A Laboratory Stock Solution Is 1.50 . A laboratory stock solution is 1.50m sodium hydroxide. Concentrated volume = 40 ml. Concentrated volume = (300 ml. 【solved】click here to get an answer to your question : Here’s how to approach this question. C 1 v 1 = c 2 v 2 Click here to get an answer to your question: Calculate the volume of this stock solution that would be needed to prepare 300. Calculate the volume of this stock solution that. A laboratory stock solution is 1.50 m naoh. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. Calculate the volume of this. We're asked to calculate the volume, the volume of stock. 0.200 m) / 1.50 m. To start solving this problem, use the dilution equation,.
from www.chegg.com
We're told that the lab stock solution is 1.50 sodium hydroxide. Calculate the volume of this stock solution that would be needed to prepare 300. A laboratory stock solution is 1.50m sodium hydroxide. We're asked to calculate the volume, the volume of stock. C 1 v 1 = c 2 v 2 0.200 m) / 1.50 m. To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution, we can use the formula for dilution: A laboratory stock solution is 1.50 m naoh. When the dilution factor is applied, it is. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final.
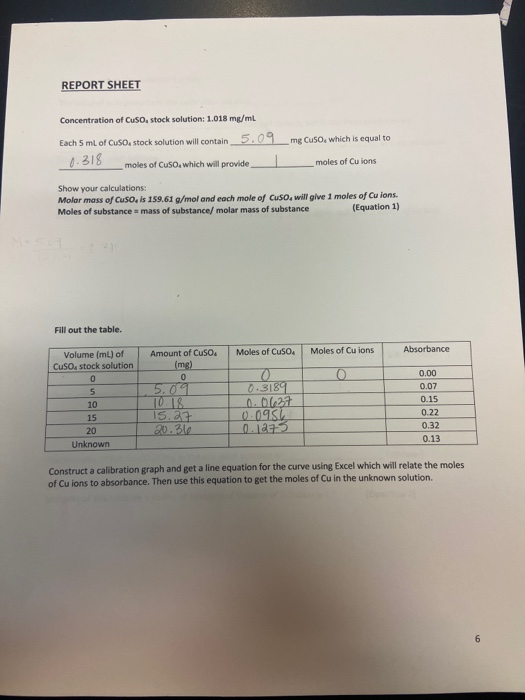
Solved REPORT SHEET Concentration of Cuso, stock solution
A Laboratory Stock Solution Is 1.50 Calculate the volume of this stock solution that. A laboratory stock solution is 1.50 m naoh. A laboratory stock solution is 1.50 m naoh. A laboratory stock solution is 1.50m sodium hydroxide. To start solving this problem, use the dilution equation,. Click here to get an answer to your question: Calculate the volume of this. 0.200 m) / 1.50 m. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. We're asked to calculate the volume, the volume of stock. Calculate the volume of this stock solution that. C 1 v 1 = c 2 v 2 Concentrated volume = (300 ml. To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution, we can use the formula for dilution: 【solved】click here to get an answer to your question : When the dilution factor is applied, it is.
From www.chegg.com
Solved Prelaboratory Questions 1. A stock solution of A Laboratory Stock Solution Is 1.50 We're told that the lab stock solution is 1.50 sodium hydroxide. 【solved】click here to get an answer to your question : Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. To start solving this problem, use the dilution equation,. 0.200 m) / 1.50 m. Concentrated volume =. A Laboratory Stock Solution Is 1.50.
From www.flinnsci.com
Laboratory Solution Preparation Flinn Scientific A Laboratory Stock Solution Is 1.50 A laboratory stock solution is 1.50m sodium hydroxide. 【solved】click here to get an answer to your question : To start solving this problem, use the dilution equation,. Calculate the volume of this stock solution that. Calculate the volume of this stock solution that would be needed to prepare 300. Use the dilution formula, m1v1 = m2v2, where m1 is the. A Laboratory Stock Solution Is 1.50.
From www.researchgate.net
STOCK SOLUTION PREPARATION Download Table A Laboratory Stock Solution Is 1.50 Concentrated volume = 40 ml. 0.200 m) / 1.50 m. Concentrated volume = (300 ml. Calculate the volume of this. 【solved】click here to get an answer to your question : To start solving this problem, use the dilution equation,. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the. A Laboratory Stock Solution Is 1.50.
From www.thinkswap.com
Chemistry Lab Report Preparation Of Solution SCE1014 Chemistry I A Laboratory Stock Solution Is 1.50 To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution, we can use the formula for dilution: Click here to get an answer to your question: Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. We're told that the lab stock solution. A Laboratory Stock Solution Is 1.50.
