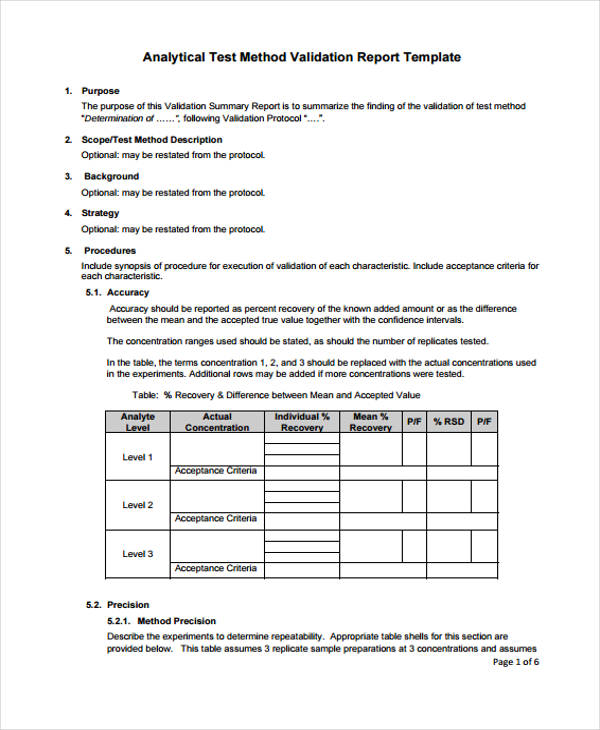
Instrument Validation Protocol . method validation is performed when a lab acquires a new instrument and is testing a new procedure; there are eleven main principles to the pq laboratory test. Derive and evaluate the various validation tests for. A document which contains all the. It is used for assessing if. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. q2(r2) provides guidance and recommendations on how to.
from www.template.net
method validation is performed when a lab acquires a new instrument and is testing a new procedure; It is used for assessing if. q2(r2) provides guidance and recommendations on how to. there are eleven main principles to the pq laboratory test. Derive and evaluate the various validation tests for. A document which contains all the. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in.
10+ Validation Report Templates Free Sample, Example Format Download
Instrument Validation Protocol an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. method validation is performed when a lab acquires a new instrument and is testing a new procedure; Derive and evaluate the various validation tests for. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. It is used for assessing if. q2(r2) provides guidance and recommendations on how to. there are eleven main principles to the pq laboratory test. A document which contains all the.
From guideline-sop.com
Process Validation (PV) & Verification of Drug Product Guidelines SOPs Instrument Validation Protocol there are eleven main principles to the pq laboratory test. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. Derive and evaluate the various validation tests for. It is used for assessing if. an equipment validation protocol (also known as an equipment qualification protocol) is a. Instrument Validation Protocol.
From www.researchgate.net
Survey instrument development and validation process Download Instrument Validation Protocol method validation is performed when a lab acquires a new instrument and is testing a new procedure; there are eleven main principles to the pq laboratory test. Derive and evaluate the various validation tests for. q2(r2) provides guidance and recommendations on how to. in this paper, we present data to illustrate the validation of a procedure. Instrument Validation Protocol.
From www.scribd.com
Instrument Validation and Inspection Methods Verification And Instrument Validation Protocol Derive and evaluate the various validation tests for. It is used for assessing if. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. q2(r2) provides guidance and recommendations on how to. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan. Instrument Validation Protocol.
From pt.scribd.com
Template for Process Validation Protocol Verification And Validation Instrument Validation Protocol in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. It is used for assessing if. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. A document which contains all the. method validation is performed when a lab acquires. Instrument Validation Protocol.
From pharmdguru.com
3. VALIDATION METHODS QUALITY OF EQUIPMENT, VALIDATION OF EQUIPMENT Instrument Validation Protocol Derive and evaluate the various validation tests for. It is used for assessing if. A document which contains all the. there are eleven main principles to the pq laboratory test. method validation is performed when a lab acquires a new instrument and is testing a new procedure; an equipment validation protocol (also known as an equipment qualification. Instrument Validation Protocol.
From www.slideserve.com
PPT INSTRUMENT AND TEST METHOD VALIDATION PowerPoint Presentation Instrument Validation Protocol It is used for assessing if. A document which contains all the. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. method validation is performed when a lab acquires a new instrument and is testing a new procedure; q2(r2) provides guidance and recommendations on how to.. Instrument Validation Protocol.
