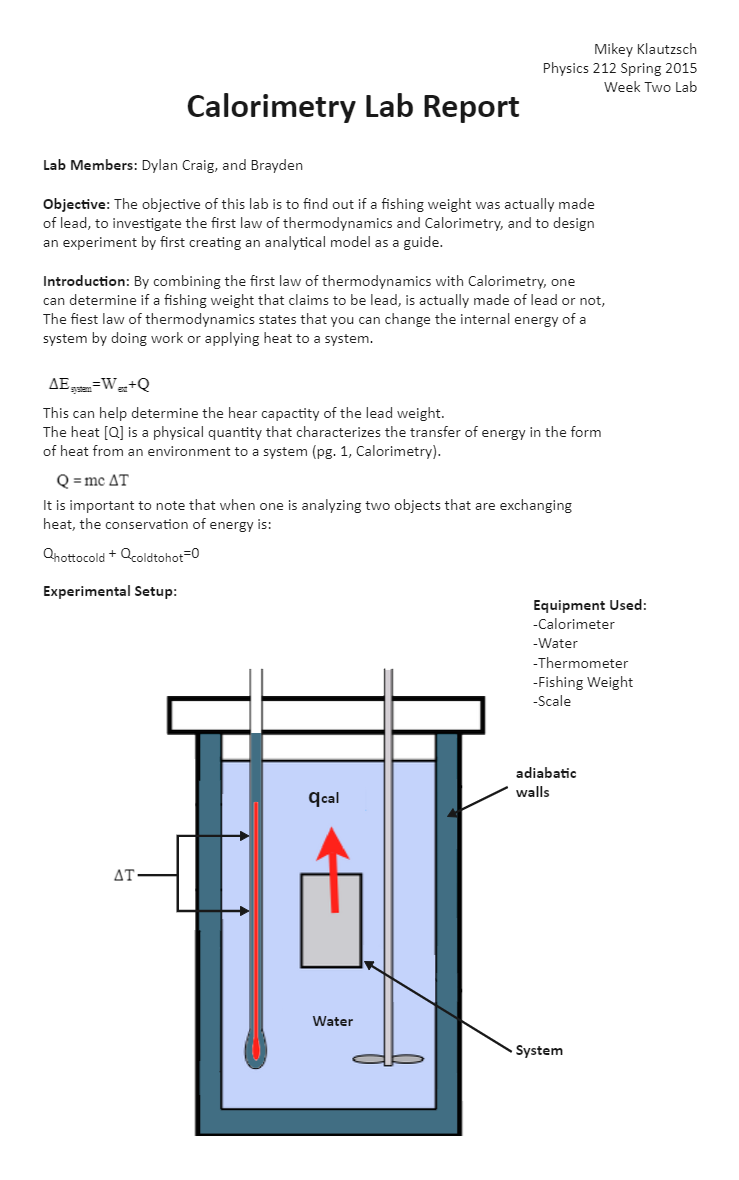
Calorimetry Of Heat . Chemists use calorimetry to determine the. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. Calorimetry is used to measure amounts of heat transferred to or from a substance. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. To do so, the heat is exchanged with a calibrated object (calorimeter). One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction.
from www.edrawmax.com
A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. Chemists use calorimetry to determine the. Calorimetry is used to measure amounts of heat transferred to or from a substance. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process. To do so, the heat is exchanged with a calibrated object (calorimeter).
Calorimetry Lab Report EdrawMax Template
Calorimetry Of Heat One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is used to measure amounts of heat transferred to or from a substance. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. To do so, the heat is exchanged with a calibrated object (calorimeter). One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Chemists use calorimetry to determine the. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction.
From www.youtube.com
09.05 Calorimetry YouTube Calorimetry Of Heat One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. Chemists use. Calorimetry Of Heat.
From www.edrawmax.com
Calorimetry Lab Report EdrawMax Template Calorimetry Of Heat Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. Calorimetry is the process of measuring the amount of. Calorimetry Of Heat.
From users.highland.edu
Calorimetry Calorimetry Of Heat Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from. Calorimetry Of Heat.
From printablelistsaylor.z19.web.core.windows.net
Heat And Calorimetry Worksheets Calorimetry Of Heat One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. Chemists use calorimetry to determine the.. Calorimetry Of Heat.
From www.slideserve.com
PPT Calorimetry PowerPoint Presentation, free download ID6912350 Calorimetry Of Heat A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. One technique we can use to measure the amount of heat. Calorimetry Of Heat.
From janiyahabbgates.blogspot.com
Calorimetry Specific Heat Capacity of Metals Lab Report JaniyahabbGates Calorimetry Of Heat We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical. Calorimetry Of Heat.
From www.youtube.com
CHEMISTRY 101 Constant volume calorimetry YouTube Calorimetry Of Heat A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the measurement of the. Calorimetry Of Heat.
From www.baamboozle.com
Heat Gain and Heat Loss Baamboozle Baamboozle The Most Fun Calorimetry Of Heat One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. To do so, the heat is. Calorimetry Of Heat.
From www.docsity.com
Calorimetry lab report Study Guides, Projects, Research Chemistry Calorimetry Of Heat Calorimetry is used to measure amounts of heat transferred to or from a substance. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. Chemists use calorimetry to determine the. Calorimetry. Calorimetry Of Heat.
From saylordotorg.github.io
Calorimetry Calorimetry Of Heat Chemists use calorimetry to determine the. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. Calorimetry is used to measure amounts of heat transferred to or from a substance. Calorimetry is the measurement of the transfer of. Calorimetry Of Heat.
From www.scienceabc.com
Molar Heat Capacity Definition, Formula, Equation, Calculation Calorimetry Of Heat We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the process of measuring the amount of heat released or absorbed during a. Calorimetry Of Heat.
