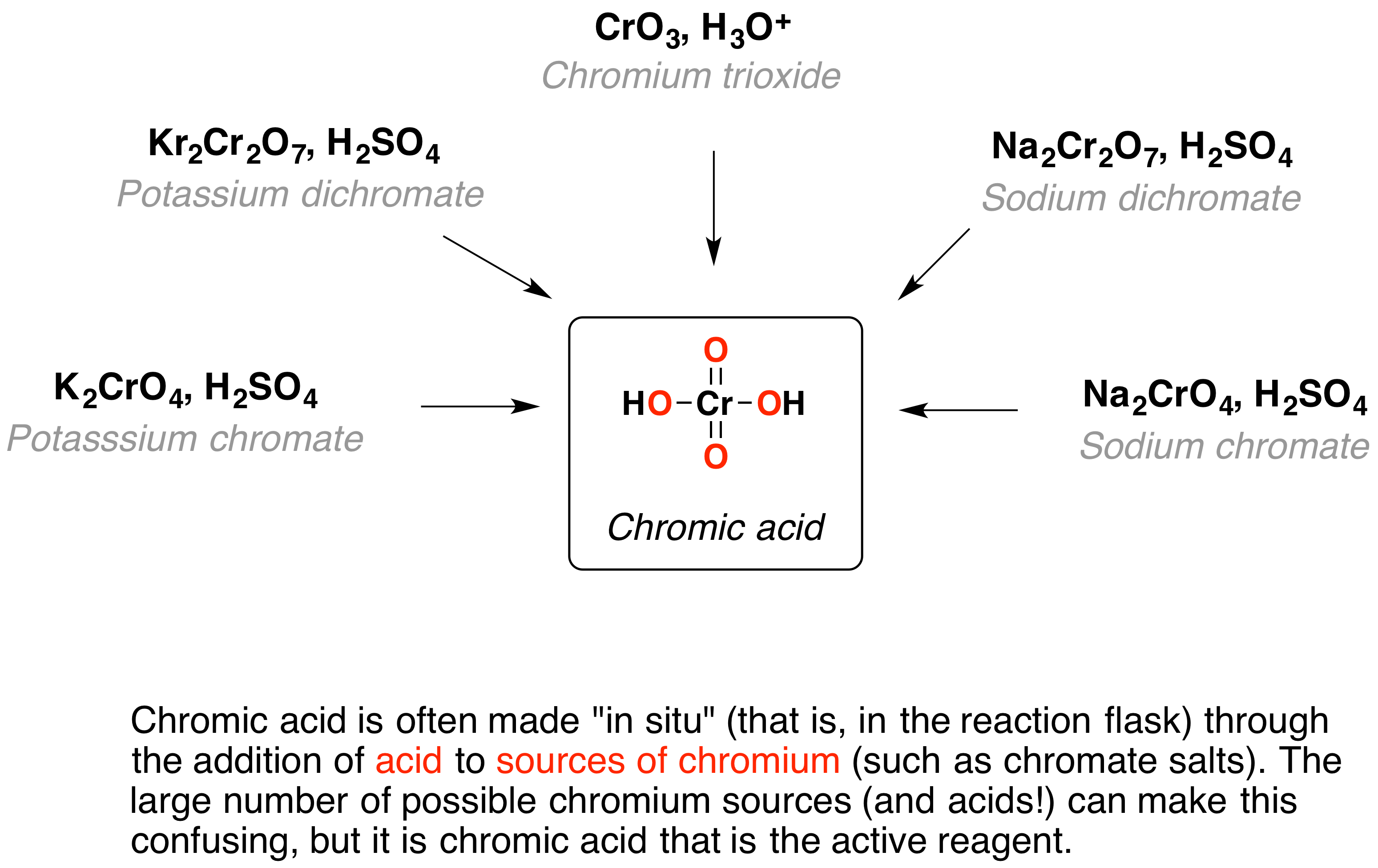
Oxidation Reagents And Catalysts In Green Chemistry . A substance that initiates or increases the. These reactions are coupled, occur together and are referred to as redox reactions. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. Waste formation caused by stoichiometric reagents,. oxidation catalysts for green chemistry. Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. selective catalysis enables an efficient utilisation of resources and the design of benign processes. creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to.
from chem.libretexts.org
the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. A substance that initiates or increases the. Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. These reactions are coupled, occur together and are referred to as redox reactions. oxidation catalysts for green chemistry. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. selective catalysis enables an efficient utilisation of resources and the design of benign processes. Waste formation caused by stoichiometric reagents,. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite.