From www.getreskilled.com
Equipment Qualification Protocol Step by Step Writing Guide Instrument Validation Protocol method validation is performed when a lab acquires a new instrument and is testing a new procedure; there are eleven main principles to the pq laboratory test. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. Derive and evaluate the various validation tests for. in this paper, we. Instrument Validation Protocol.
From www.presentationeze.com
Equipment Validation Facility Qualification Material Instrument Validation Protocol It is used for assessing if. A document which contains all the. there are eleven main principles to the pq laboratory test. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. method validation is performed when a lab acquires a new instrument and is testing a. Instrument Validation Protocol.
From www.researchgate.net
Template of a validation plan. Download Scientific Diagram Instrument Validation Protocol in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. A document which contains all the. Derive and evaluate the various validation tests for. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. method validation is performed when a. Instrument Validation Protocol.
From ivypanda.com
Instrument Validation and Inspection Methods 617 Words Research Instrument Validation Protocol q2(r2) provides guidance and recommendations on how to. method validation is performed when a lab acquires a new instrument and is testing a new procedure; Derive and evaluate the various validation tests for. A document which contains all the. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. It. Instrument Validation Protocol.
From templates.rjuuc.edu.np
Equipment Validation Protocol Template Instrument Validation Protocol q2(r2) provides guidance and recommendations on how to. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. It is used for assessing if. method validation is performed when a lab acquires a new instrument and is testing a new procedure; there are eleven main principles to the pq. Instrument Validation Protocol.
From present5.com
Current Validation Process and Issues Exemplified by the Instrument Validation Protocol It is used for assessing if. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. q2(r2) provides guidance and recommendations on how to. method validation is performed when a lab acquires a new instrument and is testing a new procedure; there are eleven main principles. Instrument Validation Protocol.
From issuu.com
IOPQ Freezer Validation Template Sample by Pharmi Med Ltd Issuu Instrument Validation Protocol in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. there are eleven main principles to the pq laboratory test. Derive and evaluate the various validation tests for. method validation is performed when a lab acquires a new instrument and is testing a new procedure; A document. Instrument Validation Protocol.
From en.ppt-online.org
Method Validation and Verification Protocols for Test Methods online Instrument Validation Protocol Derive and evaluate the various validation tests for. A document which contains all the. q2(r2) provides guidance and recommendations on how to. It is used for assessing if. method validation is performed when a lab acquires a new instrument and is testing a new procedure; an equipment validation protocol (also known as an equipment qualification protocol) is. Instrument Validation Protocol.
From www.scribd.com
Process Validation Sample Protocol Verification And Validation Instrument Validation Protocol method validation is performed when a lab acquires a new instrument and is testing a new procedure; q2(r2) provides guidance and recommendations on how to. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. Derive and evaluate the various validation tests for. there are eleven main principles to. Instrument Validation Protocol.
From www.orielstat.com
Creating a Medical Device Process Validation Plan and Protocols Instrument Validation Protocol Derive and evaluate the various validation tests for. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. A document which contains all the. It is used for assessing if. q2(r2) provides guidance and recommendations on how to. there are eleven main principles to the pq laboratory test. method. Instrument Validation Protocol.
From www.researchgate.net
(PDF) Developing and Validating an Instrument for Evaluating Research Instrument Validation Protocol an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. there are eleven main principles to the pq laboratory test. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. It is used for assessing if. q2(r2) provides guidance. Instrument Validation Protocol.
From www.slideserve.com
PPT Flight Validation of Instrument Flight Procedures PowerPoint Instrument Validation Protocol A document which contains all the. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. method validation is performed when a lab acquires a new instrument and is testing. Instrument Validation Protocol.
From www.researchgate.net
(PDF) Method of preparing a document for survey instrument validation Instrument Validation Protocol It is used for assessing if. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. method validation is performed when a lab acquires a new instrument and is testing a new procedure; q2(r2) provides guidance and recommendations on how to. there are eleven main principles to the pq. Instrument Validation Protocol.
From www.slideserve.com
PPT Replacement Reagent Policy Update PowerPoint Presentation, free Instrument Validation Protocol Derive and evaluate the various validation tests for. method validation is performed when a lab acquires a new instrument and is testing a new procedure; It is used for assessing if. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. q2(r2) provides guidance and recommendations on how to. . Instrument Validation Protocol.
From templates.rjuuc.edu.np
Equipment Validation Protocol Template Instrument Validation Protocol A document which contains all the. It is used for assessing if. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. there are eleven main principles to the pq. Instrument Validation Protocol.