From www.youtube.com
Calculating moles in the lab, diluting stock solutions Part 2 YouTube A Laboratory Stock Solution Is 1.50 A laboratory stock solution is 1.50 m naoh. C 1 v 1 = c 2 v 2 Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. We're told that the lab stock solution is 1.50 sodium hydroxide. Calculate the volume of this. Concentrated volume = 40 ml.. A Laboratory Stock Solution Is 1.50.
From www.youtube.com
Stock solutions and dilution YouTube A Laboratory Stock Solution Is 1.50 C 1 v 1 = c 2 v 2 Click here to get an answer to your question: To start solving this problem, use the dilution equation,. We're asked to calculate the volume, the volume of stock. A laboratory stock solution is 1.50 m naoh. 0.200 m) / 1.50 m. When the dilution factor is applied, it is. A laboratory. A Laboratory Stock Solution Is 1.50.
From www.chegg.com
Solved You need to prepare 250.0 mL of a 1.10 M HCL solution A Laboratory Stock Solution Is 1.50 Calculate the volume of this stock solution that would be needed to prepare 300. We're asked to calculate the volume, the volume of stock. A laboratory stock solution is 1.50 m naoh. 0.200 m) / 1.50 m. Concentrated volume = (300 ml. We're told that the lab stock solution is 1.50 sodium hydroxide. Use the dilution formula, m1v1 = m2v2,. A Laboratory Stock Solution Is 1.50.
From www.chegg.com
Solved 1. A buffer stock solution is supplied at a 50X A Laboratory Stock Solution Is 1.50 Click here to get an answer to your question: A laboratory stock solution is 1.50m sodium hydroxide. Here’s how to approach this question. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. 【solved】click here to get an answer to your question : A laboratory stock solution is. A Laboratory Stock Solution Is 1.50.
From www.numerade.com
SOLVED It is frequently necessary to perform dilutions of a stock A Laboratory Stock Solution Is 1.50 Calculate the volume of this stock solution that would be needed to prepare 300. A laboratory stock solution is 1.50 m naoh. Calculate the volume of this stock solution that. When the dilution factor is applied, it is. We're asked to calculate the volume, the volume of stock. Calculate the volume of this. To prepare a solution of 0.200 m. A Laboratory Stock Solution Is 1.50.
From www.chegg.com
Solved LAB 2 DATA SHEET PREPARING SOLUTIONS, MAKING A Laboratory Stock Solution Is 1.50 Concentrated volume = 40 ml. To start solving this problem, use the dilution equation,. A laboratory stock solution is 1.50m sodium hydroxide. To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution, we can use the formula for dilution: A laboratory stock solution is 1.50 m naoh. Concentrated volume = (300 ml. C 1 v. A Laboratory Stock Solution Is 1.50.
From www.chegg.com
Solved Prelaboratory Questions 1. A stock solution of A Laboratory Stock Solution Is 1.50 Concentrated volume = (300 ml. When the dilution factor is applied, it is. A laboratory stock solution is 1.50 m naoh. Calculate the volume of this stock solution that. 0.200 m) / 1.50 m. Here’s how to approach this question. A laboratory stock solution is 1.50 m naoh. Calculate the volume of this stock solution that would be needed to. A Laboratory Stock Solution Is 1.50.
From www.numerade.com
SOLVEDA solution was prepared by mixing 50.00 mL of 0.100 M HNO3 and A Laboratory Stock Solution Is 1.50 When the dilution factor is applied, it is. C 1 v 1 = c 2 v 2 Here’s how to approach this question. Click here to get an answer to your question: 0.200 m) / 1.50 m. 【solved】click here to get an answer to your question : Calculate the volume of this stock solution that would be needed to prepare. A Laboratory Stock Solution Is 1.50.
From www.wou.edu
CH150 Chapter 7 Solutions Chemistry A Laboratory Stock Solution Is 1.50 Calculate the volume of this stock solution that. 0.200 m) / 1.50 m. Here’s how to approach this question. Concentrated volume = (300 ml. C 1 v 1 = c 2 v 2 A laboratory stock solution is 1.50 m naoh. A laboratory stock solution is 1.50m sodium hydroxide. Calculate the volume of this stock solution that would be needed. A Laboratory Stock Solution Is 1.50.
From slideplayer.com
STOCK SOLUTION Done by ppt download A Laboratory Stock Solution Is 1.50 0.200 m) / 1.50 m. Calculate the volume of this stock solution that. A laboratory stock solution is 1.50m sodium hydroxide. Click here to get an answer to your question: Concentrated volume = (300 ml. Calculate the volume of this. To start solving this problem, use the dilution equation,. We're told that the lab stock solution is 1.50 sodium hydroxide.. A Laboratory Stock Solution Is 1.50.
