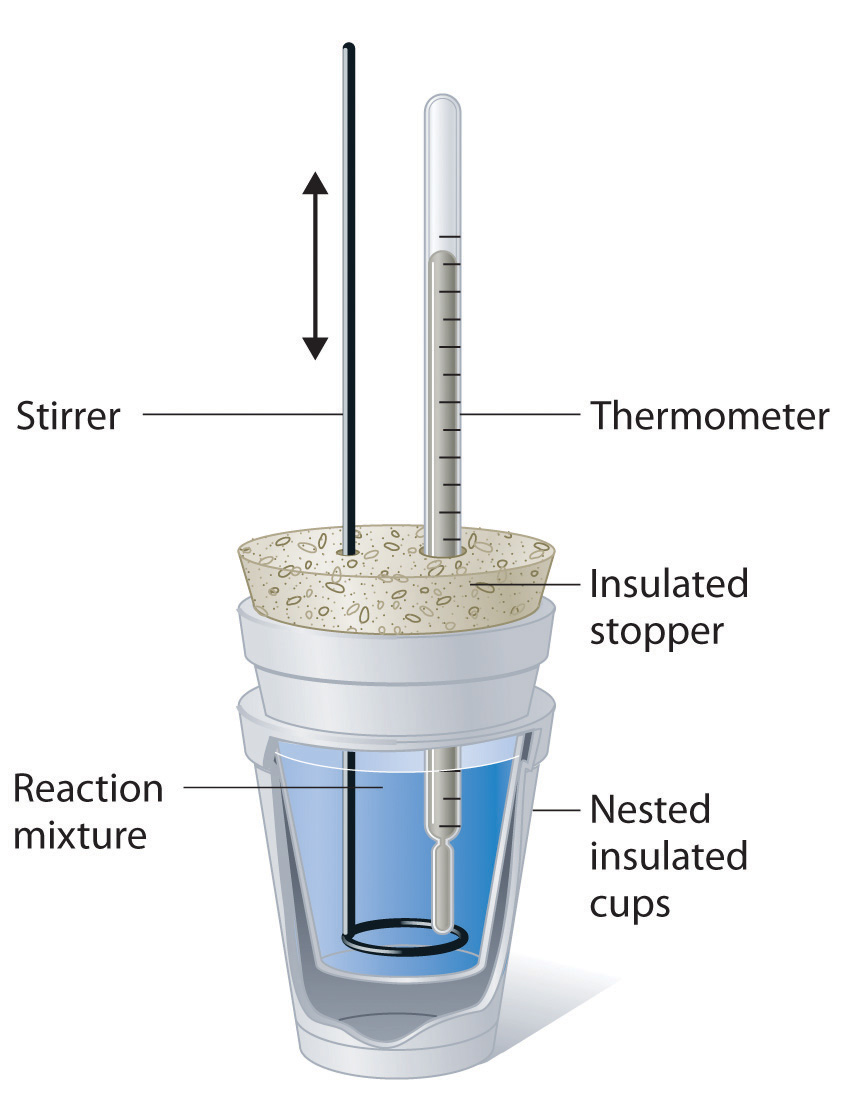
Calorimeter Is Made Up Of Which Material . A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. It mainly consists of a metallic vessel made of materials which are good. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. For example, when an exothermic. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. A calorimeter is a device used for heat measurements necessary for calorimetry. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.
from users.highland.edu
For example, when an exothermic. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. It mainly consists of a metallic vessel made of materials which are good. A calorimeter is a device used for heat measurements necessary for calorimetry. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings.
Calorimetry
Calorimeter Is Made Up Of Which Material Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is a device used for heat measurements necessary for calorimetry. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. It mainly consists of a metallic vessel made of materials which are good. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. For example, when an exothermic. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings.
From www.freeastroscience.com
Exploring Calorimetry A Journey into Heat Transfer Measurement Calorimeter Is Made Up Of Which Material It mainly consists of a metallic vessel made of materials which are good. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. For example, when an. Calorimeter Is Made Up Of Which Material.
From www.embibe.com
Explain the construction of a calorimeter Draw the necessary figure Calorimeter Is Made Up Of Which Material Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. For example, when an exothermic. Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies. Calorimeter Is Made Up Of Which Material.