10.6.2. Strong Oxidizing Agents Chemistry LibreTexts
Oxidation Reagents And Catalysts In Green Chemistry waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. Waste formation caused by stoichiometric reagents,. A substance that initiates or increases the. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. oxidation catalysts for green chemistry. These reactions are coupled, occur together and are referred to as redox reactions. selective catalysis enables an efficient utilisation of resources and the design of benign processes. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can.
From www.youtube.com
Green Oxidation of Cyclohexanol Experiment Part 1, Prelab YouTube Oxidation Reagents And Catalysts In Green Chemistry A substance that initiates or increases the. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is. Oxidation Reagents And Catalysts In Green Chemistry.
From www.science.org
Green, Catalytic Oxidation of Alcohols in Water Science Oxidation Reagents And Catalysts In Green Chemistry Waste formation caused by stoichiometric reagents,. creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. waste formation caused. Oxidation Reagents And Catalysts In Green Chemistry.
From www.slideserve.com
PPT New and Improved Green Experiments for the Organic Chemistry Lab Oxidation Reagents And Catalysts In Green Chemistry selective catalysis enables an efficient utilisation of resources and the design of benign processes. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. this first book to focus on catalytic processes. Oxidation Reagents And Catalysts In Green Chemistry.
From www.mdpi.com
Catalysts Free FullText General and Prospective Views on Oxidation Oxidation Reagents And Catalysts In Green Chemistry A substance that initiates or increases the. These reactions are coupled, occur together and are referred to as redox reactions. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. Waste formation caused by stoichiometric reagents,. oxidation catalysts for green chemistry. creating a green oxidation catalyst a priori is a complex. Oxidation Reagents And Catalysts In Green Chemistry.
From chemistry-reaction.com
Organic Chemistry Reagents Table « Organic Chemistry Reaction Oxidation Reagents And Catalysts In Green Chemistry selective catalysis enables an efficient utilisation of resources and the design of benign processes. These reactions are coupled, occur together and are referred to as redox reactions. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction. Oxidation Reagents And Catalysts In Green Chemistry.
From pubs.rsc.org
Oxidation of secondary alcohols using solidsupported hypervalent Oxidation Reagents And Catalysts In Green Chemistry These reactions are coupled, occur together and are referred to as redox reactions. creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. A substance that initiates or increases the. Oxidation reactions involve the loss of. Oxidation Reagents And Catalysts In Green Chemistry.
From chemistrycommunity.nature.com
Green Oxidation of Indoles using halide Catalysis Nature Research Oxidation Reagents And Catalysts In Green Chemistry These reactions are coupled, occur together and are referred to as redox reactions. selective catalysis enables an efficient utilisation of resources and the design of benign processes. Waste formation caused by stoichiometric reagents,. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. A substance that initiates or increases the. Oxidation. Oxidation Reagents And Catalysts In Green Chemistry.
From www.science.org
A "Green" Route to Adipic Acid Direct Oxidation of Cyclohexenes with Oxidation Reagents And Catalysts In Green Chemistry creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. A substance that initiates or increases the. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. Waste formation. Oxidation Reagents And Catalysts In Green Chemistry.
From owlcation.com
AS Chemistry Redox Reactions and Group 2 Elements Owlcation Oxidation Reagents And Catalysts In Green Chemistry Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. Waste formation caused by stoichiometric reagents,. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. A substance that initiates or increases the. selective catalysis enables an efficient. Oxidation Reagents And Catalysts In Green Chemistry.
From www.essentialchemicalindustry.org
Catalysis in industry Oxidation Reagents And Catalysts In Green Chemistry creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. oxidation catalysts for green chemistry. this first book to focus on catalytic processes from the. Oxidation Reagents And Catalysts In Green Chemistry.
From royalsocietypublishing.org
Engineering a more sustainable world through catalysis and green Oxidation Reagents And Catalysts In Green Chemistry These reactions are coupled, occur together and are referred to as redox reactions. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. creating a green oxidation catalyst. Oxidation Reagents And Catalysts In Green Chemistry.