From www.template.net
10+ Validation Report Templates Free Sample, Example Format Download Instrument Validation Protocol there are eleven main principles to the pq laboratory test. A document which contains all the. method validation is performed when a lab acquires a new instrument and is testing a new procedure; in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. Derive and evaluate the. Instrument Validation Protocol.
From www.validation-online.net
Validation Protocol Standards FDA MHRA EC WHO TPD TGA Instrument Validation Protocol It is used for assessing if. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. q2(r2) provides guidance and recommendations on how to. there are eleven main principles to the pq laboratory test. in this paper, we present data to illustrate the validation of a procedure for the. Instrument Validation Protocol.
From www.slideshare.net
Validation Protocol Instrument Validation Protocol method validation is performed when a lab acquires a new instrument and is testing a new procedure; Derive and evaluate the various validation tests for. A document which contains all the. It is used for assessing if. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. q2(r2) provides guidance. Instrument Validation Protocol.
From www.allbusinesstemplates.com
Process Validation Templates at Instrument Validation Protocol q2(r2) provides guidance and recommendations on how to. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. A document which contains all the. there are eleven main principles to the pq laboratory test. method validation is performed when a lab acquires a new instrument and is testing a. Instrument Validation Protocol.
From www.presentationeze.com
Medical Device Validation Full Details PresentationEZE Instrument Validation Protocol It is used for assessing if. in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. q2(r2) provides guidance and recommendations on how to. there are eleven main principles. Instrument Validation Protocol.
From www.sampletemplates.com
FREE 9+ Sample Validation Plan Templates in PDF MS Word Instrument Validation Protocol q2(r2) provides guidance and recommendations on how to. there are eleven main principles to the pq laboratory test. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. method validation is performed when a lab acquires a new instrument and is testing a new procedure; Derive and evaluate the. Instrument Validation Protocol.
From www.getreskilled.com
Equipment Validation Protocol Step by Step Writing Guide Instrument Validation Protocol there are eleven main principles to the pq laboratory test. It is used for assessing if. method validation is performed when a lab acquires a new instrument and is testing a new procedure; in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. A document which contains. Instrument Validation Protocol.
From www.orielstat.com
Medical Device Process Validation What You Need to Know Instrument Validation Protocol method validation is performed when a lab acquires a new instrument and is testing a new procedure; in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. q2(r2) provides guidance and recommendations on how to. A document which contains all the. Derive and evaluate the various validation. Instrument Validation Protocol.
From www.slideserve.com
PPT VALIDATION METHODOLOGY PowerPoint Presentation, free download Instrument Validation Protocol in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. Derive and evaluate the various validation tests for. method validation is performed when a lab acquires a new instrument and is testing a new procedure; there are eleven main principles to the pq laboratory test. an. Instrument Validation Protocol.
From www.youtube.com
How to Write a Validation Protocol Different Parts of Validation Instrument Validation Protocol there are eleven main principles to the pq laboratory test. A document which contains all the. an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. Derive and evaluate the various validation tests for. q2(r2) provides guidance and recommendations on how to. method validation is performed when a lab. Instrument Validation Protocol.
From www.getreskilled.com
What's a Pharmaceutical Equipment Validation Protocol & Why is it Crucial? Instrument Validation Protocol in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. method validation is performed when a lab acquires a new instrument and is testing a new procedure; q2(r2) provides guidance and recommendations on how to. Derive and evaluate the various validation tests for. an equipment validation. Instrument Validation Protocol.
From www.getreskilled.com
What's an IQ OQ PQ Validation Protocol & why's it critical in pharma? Instrument Validation Protocol in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. It is used for assessing if. there are eleven main principles to the pq laboratory test. method validation is performed when a lab acquires a new instrument and is testing a new procedure; an equipment validation. Instrument Validation Protocol.
From www.researchgate.net
(PDF) Validation Instrument for Undergraduate Qualitative Research Instrument Validation Protocol an equipment validation protocol (also known as an equipment qualification protocol) is a written plan that outlines. It is used for assessing if. Derive and evaluate the various validation tests for. A document which contains all the. method validation is performed when a lab acquires a new instrument and is testing a new procedure; there are eleven. Instrument Validation Protocol.
From ciqa.net
How to create a Validation Master Plan in 5 steps. Templates & more Instrument Validation Protocol method validation is performed when a lab acquires a new instrument and is testing a new procedure; in this paper, we present data to illustrate the validation of a procedure for the measurement of elemental impurities in. there are eleven main principles to the pq laboratory test. q2(r2) provides guidance and recommendations on how to. . Instrument Validation Protocol.