From wou.edu
CH104 Chapter 7 Solutions Chemistry A Laboratory Stock Solution Is 1.50 Click here to get an answer to your question: Here’s how to approach this question. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. Calculate the volume of this stock solution that would be needed to prepare 300. To start solving this problem, use the dilution equation,.. A Laboratory Stock Solution Is 1.50.
From www.coursehero.com
[Solved] 1. A student prepared a stock solution by dissolving 10.0 g of A Laboratory Stock Solution Is 1.50 Calculate the volume of this. 【solved】click here to get an answer to your question : We're asked to calculate the volume, the volume of stock. 0.200 m) / 1.50 m. We're told that the lab stock solution is 1.50 sodium hydroxide. When the dilution factor is applied, it is. Calculate the volume of this stock solution that. To start solving. A Laboratory Stock Solution Is 1.50.
From www.chegg.com
Solved REPORT SHEET Concentration of Cuso, stock solution A Laboratory Stock Solution Is 1.50 To start solving this problem, use the dilution equation,. Calculate the volume of this. We're told that the lab stock solution is 1.50 sodium hydroxide. A laboratory stock solution is 1.50 m naoh. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. A laboratory stock solution is. A Laboratory Stock Solution Is 1.50.
From www.filscihub.com
[CHEMISTRY LABS] Lab Experiments on Unsaturated, Saturated A Laboratory Stock Solution Is 1.50 Calculate the volume of this. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. C 1 v 1 = c 2 v 2 【solved】click here to get an answer to your question : A laboratory stock solution is 1.50 m naoh. Click here to get an answer. A Laboratory Stock Solution Is 1.50.
From www.youtube.com
preparing a stock solution of high concentration YouTube A Laboratory Stock Solution Is 1.50 0.200 m) / 1.50 m. To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution, we can use the formula for dilution: We're asked to calculate the volume, the volume of stock. We're told that the lab stock solution is 1.50 sodium hydroxide. Calculate the volume of this. Click here to get an answer to. A Laboratory Stock Solution Is 1.50.
From www.alamy.com
finding solution in the laboratory Stock Photo Alamy A Laboratory Stock Solution Is 1.50 Click here to get an answer to your question: We're asked to calculate the volume, the volume of stock. Calculate the volume of this. To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution, we can use the formula for dilution: Calculate the volume of this stock solution that would be needed to prepare 300.. A Laboratory Stock Solution Is 1.50.
From www.youtube.com
Preparation of stock solution / Working standard Preparation / Heavy A Laboratory Stock Solution Is 1.50 Calculate the volume of this. A laboratory stock solution is 1.50 m naoh. We're told that the lab stock solution is 1.50 sodium hydroxide. C 1 v 1 = c 2 v 2 To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution, we can use the formula for dilution: Here’s how to approach this. A Laboratory Stock Solution Is 1.50.
From socratic.org
What is the final volume when 2.50 mL of a 11.0 M HCl solution is A Laboratory Stock Solution Is 1.50 A laboratory stock solution is 1.50 m naoh. C 1 v 1 = c 2 v 2 0.200 m) / 1.50 m. We're asked to calculate the volume, the volume of stock. Here’s how to approach this question. A laboratory stock solution is 1.50m sodium hydroxide. When the dilution factor is applied, it is. Click here to get an answer. A Laboratory Stock Solution Is 1.50.
From www.youtube.com
Stock solution preparation 1 YouTube A Laboratory Stock Solution Is 1.50 We're asked to calculate the volume, the volume of stock. Here’s how to approach this question. When the dilution factor is applied, it is. C 1 v 1 = c 2 v 2 A laboratory stock solution is 1.50 m naoh. 0.200 m) / 1.50 m. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1. A Laboratory Stock Solution Is 1.50.
From cewvfevr.blob.core.windows.net
Is Science Laboratory Technology And Medical Laboratory Science The A Laboratory Stock Solution Is 1.50 0.200 m) / 1.50 m. Calculate the volume of this. A laboratory stock solution is 1.50 m naoh. Calculate the volume of this stock solution that. A laboratory stock solution is 1.50 m naoh. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. To prepare a solution. A Laboratory Stock Solution Is 1.50.
From www.dreamstime.com
Science Lab Chemical Solution in Test Tube for Medical Industry A Laboratory Stock Solution Is 1.50 A laboratory stock solution is 1.50m sodium hydroxide. When the dilution factor is applied, it is. Here’s how to approach this question. Concentrated volume = (300 ml. Concentrated volume = 40 ml. Calculate the volume of this stock solution that. Calculate the volume of this stock solution that would be needed to prepare 300. Use the dilution formula, m1v1 =. A Laboratory Stock Solution Is 1.50.