From pubs.acs.org
Green and Efficient IronCatalyzed Selective Oxidation of Olefins to Oxidation Reagents And Catalysts In Green Chemistry selective catalysis enables an efficient utilisation of resources and the design of benign processes. Waste formation caused by stoichiometric reagents,. A substance that initiates or increases the. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. oxidation catalysts for green chemistry. Hydrogen peroxide. Oxidation Reagents And Catalysts In Green Chemistry.
From www.studypool.com
SOLUTION Green oxidation reagents and catalyst Studypool Oxidation Reagents And Catalysts In Green Chemistry selective catalysis enables an efficient utilisation of resources and the design of benign processes. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. These reactions are coupled, occur together and are referred to as redox reactions. waste formation caused by stoichiometric reagents, for. Oxidation Reagents And Catalysts In Green Chemistry.
From pubs.rsc.org
Photochemical oxidation of benzylic primary and secondary alcohols Oxidation Reagents And Catalysts In Green Chemistry A substance that initiates or increases the. selective catalysis enables an efficient utilisation of resources and the design of benign processes. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished. Oxidation Reagents And Catalysts In Green Chemistry.
From pubs.acs.org
Aerobic Alcohol Oxidation Using a Copper(I)/TEMPO Catalyst System A Oxidation Reagents And Catalysts In Green Chemistry These reactions are coupled, occur together and are referred to as redox reactions. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. selective catalysis enables an efficient utilisation of resources and the design of benign processes. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction. Oxidation Reagents And Catalysts In Green Chemistry.
From chem.libretexts.org
10.6.2. Strong Oxidizing Agents Chemistry LibreTexts Oxidation Reagents And Catalysts In Green Chemistry Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. waste formation caused by stoichiometric reagents, for instance in reduction. Oxidation Reagents And Catalysts In Green Chemistry.
From chem.libretexts.org
10.6.2. Strong Oxidizing Agents Chemistry LibreTexts Oxidation Reagents And Catalysts In Green Chemistry the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. selective catalysis enables an efficient utilisation of resources and the design of benign processes. These reactions are coupled, occur together and are referred to as redox reactions. A substance that initiates or increases the. Oxidation. Oxidation Reagents And Catalysts In Green Chemistry.
From www.researchgate.net
(PDF) Threedimensional graphene oxide A promising green and Oxidation Reagents And Catalysts In Green Chemistry this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. oxidation catalysts for green chemistry. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who. Oxidation Reagents And Catalysts In Green Chemistry.
From www.pinterest.com
Future Trends in Green Chemistry Future Trends in Green Chemistry Oxidation Reagents And Catalysts In Green Chemistry the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. Waste formation caused by stoichiometric reagents,. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. selective catalysis enables an efficient utilisation of resources and the design of benign processes.. Oxidation Reagents And Catalysts In Green Chemistry.
From www.chemistrystudent.com
Catalysts (ALevel) ChemistryStudent Oxidation Reagents And Catalysts In Green Chemistry creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. A substance that initiates or increases the. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. this first book to focus on catalytic processes from. Oxidation Reagents And Catalysts In Green Chemistry.
From www.nettinc.com
What Is a Diesel Oxidation Catalyst? Nett Technologies Oxidation Reagents And Catalysts In Green Chemistry this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. Waste formation caused by stoichiometric reagents,. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. Hydrogen peroxide is an. Oxidation Reagents And Catalysts In Green Chemistry.
From www.researchgate.net
An environmental catalytic process Advanced oxidation of organic Oxidation Reagents And Catalysts In Green Chemistry oxidation catalysts for green chemistry. These reactions are coupled, occur together and are referred to as redox reactions. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. creating a green oxidation catalyst a priori is. Oxidation Reagents And Catalysts In Green Chemistry.
From www.studypool.com
SOLUTION Green oxidation reagents and catalyst Studypool Oxidation Reagents And Catalysts In Green Chemistry oxidation catalysts for green chemistry. Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. selective catalysis enables an efficient utilisation of resources and the design of benign processes. These reactions are coupled, occur together. Oxidation Reagents And Catalysts In Green Chemistry.
