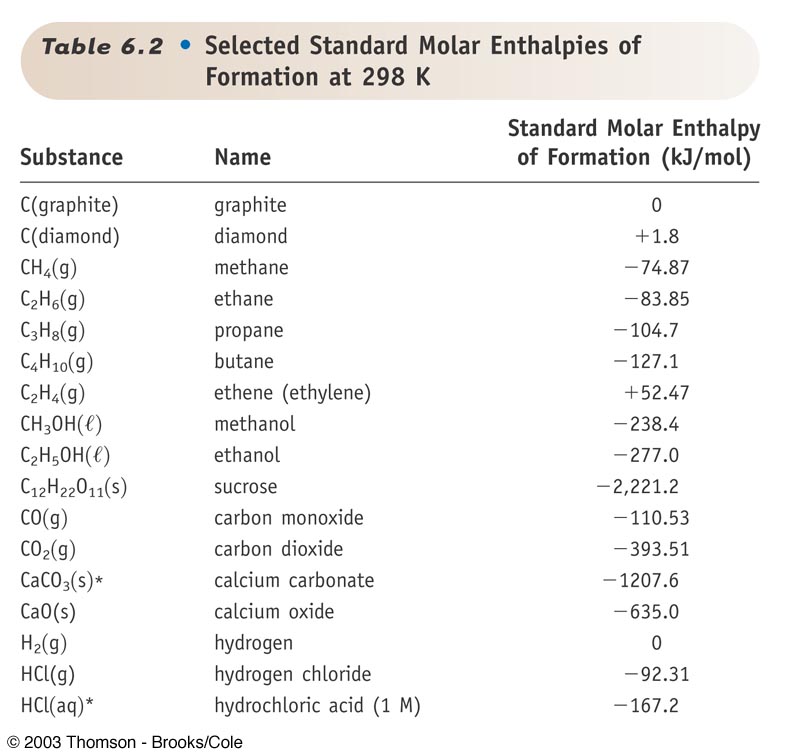
Standard Heat Formation Of Potassium Chloride . For this compound, wtt contains critically evaluated recommendations for: To find the δh reaction o, use the formula for the standard enthalpy change of formation: (please note that if more than 50 points are used for regression,. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. \[\delta h_{reaction}^o = \sum {\delta. The standard state heat of formation for the elemental form of each atom is zero. This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of.
from lessonluft.z19.web.core.windows.net
Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. To find the δh reaction o, use the formula for the standard enthalpy change of formation: For this compound, wtt contains critically evaluated recommendations for: This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. The standard state heat of formation for the elemental form of each atom is zero. \[\delta h_{reaction}^o = \sum {\delta. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. (please note that if more than 50 points are used for regression,. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements.
Heat Of Formation Chart
Standard Heat Formation Of Potassium Chloride \[\delta h_{reaction}^o = \sum {\delta. For this compound, wtt contains critically evaluated recommendations for: This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. (please note that if more than 50 points are used for regression,. To find the δh reaction o, use the formula for the standard enthalpy change of formation: 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard state heat of formation for the elemental form of each atom is zero. \[\delta h_{reaction}^o = \sum {\delta. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including.
From www.researchgate.net
Heat capacity of potassium chloride. Download Scientific Diagram Standard Heat Formation Of Potassium Chloride To find the δh reaction o, use the formula for the standard enthalpy change of formation: 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including.. Standard Heat Formation Of Potassium Chloride.
From www.lookfordiagnosis.com
Potassium chloride Standard Heat Formation Of Potassium Chloride Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. To find the δh reaction o, use the formula for the standard enthalpy change of formation: 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of.. Standard Heat Formation Of Potassium Chloride.
From www.chem.fsu.edu
CHM1045 Enthalpy Lecture Standard Heat Formation Of Potassium Chloride For this compound, wtt contains critically evaluated recommendations for: To find the δh reaction o, use the formula for the standard enthalpy change of formation: The standard state heat of formation for the elemental form of each atom is zero. \[\delta h_{reaction}^o = \sum {\delta. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of. Standard Heat Formation Of Potassium Chloride.
From www.chegg.com
Solved A solubility curve for KCl in water as a function of Standard Heat Formation Of Potassium Chloride This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. (please note that if more than 50 points are used for regression,. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard state heat of formation. Standard Heat Formation Of Potassium Chloride.
From ar.inspiredpencil.com
Potassium Chloride Diagram Standard Heat Formation Of Potassium Chloride The standard state heat of formation for the elemental form of each atom is zero. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. To find the. Standard Heat Formation Of Potassium Chloride.
From animalia-life.club
Potassium Chloride Shell Model Standard Heat Formation Of Potassium Chloride To find the δh reaction o, use the formula for the standard enthalpy change of formation: Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. \[\delta h_{reaction}^o = \sum {\delta. The standard state heat of formation for the elemental form of each atom is zero. 193 rows in. Standard Heat Formation Of Potassium Chloride.