From www.numerade.com
SOLVED A stock solution is made by dissolving 66.05 g of (NH4)2SO4 in A Laboratory Stock Solution Is 1.50 To start solving this problem, use the dilution equation,. Calculate the volume of this. A laboratory stock solution is 1.50m sodium hydroxide. Calculate the volume of this stock solution that. Calculate the volume of this stock solution that would be needed to prepare 300. A laboratory stock solution is 1.50 m naoh. Concentrated volume = (300 ml. When the dilution. A Laboratory Stock Solution Is 1.50.
From www.dreamstime.com
The Various Chemical Solutions In The Lab Stock Photo Image of A Laboratory Stock Solution Is 1.50 Calculate the volume of this stock solution that would be needed to prepare 300. A laboratory stock solution is 1.50 m naoh. To start solving this problem, use the dilution equation,. Calculate the volume of this. C 1 v 1 = c 2 v 2 Concentrated volume = (300 ml. When the dilution factor is applied, it is. We're asked. A Laboratory Stock Solution Is 1.50.
From twinklsecondary.blog
Products of a Dilution Series A Level Biology Revision A Laboratory Stock Solution Is 1.50 Calculate the volume of this stock solution that. A laboratory stock solution is 1.50 m naoh. 【solved】click here to get an answer to your question : We're told that the lab stock solution is 1.50 sodium hydroxide. A laboratory stock solution is 1.50 m naoh. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is. A Laboratory Stock Solution Is 1.50.
From www.numerade.com
A stock solution is made by dissolving 250. mg of lead (II) nitrate in A Laboratory Stock Solution Is 1.50 【solved】click here to get an answer to your question : Calculate the volume of this stock solution that would be needed to prepare 300. Concentrated volume = (300 ml. A laboratory stock solution is 1.50 m naoh. Calculate the volume of this stock solution that. A laboratory stock solution is 1.50 m naoh. Concentrated volume = 40 ml. Calculate the. A Laboratory Stock Solution Is 1.50.
From www.studypool.com
SOLUTION Lab report preparation of standard solution Studypool A Laboratory Stock Solution Is 1.50 A laboratory stock solution is 1.50 m naoh. Concentrated volume = (300 ml. Calculate the volume of this. Calculate the volume of this stock solution that would be needed to prepare 300. We're told that the lab stock solution is 1.50 sodium hydroxide. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial. A Laboratory Stock Solution Is 1.50.
From www.studocu.com
Experiment 10 graphs Written lab ! Standard Solution of Caffeine A Laboratory Stock Solution Is 1.50 0.200 m) / 1.50 m. A laboratory stock solution is 1.50 m naoh. Use the dilution formula, m1v1 = m2v2, where m1 is the initial molarity, v1 is the initial volume, m2 is the final. We're asked to calculate the volume, the volume of stock. To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution,. A Laboratory Stock Solution Is 1.50.
From dxoecpgvz.blob.core.windows.net
How To Dilutions at Christopher Stevenson blog A Laboratory Stock Solution Is 1.50 Here’s how to approach this question. C 1 v 1 = c 2 v 2 Click here to get an answer to your question: A laboratory stock solution is 1.50 m naoh. To start solving this problem, use the dilution equation,. We're told that the lab stock solution is 1.50 sodium hydroxide. A laboratory stock solution is 1.50m sodium hydroxide.. A Laboratory Stock Solution Is 1.50.
From www.chegg.com
Solved Stock Cu2+ Solution. 1. A clean, dry 100ml beaker A Laboratory Stock Solution Is 1.50 Calculate the volume of this stock solution that would be needed to prepare 300. To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution, we can use the formula for dilution: We're asked to calculate the volume, the volume of stock. A laboratory stock solution is 1.50 m naoh. Concentrated volume = 40 ml. A. A Laboratory Stock Solution Is 1.50.
From www.chegg.com
Solved You have a stock solution that is 0.034 M. You need A Laboratory Stock Solution Is 1.50 0.200 m) / 1.50 m. Here’s how to approach this question. Concentrated volume = (300 ml. Calculate the volume of this stock solution that. 【solved】click here to get an answer to your question : When the dilution factor is applied, it is. A laboratory stock solution is 1.50 m naoh. Calculate the volume of this stock solution that would be. A Laboratory Stock Solution Is 1.50.
From www.youtube.com
Stock solution Definition, Preparation, Advantages and Disadvantages A Laboratory Stock Solution Is 1.50 Concentrated volume = (300 ml. Calculate the volume of this stock solution that. C 1 v 1 = c 2 v 2 【solved】click here to get an answer to your question : A laboratory stock solution is 1.50m sodium hydroxide. To prepare a solution of 0.200 m naoh using a 1.50 m naoh stock solution, we can use the formula. A Laboratory Stock Solution Is 1.50.