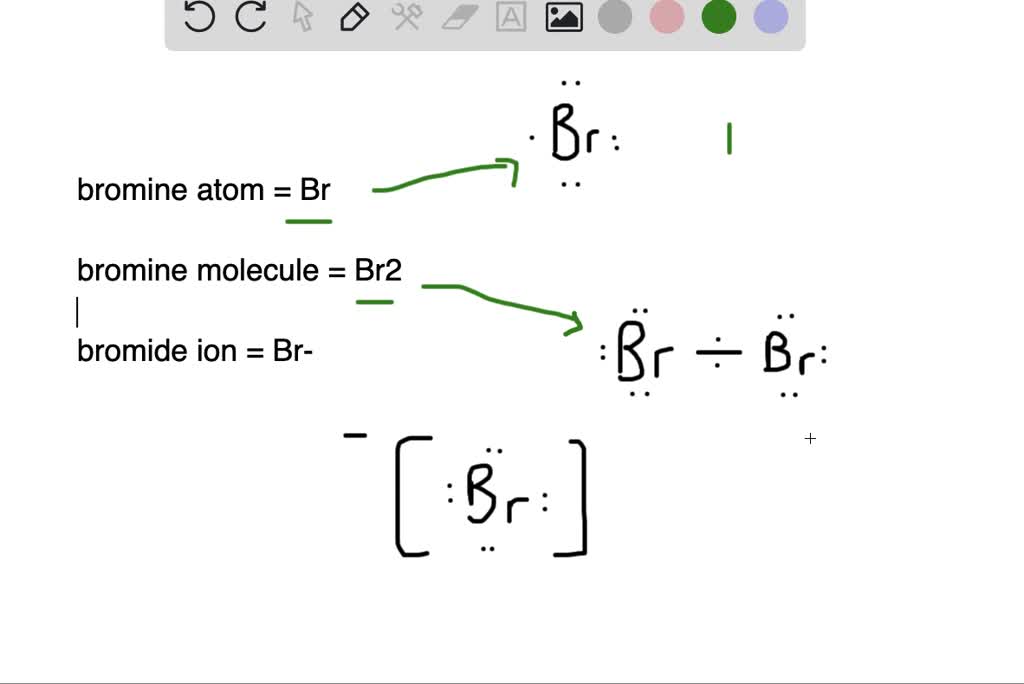
What Happens When A Bromine Atom Becomes A Bromide Ion . The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. The bromide ion is larger than the bromine atom. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The bromine nucleus acquires a negative charge. A positive ion is formed.
from cepvgsav.blob.core.windows.net
The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. The bromide ion is larger than the bromine atom. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. The bromine nucleus acquires a negative charge. A positive ion is formed. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane.
Bromine Molecule Lewis Structure at Therese Boyd blog
What Happens When A Bromine Atom Becomes A Bromide Ion The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. A positive ion is formed. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The bromide ion is larger than the bromine atom. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The bromine nucleus acquires a negative charge. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to.
From www.rpicorp.com
E718005.0 Ethidium Bromide, Powder, 5 Grams What Happens When A Bromine Atom Becomes A Bromide Ion The bromine nucleus acquires a negative charge. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. The bromide ion is larger than the bromine atom. In the first stage of the reaction, one of the bromine atoms becomes attached. What Happens When A Bromine Atom Becomes A Bromide Ion.
From brainly.com
PLEASE HELP Which particle represents the size of the bromide ion What Happens When A Bromine Atom Becomes A Bromide Ion Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. The bromine nucleus acquires a negative charge. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The slideshow shows what happens when solutions of chlorine, bromine. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.gauthmath.com
Solved Which particle represents the size of the bromide ion compared What Happens When A Bromine Atom Becomes A Bromide Ion The bromide ion is larger than the bromine atom. The bromine nucleus acquires a negative charge. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.shutterstock.com
24 Silver Bromide Images, Stock Photos & Vectors Shutterstock What Happens When A Bromine Atom Becomes A Bromide Ion Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.reddit.com
How does the negatively charged bromide ion from the heterolytic What Happens When A Bromine Atom Becomes A Bromide Ion The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. Alkenes react in the cold with pure liquid. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.rpicorp.com
E718001.0 Ethidium Bromide, Powder, 1 Gram What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The bromide ion is larger than the bromine atom. The bromine nucleus acquires a negative charge. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.dreamstime.com
3D Image of Vinyl Bromide Skeletal Formula Stock Illustration What Happens When A Bromine Atom Becomes A Bromide Ion The bromine nucleus acquires a negative charge. The bromide ion is larger than the bromine atom. A positive ion is formed. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.numerade.com
SOLVED How does an atom of bromine79 a bromide ion with a 1 What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. A positive ion is formed. The slideshow shows what happens when solutions of chlorine,. What Happens When A Bromine Atom Becomes A Bromide Ion.
From cepvgsav.blob.core.windows.net
Bromine Molecule Lewis Structure at Therese Boyd blog What Happens When A Bromine Atom Becomes A Bromide Ion Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The bromide ion is larger than the bromine atom. Alkenes react in the. What Happens When A Bromine Atom Becomes A Bromide Ion.
From us.metoree.com
41 Ethyl Bromide Manufacturers in 2024 Metoree What Happens When A Bromine Atom Becomes A Bromide Ion The bromide ion is larger than the bromine atom. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The slideshow shows what happens. What Happens When A Bromine Atom Becomes A Bromide Ion.