From www.nagwa.com
Question Video Determining the Correct Formula to Use in Order to Calorimetry Of Heat Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical. Calorimetry Of Heat.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec Calorimetry Of Heat One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. To do so, the heat is. Calorimetry Of Heat.
From grade12uchemistry.weebly.com
Calorimetry Grade12UChemistry Calorimetry Of Heat Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make. Calorimetry Of Heat.
From www.youtube.com
050 Calorimetry YouTube Calorimetry Of Heat Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. To do so, the heat is exchanged with a calibrated object (calorimeter). One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. A container that prevents heat transfer in or out. Calorimetry Of Heat.
From escolagersonalvesgui.blogspot.com
Chemistry Worksheet Heat And Calorimetry Problems Escolagersonalvesgui Calorimetry Of Heat Calorimetry is used to measure amounts of heat transferred to or from a substance. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. We will use the term “calorimetry problem”. Calorimetry Of Heat.
From www.tessshebaylo.com
Equation For Calorimetry Specific Heat Tessshebaylo Calorimetry Of Heat Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. Calorimetry is used to measure amounts of heat transferred to or from a substance. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. To do so, the. Calorimetry Of Heat.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec Calorimetry Of Heat To do so, the heat is exchanged with a calibrated object (calorimeter). Calorimetry is used to measure amounts of heat transferred to or from a substance. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or. Calorimetry Of Heat.
From www.sliderbase.com
Basic Thermochemistry Presentation Chemistry Calorimetry Of Heat Calorimetry is used to measure amounts of heat transferred to or from a substance. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. Calorimetry is a field of thermochemistry. Calorimetry Of Heat.
From pressbooks.online.ucf.edu
10.2 Calorimetry Chemistry Fundamentals Calorimetry Of Heat Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. To do. Calorimetry Of Heat.
From www.wizeprep.com
Calorimetry Wize University Chemistry Textbook Wizeprep Calorimetry Of Heat To do so, the heat is exchanged with a calibrated object (calorimeter). Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. One technique we can use to measure the amount. Calorimetry Of Heat.
From www.youtube.com
Energy 5 Calorimetry/Specific Heat Lab YouTube Calorimetry Of Heat One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process. To do so, the heat is exchanged with a calibrated object (calorimeter). Calorimetry is the process. Calorimetry Of Heat.
From people.chem.umass.edu
to Adobe GoLive 6 Calorimetry Of Heat We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. One technique we can use to measure the amount of heat involved in a chemical or physical process. Calorimetry Of Heat.
From www.youtube.com
BASIC PRINCIPLE OF CALORIMETRY YouTube Calorimetry Of Heat Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. Calorimetry is the process of measuring the amount of heat released or absorbed during a. Calorimetry Of Heat.
From www.youtube.com
Principle of Calorimetry YouTube Calorimetry Of Heat We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. To do so, the heat is exchanged with a calibrated object (calorimeter). One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is the. Calorimetry Of Heat.
From www.chegg.com
Solved connect Calorimetry TRY. HEAT Calorimetry Of Heat To do so, the heat is exchanged with a calibrated object (calorimeter). Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. A container that prevents heat transfer in. Calorimetry Of Heat.
From ecampusontario.pressbooks.pub
5.2 Calorimetry Chemistry Calorimetry Of Heat Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. To do so, the heat is exchanged with a calibrated object (calorimeter). Chemists use calorimetry to determine the. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. We. Calorimetry Of Heat.
From www.studocu.com
Calorimetry and Heat Transfer Calorimetry and Heat Transfer Objective Calorimetry Of Heat Chemists use calorimetry to determine the. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. A container that prevents heat transfer in or out is called a calorimeter, and. Calorimetry Of Heat.
From learningcampusscarf.z13.web.core.windows.net
How To Calculate Heat Of Reaction Calorimetry Calorimetry Of Heat Chemists use calorimetry to determine the. Calorimetry is used to measure amounts of heat transferred to or from a substance. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are. Calorimetry Of Heat.
From www.slideserve.com
PPT Chapter 11 PowerPoint Presentation, free download ID1084959 Calorimetry Of Heat A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Calorimetry is a field of thermochemistry. Calorimetry Of Heat.
From www.youtube.com
Thermal Properties of Matter Class 11 Physics Calorimetry Principle Calorimetry Of Heat One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. Chemists use calorimetry to determine the. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. To do so, the heat is exchanged with a. Calorimetry Of Heat.
From ayanahcristien.blogspot.com
20+ Calculating Heat Of Reaction From ConstantPressure Calorimetry Calorimetry Of Heat Chemists use calorimetry to determine the. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called calorimetry. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their. Calorimetry Of Heat.
From courses.lumenlearning.com
Calorimetry Chemistry for Majors Calorimetry Of Heat We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. Chemists use calorimetry to determine the. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity) is called. Calorimetry Of Heat.
From answerhappy.com
CALORIMETRY HEAT CAPACITY OF A CALORIMETER NTRODUCTION Lab Data Calorimetry Of Heat Calorimetry is the measurement of the transfer of heat into or out of a system during a chemical reaction or physical process. We will use the term “calorimetry problem” to refer to any problem in which the objects concerned are thermally isolated from their surroundings. Chemists use calorimetry to determine the. One technique we can use to measure the amount. Calorimetry Of Heat.
From www.linstitute.net
Edexcel A Level Chemistry复习笔记1.8.3 Calorimetry翰林国际教育 Calorimetry Of Heat To do so, the heat is exchanged with a calibrated object (calorimeter). Calorimetry is a field of thermochemistry that measures the amount of heat involved in a physical or chemical reaction. A container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements (typically of heat or specific heat capacity). Calorimetry Of Heat.