From www.chegg.com
Solved Use a standard enthalpies of formation table to Standard Heat Formation Of Potassium Chloride 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. To find the δh reaction o, use the formula for the standard enthalpy change of formation: This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. Standard enthalpy. Standard Heat Formation Of Potassium Chloride.
From www.priyamstudycentre.com
Potassium Chloride (KCl) Uses, Medication, Chemistry Standard Heat Formation Of Potassium Chloride (please note that if more than 50 points are used for regression,. The standard state heat of formation for the elemental form of each atom is zero. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or. Standard Heat Formation Of Potassium Chloride.
From www.researchgate.net
Potassium chloride standardised Download Table Standard Heat Formation Of Potassium Chloride Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. To find the δh reaction o, use the formula for the standard enthalpy change of formation: For this compound, wtt contains critically evaluated recommendations for: \[\delta h_{reaction}^o = \sum {\delta. The standard state heat of formation for the elemental. Standard Heat Formation Of Potassium Chloride.
From www.youtube.com
How to Accurately Calculate the Heat of Dissolution of Potassium Standard Heat Formation Of Potassium Chloride \[\delta h_{reaction}^o = \sum {\delta. (please note that if more than 50 points are used for regression,. To find the δh reaction o, use the formula for the standard enthalpy change of formation: Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. The enthalpy of formation (\(δh_{f}\)) is. Standard Heat Formation Of Potassium Chloride.
From www.chegg.com
Solved et ThermodynamicS The standard heat of formation of Standard Heat Formation Of Potassium Chloride This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. (please note that if more than 50 points are used for regression,. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. For this compound, wtt contains critically evaluated recommendations for: To. Standard Heat Formation Of Potassium Chloride.
From animalia-life.club
Potassium Chloride Shell Model Standard Heat Formation Of Potassium Chloride To find the δh reaction o, use the formula for the standard enthalpy change of formation: (please note that if more than 50 points are used for regression,. This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. For this compound, wtt contains critically evaluated recommendations for: The standard state heat. Standard Heat Formation Of Potassium Chloride.
From quizzlistreplevies.z13.web.core.windows.net
Heat Of Formation Equations Standard Heat Formation Of Potassium Chloride (please note that if more than 50 points are used for regression,. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. To find the δh reaction o, use the formula for the standard enthalpy change of formation: For this compound, wtt contains critically evaluated recommendations for: The enthalpy. Standard Heat Formation Of Potassium Chloride.
From www.numerade.com
SOLVED QUESTION 14 In the presence of heat potassium chlorate Standard Heat Formation Of Potassium Chloride To find the δh reaction o, use the formula for the standard enthalpy change of formation: The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. The standard state heat of formation for the elemental form of each atom is zero. Standard enthalpy change of formation (data table) these tables include. Standard Heat Formation Of Potassium Chloride.
From ar.inspiredpencil.com
Potassium Chloride Lewis Structure Standard Heat Formation Of Potassium Chloride The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. For this compound, wtt contains critically evaluated recommendations for: The standard state heat of formation for the elemental form of each atom is zero. To find the δh reaction o, use the formula for the standard enthalpy change of formation: (please. Standard Heat Formation Of Potassium Chloride.
From worksheetzonepatine.z14.web.core.windows.net
How To Determine The Heat Of Formation Standard Heat Formation Of Potassium Chloride The standard state heat of formation for the elemental form of each atom is zero. (please note that if more than 50 points are used for regression,. To find the δh reaction o, use the formula for the standard enthalpy change of formation: This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800. Standard Heat Formation Of Potassium Chloride.
From www.examples.com
Potassium Chloride (KCl) Definition, Structure, Properties, Uses Standard Heat Formation Of Potassium Chloride To find the δh reaction o, use the formula for the standard enthalpy change of formation: 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. \[\delta h_{reaction}^o = \sum {\delta. (please note that if more than 50 points are used for regression,. Standard enthalpy change of. Standard Heat Formation Of Potassium Chloride.
From www.showme.com
Standard heat of formation Science, Chemistry, thermochemistry ShowMe Standard Heat Formation Of Potassium Chloride The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. The standard state heat of formation for the elemental form of each atom is zero. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. This examination indicated that the. Standard Heat Formation Of Potassium Chloride.
From animalia-life.club
Potassium Chloride Shell Model Standard Heat Formation Of Potassium Chloride This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. \[\delta h_{reaction}^o = \sum {\delta. (please note that if more than 50 points are used for regression,. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. For. Standard Heat Formation Of Potassium Chloride.
From sciencekitstore.com
Potassium Chloride, USP/FCC, Fine crystal Standard Heat Formation Of Potassium Chloride For this compound, wtt contains critically evaluated recommendations for: This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. (please note that if more than 50 points are used for regression,. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. Standard Heat Formation Of Potassium Chloride.
From www.chegg.com
Solved Calculate the standard heat of the neutralization Standard Heat Formation Of Potassium Chloride This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. For this compound, wtt contains critically evaluated recommendations for: (please note that if more than 50 points are used for regression,. The standard state heat of formation for the elemental form of each atom is zero. To find the δh reaction. Standard Heat Formation Of Potassium Chloride.