From www.shaalaa.com
Explain the construction of a calorimeter. Draw the necessary figure Calorimeter Is Made Up Of Which Material A calorimeter is a device used for heat measurements necessary for calorimetry. For example, when an exothermic. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat. Calorimeter Is Made Up Of Which Material.
From www.slideserve.com
PPT Calorimetry PowerPoint Presentation, free download ID3850751 Calorimeter Is Made Up Of Which Material For example, when an exothermic. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. A calorimeter is a device used for heat measurements necessary for. Calorimeter Is Made Up Of Which Material.
From www.scienceabc.com
Molar Heat Capacity Definition, Formula, Equation, Calculation Calorimeter Is Made Up Of Which Material For example, when an exothermic. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. It mainly consists of a metallic vessel made of materials which are good. A calorimeter is a device used for heat measurements necessary for calorimetry. A calorimeter is made up of a conducting vessel to hold. Calorimeter Is Made Up Of Which Material.
From www.youtube.com
How to Draw a Calorimeter Step by Step Drawing Tutorial YouTube Calorimeter Is Made Up Of Which Material A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. For example, when an exothermic. A calorimeter is a device used for heat measurements necessary for calorimetry. Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings.. Calorimeter Is Made Up Of Which Material.
From slidetodoc.com
Differential Scanning Calorimetry By Dr Kundan Tayade Calorimeter Calorimeter Is Made Up Of Which Material It mainly consists of a metallic vessel made of materials which are good. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is a. Calorimeter Is Made Up Of Which Material.
From eduinput.com
CalorimeterDefinition, History, Construction, Types, And Uses Calorimeter Is Made Up Of Which Material A calorimeter is a device used for heat measurements necessary for calorimetry. Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. Calorimetery is an application of. Calorimeter Is Made Up Of Which Material.
From www.studypool.com
SOLUTION What is glass calorimeter explain with diagram and example Calorimeter Is Made Up Of Which Material Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. A calorimeter is a device used to measure the amount of heat. Calorimeter Is Made Up Of Which Material.
From users.highland.edu
Calorimetry Calorimeter Is Made Up Of Which Material Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. For example, when an exothermic. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is made up of a conducting vessel to hold. Calorimeter Is Made Up Of Which Material.
From engineeringlearn.com
Bomb Calorimeter Definition, Construction, Diagram, Working & Uses Calorimeter Is Made Up Of Which Material Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. A calorimeter is a device used for heat. Calorimeter Is Made Up Of Which Material.
From www.youtube.com
Measuring Energy at Constant Volume Using a Bomb Calorimeter YouTube Calorimeter Is Made Up Of Which Material It mainly consists of a metallic vessel made of materials which are good. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. A calorimeter is. Calorimeter Is Made Up Of Which Material.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec Calorimeter Is Made Up Of Which Material Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is a device used for heat measurements necessary for calorimetry. Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. A calorimeter is a device used to measure the. Calorimeter Is Made Up Of Which Material.
From faqguide.co
What does a calorimeter do? Explained by FAQGuide Calorimeter Is Made Up Of Which Material It mainly consists of a metallic vessel made of materials which are good. For example, when an exothermic. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction. Calorimeter Is Made Up Of Which Material.
From www.youtube.com
Constructing a Calorimeter YouTube Calorimeter Is Made Up Of Which Material Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. For example, when an exothermic. A calorimeter is a device used for heat measurements necessary for calorimetry. Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. A calorimeter is a. Calorimeter Is Made Up Of Which Material.
From www.vedantu.com
Bomb Calorimeter Learn Important Terms and Concepts Calorimeter Is Made Up Of Which Material A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. It mainly consists of a metallic vessel made of materials which are good. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the.. Calorimeter Is Made Up Of Which Material.
From www.jove.com
Thermochemistry Constant Volume Calorimetry JoVE Book Calorimeter Is Made Up Of Which Material A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimeters are designed to minimize energy exchange between the system being studied and. Calorimeter Is Made Up Of Which Material.
From www.britannica.com
Calorimeter Definition, Uses, Diagram, & Facts Britannica Calorimeter Is Made Up Of Which Material A calorimeter is a device used for heat measurements necessary for calorimetry. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. For example, when an exothermic. It mainly consists of. Calorimeter Is Made Up Of Which Material.
From www.savemyexams.com
Calorimetry Experiments SL IB Chemistry Revision Notes 2025 Save My Calorimeter Is Made Up Of Which Material A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. Calorimetery is an application of the first law of thermodynamics to heat transfer,. Calorimeter Is Made Up Of Which Material.