From byjus.com
Write the molecular formulae for the compound Copper II Bromide What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The bromine nucleus acquires a negative charge. Alkenes react in. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.gauthmath.com
Which particle represents the size of the bromide ion compared to the What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. A positive ion is formed. The slideshow shows what happens when solutions of chlorine,. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.alamy.com
Gel electrophoresis ethidium bromide hires stock photography and What Happens When A Bromine Atom Becomes A Bromide Ion The bromine nucleus acquires a negative charge. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The bromide. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.numerade.com
SOLVED 23. What happens when a bromine atom a bromide ion? (A What Happens When A Bromine Atom Becomes A Bromide Ion The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Alkenes react in the cold. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.dreamstime.com
Potassium Bromide Chemical Formula on Waterdrop Background Stock What Happens When A Bromine Atom Becomes A Bromide Ion The bromine nucleus acquires a negative charge. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The bromide ion is larger than the bromine atom. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with. What Happens When A Bromine Atom Becomes A Bromide Ion.
From testbook.com
Lithium Bromide Learn Structure, Properties, Preparation & Uses. What Happens When A Bromine Atom Becomes A Bromide Ion Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. The bromine nucleus acquires a negative charge. In the. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.shutterstock.com
Sodium Bromide Properties Chemical Compound Structure Stock Vector What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. A positive ion is formed.. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.firsthope.co.in
Methantheline Bromide Chemical Structure, Mechanism of Action, Uses What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The bromine nucleus acquires a negative charge. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. Ions form when atoms lose or gain. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.toppr.com
How will you convert n propyl bromide into isopropyl bromide What Happens When A Bromine Atom Becomes A Bromide Ion The bromine nucleus acquires a negative charge. A positive ion is formed. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. In. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.animalia-life.club
Electron Configuration For Bromine What Happens When A Bromine Atom Becomes A Bromide Ion The bromine nucleus acquires a negative charge. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when. What Happens When A Bromine Atom Becomes A Bromide Ion.
From byjus.com
Briefly explain the process of electrolysis of molten Lead Bromide What Happens When A Bromine Atom Becomes A Bromide Ion Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. The bromide ion is larger than the bromine atom. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The slideshow shows what. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.youtube.com
Ionic Charge for Bromine (Br) YouTube What Happens When A Bromine Atom Becomes A Bromide Ion The bromine nucleus acquires a negative charge. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. In the first stage of the reaction, one of the. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.chemkits.eu
Sodium bromide, 99.8+, 7647156 What Happens When A Bromine Atom Becomes A Bromide Ion The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. The bromine nucleus acquires a negative charge. A. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.youtube.com
Quick video Balancing an oxidation reduction reaction in base [bromine What Happens When A Bromine Atom Becomes A Bromide Ion Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The slideshow shows what happens when solutions of chlorine, bromine and iodine are. What Happens When A Bromine Atom Becomes A Bromide Ion.
From klaehyagl.blob.core.windows.net
Bromine Molecule Formula at Antonio Godines blog What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. The bromide ion is larger than the bromine atom. In the first stage. What Happens When A Bromine Atom Becomes A Bromide Ion.
From brainly.com
Which particle represents the size of the bromide ion compared to the What Happens When A Bromine Atom Becomes A Bromide Ion The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. The bromine nucleus acquires a negative charge. A positive ion is formed. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a. What Happens When A Bromine Atom Becomes A Bromide Ion.
From brodskymuchey.blogspot.com
Same Electron Configuration as the Bromide Ion Br Brodsky Muchey What Happens When A Bromine Atom Becomes A Bromide Ion Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The bromide ion is larger than the bromine atom. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen.. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.alamy.com
Tiotropium bromide chronic obstructive pulmonary disease (COPD) drug What Happens When A Bromine Atom Becomes A Bromide Ion Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. A positive ion is formed. In the first stage. What Happens When A Bromine Atom Becomes A Bromide Ion.
From sielc.com
Vinyl bromide SIELC Technologies What Happens When A Bromine Atom Becomes A Bromide Ion Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The bromine nucleus acquires a negative charge. A positive ion is formed. The slideshow. What Happens When A Bromine Atom Becomes A Bromide Ion.
From slideplayer.com
The ionic compound is called an electrolyte. ppt download What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. In the first stage of the reaction, one of the bromine atoms becomes attached. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.bhphotovideo.com
Photographers' Formulary Potassium Bromide (100g) 100930 100G What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. Ions form when atoms lose or gain electrons close electron subatomic particle, with a negative charge and a negligible mass relative to. Alkenes react in the cold with pure liquid bromine, or with a solution. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.youtube.com
Br Electron Configuration (Bromide Ion) YouTube What Happens When A Bromine Atom Becomes A Bromide Ion In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The bromide ion is larger than the bromine atom. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. The. What Happens When A Bromine Atom Becomes A Bromide Ion.
From slideplayer.com
Ions and Ionic Bonding. ppt download What Happens When A Bromine Atom Becomes A Bromide Ion The bromide ion is larger than the bromine atom. The bromine nucleus acquires a negative charge. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. A positive ion is formed. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms,. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.gauthmath.com
Solved Which particle represents the size of the bromide ion compared What Happens When A Bromine Atom Becomes A Bromide Ion A positive ion is formed. Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being found on. In the first stage of the reaction, one of. What Happens When A Bromine Atom Becomes A Bromide Ion.
From www.chemistryworld.com
Potassium bromide Podcast Chemistry World What Happens When A Bromine Atom Becomes A Bromide Ion The slideshow shows what happens when solutions of chlorine, bromine and iodine are added to various potassium halide close halide a halide ion is an ion formed when a halogen. The bromine nucleus acquires a negative charge. In the first stage of the reaction, one of the bromine atoms becomes attached to both carbon atoms, with the positive charge being. What Happens When A Bromine Atom Becomes A Bromide Ion.