From www.numerade.com
SOLVEDConsider the formation of a solution of aqueous potassium Standard Heat Formation Of Potassium Chloride For this compound, wtt contains critically evaluated recommendations for: To find the δh reaction o, use the formula for the standard enthalpy change of formation: Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. The standard state heat of formation for the elemental form of each atom is. Standard Heat Formation Of Potassium Chloride.
From wisc.pb.unizin.org
Acids, Bases, Neutralization, and GasForming Reactions (M3Q34) UW Standard Heat Formation Of Potassium Chloride To find the δh reaction o, use the formula for the standard enthalpy change of formation: (please note that if more than 50 points are used for regression,. The standard state heat of formation for the elemental form of each atom is zero. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a. Standard Heat Formation Of Potassium Chloride.
From www.thesciencehive.co.uk
Lattice Enthalpy* — the science sauce Standard Heat Formation Of Potassium Chloride \[\delta h_{reaction}^o = \sum {\delta. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. For this compound, wtt contains critically evaluated recommendations for: This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. The standard state heat of formation. Standard Heat Formation Of Potassium Chloride.
From www.researchgate.net
Calculated enthalpy increments of solid and liquid potassium chloride Standard Heat Formation Of Potassium Chloride The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. \[\delta h_{reaction}^o = \sum {\delta. The standard state heat of formation for the elemental form of each atom is zero. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the. Standard Heat Formation Of Potassium Chloride.
From readingandwritingprojectcom.web.fc2.com
potassium chloride ionic or covalent Standard Heat Formation Of Potassium Chloride For this compound, wtt contains critically evaluated recommendations for: This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. To find the δh reaction o, use the formula. Standard Heat Formation Of Potassium Chloride.
From animalia-life.club
Potassium Chloride Shell Model Standard Heat Formation Of Potassium Chloride Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. For this compound, wtt contains critically evaluated recommendations for: \[\delta h_{reaction}^o = \sum {\delta. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. (please note. Standard Heat Formation Of Potassium Chloride.
From www.toppr.com
Draw electron dot representation for the formation of potassium chloride. Standard Heat Formation Of Potassium Chloride For this compound, wtt contains critically evaluated recommendations for: Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation. Standard Heat Formation Of Potassium Chloride.
From lessonluft.z19.web.core.windows.net
Heat Of Formation Chart Standard Heat Formation Of Potassium Chloride To find the δh reaction o, use the formula for the standard enthalpy change of formation: This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. \[\delta h_{reaction}^o = \sum {\delta. For this compound, wtt contains critically evaluated recommendations for: The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies. Standard Heat Formation Of Potassium Chloride.
From www.researchgate.net
The wavelength versus absorption for the potassium chloride (KCl Standard Heat Formation Of Potassium Chloride For this compound, wtt contains critically evaluated recommendations for: The standard state heat of formation for the elemental form of each atom is zero. To find the δh reaction o, use the formula for the standard enthalpy change of formation: Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources,. Standard Heat Formation Of Potassium Chloride.
From www.numerade.com
SOLVED a) Calculate the standard heat of the neutralization reaction Standard Heat Formation Of Potassium Chloride \[\delta h_{reaction}^o = \sum {\delta. 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. The standard state heat of formation for the elemental form of each atom is zero. Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a. Standard Heat Formation Of Potassium Chloride.
From www.slideserve.com
PPT Ch. 10 Chemical Bonding PowerPoint Presentation, free download Standard Heat Formation Of Potassium Chloride To find the δh reaction o, use the formula for the standard enthalpy change of formation: The enthalpy of formation (\(δh_{f}\)) is the enthalpy change that accompanies the formation of a compound from its elements. This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. Standard enthalpy change of formation (data. Standard Heat Formation Of Potassium Chloride.
From www.chegg.com
Solved a) Calculate the standard heat of the neutralization Standard Heat Formation Of Potassium Chloride Standard enthalpy change of formation (data table) these tables include heat of formation data gathered from a variety of sources, including. To find the δh reaction o, use the formula for the standard enthalpy change of formation: The standard state heat of formation for the elemental form of each atom is zero. The enthalpy of formation (\(δh_{f}\)) is the enthalpy. Standard Heat Formation Of Potassium Chloride.
From ar.inspiredpencil.com
Potassium Chloride Diagram Standard Heat Formation Of Potassium Chloride The standard state heat of formation for the elemental form of each atom is zero. \[\delta h_{reaction}^o = \sum {\delta. To find the δh reaction o, use the formula for the standard enthalpy change of formation: 193 rows in chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of. This. Standard Heat Formation Of Potassium Chloride.
From www.numerade.com
Use the following data for potassium chloride to Standard Heat Formation Of Potassium Chloride For this compound, wtt contains critically evaluated recommendations for: This examination indicated that the nist recommendation of heating standard reference material 1655, potassium chloride, at 800 k for. The standard state heat of formation for the elemental form of each atom is zero. To find the δh reaction o, use the formula for the standard enthalpy change of formation: Standard. Standard Heat Formation Of Potassium Chloride.