From saylordotorg.github.io
Calorimetry Calorimeter Is Made Up Of Which Material For example, when an exothermic. It mainly consists of a metallic vessel made of materials which are good. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. Calorimeters are designed. Calorimeter Is Made Up Of Which Material.
From courses.lumenlearning.com
Calorimetry Chemistry for Majors Calorimeter Is Made Up Of Which Material Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. For example, when an exothermic. It mainly consists of a metallic vessel made of materials which are good. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is. Calorimeter Is Made Up Of Which Material.
From courses.lumenlearning.com
Calorimetry Chemistry I Calorimeter Is Made Up Of Which Material Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. It mainly consists of a metallic vessel made of materials which are good. A calorimeter is a. Calorimeter Is Made Up Of Which Material.
From www.youtube.com
BASIC PRINCIPLE OF CALORIMETRY YouTube Calorimeter Is Made Up Of Which Material A calorimeter is a device used for heat measurements necessary for calorimetry. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. For example, when an exothermic. Calorimeter, device for measuring the heat developed during a. Calorimeter Is Made Up Of Which Material.
From www.studypool.com
SOLUTION Bomb calorimeter study material Studypool Calorimeter Is Made Up Of Which Material It mainly consists of a metallic vessel made of materials which are good. For example, when an exothermic. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. Calorimetery is an application of the first law of thermodynamics to heat transfer, and. Calorimeter Is Made Up Of Which Material.
From stock.adobe.com
Vettoriale Stock illustration of chemistry and physics, Calorimeter Calorimeter Is Made Up Of Which Material A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. For example, when an exothermic. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us. Calorimeter Is Made Up Of Which Material.
From www.thoughtco.com
Calorimeter Definition in Chemistry Calorimeter Is Made Up Of Which Material It mainly consists of a metallic vessel made of materials which are good. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is. Calorimeter Is Made Up Of Which Material.
From byjus.com
The calorimeter is commonly made up of……..because it has……. Calorimeter Is Made Up Of Which Material For example, when an exothermic. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. It mainly consists of a metallic vessel made of materials which are good. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container. Calorimeter Is Made Up Of Which Material.
From www.pinterest.com.au
Pin on Science Friday Calorimeter Is Made Up Of Which Material A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of. Calorimeter Is Made Up Of Which Material.
From www.youtube.com
Thermal Properties of Matter Class 11 Physics Calorimetry Principle Calorimeter Is Made Up Of Which Material Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict heat passage from the. It mainly consists of a metallic vessel made. Calorimeter Is Made Up Of Which Material.
From grade12uchemistry.weebly.com
Calorimetry Grade12UChemistry Calorimeter Is Made Up Of Which Material A calorimeter is a device used for heat measurements necessary for calorimetry. For example, when an exothermic. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. A calorimeter is made up of a conducting vessel to hold the sample, a stirrer, a thermometer, a dewar or insulating container to restrict. Calorimeter Is Made Up Of Which Material.
From www.slideserve.com
PPT a “ Calorimeter ” PowerPoint Presentation, free download ID7050684 Calorimeter Is Made Up Of Which Material Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. For example,. Calorimeter Is Made Up Of Which Material.
From courses.lumenlearning.com
Calorimetry General Chemistry Calorimeter Is Made Up Of Which Material Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. For example, when an exothermic. A calorimeter is made up of a conducting vessel to hold. Calorimeter Is Made Up Of Which Material.
From kaffee.50webs.com
Lab Calorimetry Calorimeter Is Made Up Of Which Material A calorimeter is a device used for heat measurements necessary for calorimetry. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimeters are designed to minimize. Calorimeter Is Made Up Of Which Material.
From www.science-revision.co.uk
Calorimetry Calorimeter Is Made Up Of Which Material A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. For example, when an exothermic. Calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction and for calculating the heat capacity. It mainly consists of a metallic vessel made of materials which are good. A calorimeter is. Calorimeter Is Made Up Of Which Material.
From scienceinfo.com
Calorimeter Definition, Types and Uses Calorimeter Is Made Up Of Which Material Calorimeters are designed to minimize energy exchange between the system being studied and its surroundings. A calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimetery is an application of the first law of thermodynamics to heat transfer, and allows us to measure the enthalpies of reaction or the. It mainly. Calorimeter Is Made Up Of Which Material.