine Free Radical The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. Full ground. Electron Configuration Of Chlorine Free Radical.
From www.shutterstock.com
Atom Chlorine This Diagram Shows Electron Stock Vector 328668782 Electron Configuration Of Chlorine Free Radical 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. Full ground state electron configuration: in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that. Electron Configuration Of Chlorine Free Radical.
From brokeasshome.com
Periodic Table Chlorine Electrons Electron Configuration Of Chlorine Free Radical The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. a radical is a species with an unpaired electron. Full ground state electron configuration: in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. 1s 2 2s 2 2p 6. Electron Configuration Of Chlorine Free Radical.
From giowjvcvu.blob.core.windows.net
Chlorine Ion Valence Electrons at John Andrade blog Electron Configuration Of Chlorine Free Radical The chlorine molecule has a covalent bond joining the 2 chlorine. Full ground state electron configuration: 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. species in which an atom. Electron Configuration Of Chlorine Free Radical.
From chamotgallery.com
How many protons, neutrons and electrons does chlorine have? (2023) Electron Configuration Of Chlorine Free Radical revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of. Electron Configuration Of Chlorine Free Radical.
From exyccngyt.blob.core.windows.net
Chlorine Structure Type at Robert Bates blog Electron Configuration Of Chlorine Free Radical Full ground state electron configuration: 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. The chlorine molecule has a covalent bond joining the 2 chlorine. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. species in which an atom has an unpaired. Electron Configuration Of Chlorine Free Radical.
From periodictable.me
Chlorine Electron Configuration (Cl) with Orbital Diagram Electron Configuration Of Chlorine Free Radical 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. species in which an atom has an unpaired electron are called free radicals. in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. a radical is. Electron Configuration Of Chlorine Free Radical.
From chemistry291.blogspot.com
What Is the Chlorine(Cl) Electron Configuration? Electron Configuration Of Chlorine Free Radical 2) the electron configuration of chlorine is: species in which an atom has an unpaired electron are called free radicals. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. Full ground state electron configuration: The chlorine molecule has a covalent bond joining the 2 chlorine. 1s 2 2s. Electron Configuration Of Chlorine Free Radical.
From elchoroukhost.net
Chlorine Periodic Table Electron Configuration Elcho Table Electron Configuration Of Chlorine Free Radical revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. The chlorine molecule has a covalent bond joining the 2 chlorine. species in which an atom has an unpaired electron are called free radicals. in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion. Electron Configuration Of Chlorine Free Radical.
From www.sciencenewsforstudents.org
Explainer Ions and radicals in our world Science News for Students Electron Configuration Of Chlorine Free Radical revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. species in which an atom has an unpaired electron are called free radicals. in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an open. The three free. Electron Configuration Of Chlorine Free Radical.
From odinkruwherrera.blogspot.com
Finish the Electron Configuration for Chlorine 1s22s22p63s2 Electron Configuration Of Chlorine Free Radical revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. 2) the electron configuration of chlorine is: Full ground state electron configuration: 1s 2 2s 2 2p. Electron Configuration Of Chlorine Free Radical.
From mungfali.com
Chlorine Orbital Diagram Electron Configuration Of Chlorine Free Radical in order to write the chlorine electron configuration we first need to know the number of electrons for the cl atom (there. Full ground state electron configuration: revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. a radical is a species with an unpaired electron. The chlorine molecule. Electron Configuration Of Chlorine Free Radical.
From elchoroukhost.net
Chlorine Periodic Table Electron Configuration Elcho Table Electron Configuration Of Chlorine Free Radical a radical is a species with an unpaired electron. species in which an atom has an unpaired electron are called free radicals. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. in order to write the chlorine electron configuration we first need to know the number of. Electron Configuration Of Chlorine Free Radical.
From www.chemistrylearner.com
Chlorine Facts, Symbol, Discovery, Properties, Uses Electron Configuration Of Chlorine Free Radical a radical is a species with an unpaired electron. The chlorine molecule has a covalent bond joining the 2 chlorine. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. in chemistry, a radical (more precisely, a free radical) is an atom, molecule, or ion that has unpaired valence electrons or an. Electron Configuration Of Chlorine Free Radical.
From www.linstitute.net
CIE A Level Chemistry复习笔记1.1.8 Electron Configuration翰林国际教育 Electron Configuration Of Chlorine Free Radical 2) the electron configuration of chlorine is: Full ground state electron configuration: The chlorine molecule has a covalent bond joining the 2 chlorine. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. species in which an atom has an unpaired electron are called free radicals. 1s 2 2s 2 2p 6. Electron Configuration Of Chlorine Free Radical.
From valenceelectrons.com
How Many Valence Electrons Does SO2 (Sulfur Dioxide) Have? Electron Configuration Of Chlorine Free Radical revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. species in which an atom has an unpaired electron are called free radicals. Full ground state electron configuration: in order. Electron Configuration Of Chlorine Free Radical.
From www.youtube.com
Electron Configuration Diagram for Chlorine YouTube Electron Configuration Of Chlorine Free Radical The chlorine molecule has a covalent bond joining the 2 chlorine. species in which an atom has an unpaired electron are called free radicals. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by. Electron Configuration Of Chlorine Free Radical.
From exyjrvgbm.blob.core.windows.net
Cl Negative Electron Configuration at Nicki Craft blog Electron Configuration Of Chlorine Free Radical species in which an atom has an unpaired electron are called free radicals. a radical is a species with an unpaired electron. 1s 2 2s 2 2p 6 3s 2 3p 5 3) what is the molecular geometry of the following?. The three free radicals shown in the bottom panel of figure 1 are called chlorine atoms,. The. Electron Configuration Of Chlorine Free Radical.
From www.alamy.com
Chlorine electron configuration. Illustration of the atomic structure Electron Configuration Of Chlorine Free Radical 2) the electron configuration of chlorine is: a radical is a species with an unpaired electron. The chlorine molecule has a covalent bond joining the 2 chlorine. revision notes on 1.1.5 electron configuration for the aqa a level chemistry syllabus, written by the chemistry experts. Full ground state electron configuration: in chemistry, a radical (more precisely,. Electron Configuration Of Chlorine Free Radical.