From pubs.rsc.org
Oxidation of a wood extractive betulin to biologically active oxo Oxidation Reagents And Catalysts In Green Chemistry A substance that initiates or increases the. creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. the summer school on green chemistry has proved to be a stepping stone in the. Oxidation Reagents And Catalysts In Green Chemistry.
From www.mdpi.com
Catalysts Free FullText General and Prospective Views on Oxidation Oxidation Reagents And Catalysts In Green Chemistry These reactions are coupled, occur together and are referred to as redox reactions. A substance that initiates or increases the. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. selective catalysis enables an efficient utilisation of resources and the design of benign processes. oxidation catalysts for green chemistry. this. Oxidation Reagents And Catalysts In Green Chemistry.
From www.researchgate.net
(PDF) Importance of Green chemistry in oxidation and reduction Oxidation Reagents And Catalysts In Green Chemistry creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. oxidation catalysts for green chemistry. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. Hydrogen peroxide is. Oxidation Reagents And Catalysts In Green Chemistry.
From chemistrytalk.org
Common Oxidizing Agents & Reducing Agents ChemTalk Oxidation Reagents And Catalysts In Green Chemistry the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. Oxidation reactions involve the. Oxidation Reagents And Catalysts In Green Chemistry.
From www.degruyter.com
Mesoporous catalysts for catalytic oxidation of volatile organic Oxidation Reagents And Catalysts In Green Chemistry Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. creating a green oxidation catalyst a priori is a complex. Oxidation Reagents And Catalysts In Green Chemistry.
From www.researchgate.net
(PDF) Chemistry and Applications of IronTAML Catalysts in Green Oxidation Reagents And Catalysts In Green Chemistry Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. selective catalysis enables an efficient utilisation of resources and the design of benign processes. These reactions are coupled, occur together and are referred to. Oxidation Reagents And Catalysts In Green Chemistry.
From www.researchgate.net
Comparison between various oxidizing agents\KBr systems for the Oxidation Reagents And Catalysts In Green Chemistry waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. selective catalysis enables an efficient utilisation of resources and the design of benign processes. Hydrogen peroxide is an example of an oxidizing agent that will remove. Oxidation Reagents And Catalysts In Green Chemistry.
From chemistry-reaction.com
Organic oxidation reactions « Organic Chemistry Reaction Oxidation Reagents And Catalysts In Green Chemistry Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. oxidation catalysts for green chemistry. this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. the summer school on green chemistry. Oxidation Reagents And Catalysts In Green Chemistry.
From www.masterorganicchemistry.com
Oxidation and Reduction in Organic Chemistry Master Organic Chemistry Oxidation Reagents And Catalysts In Green Chemistry Waste formation caused by stoichiometric reagents,. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. selective catalysis enables an efficient utilisation of resources and the design of benign processes. A substance that initiates or increases the. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can.. Oxidation Reagents And Catalysts In Green Chemistry.
From www.masterorganicchemistry.com
Alcohol Oxidation "Strong" & "Weak" Oxidants Master Organic Chemistry Oxidation Reagents And Catalysts In Green Chemistry this first book to focus on catalytic processes from the viewpoint of green chemistry presents every important. These reactions are coupled, occur together and are referred to as redox reactions. Hydrogen peroxide is an example of an oxidizing agent that will remove hydrogen atoms. creating a green oxidation catalyst a priori is a complex task because every aspect. Oxidation Reagents And Catalysts In Green Chemistry.
From phys.org
Green catalysts with Earthabundant metals accelerate production of bio Oxidation Reagents And Catalysts In Green Chemistry These reactions are coupled, occur together and are referred to as redox reactions. the summer school on green chemistry has proved to be a stepping stone in the careers of many young researchers who wished to. Oxidation reactions involve the loss of electrons or the addition of oxygen and reduction is the opposite. waste formation caused by stoichiometric. Oxidation Reagents And Catalysts In Green Chemistry.
From www.chemistrysteps.com
Alcohol Oxidation Mechanisms and Practice Problems Chemistry Steps Oxidation Reagents And Catalysts In Green Chemistry A substance that initiates or increases the. creating a green oxidation catalyst a priori is a complex task because every aspect of the catalyst needs. These reactions are coupled, occur together and are referred to as redox reactions. waste formation caused by stoichiometric reagents, for instance in reduction or oxidation reactions, can. Hydrogen peroxide is an example of. Oxidation Reagents And Catalysts In Green Chemistry.