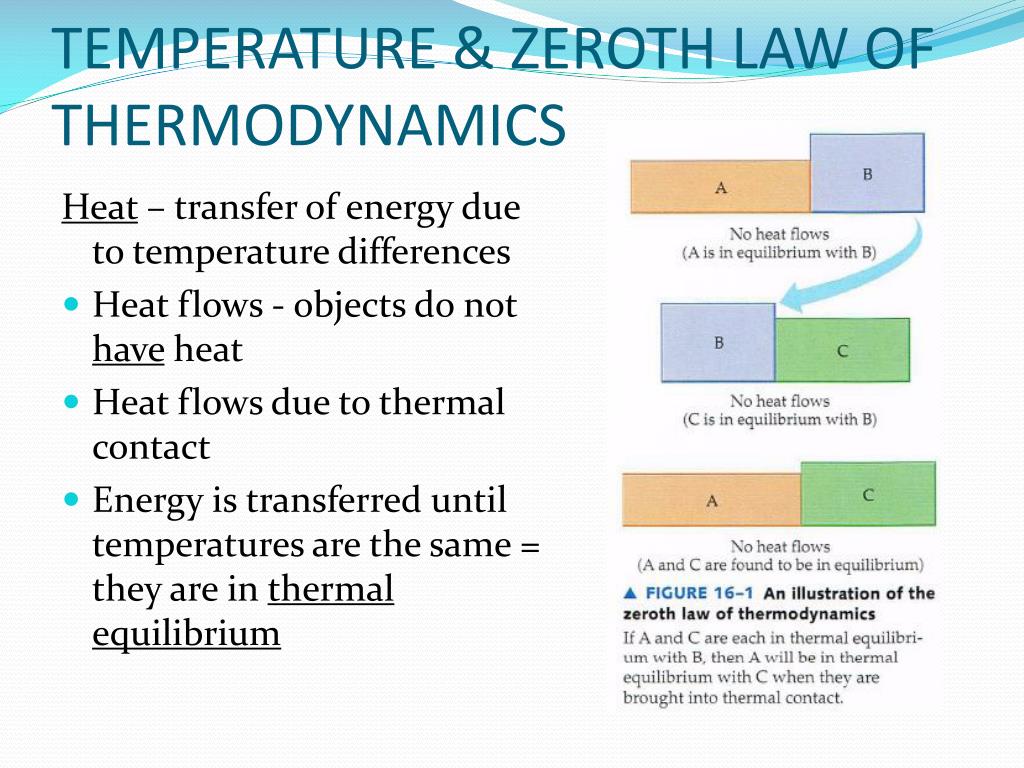
State Zeroth Law Of Thermodynamics . The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in. Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. What is the zeroth law of thermodynamics? The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. If two systems, a and b, are. Energy can neither be created nor destroyed in a chemical reaction. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal.
from www.slideserve.com
The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. If two systems, a and b, are. Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. What is the zeroth law of thermodynamics? Energy can neither be created nor destroyed in a chemical reaction. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in.
PPT TEMPERATURE & ZEROTH LAW OF THERMODYNAMICS PowerPoint Presentation ID5361849
State Zeroth Law Of Thermodynamics Energy can neither be created nor destroyed in a chemical reaction. Energy can neither be created nor destroyed in a chemical reaction. If two systems, a and b, are. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. What is the zeroth law of thermodynamics? Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other.
From www.youtube.com
Zeroth Law of Thermodynamics and Temperature measurement YouTube State Zeroth Law Of Thermodynamics Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. If two systems, a and b, are. Energy can neither be created nor destroyed in a chemical reaction. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in. State Zeroth Law Of Thermodynamics.
From pediaa.com
Difference Between First and Second Law of Thermodynamics State Zeroth Law Of Thermodynamics Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if. State Zeroth Law Of Thermodynamics.
From www.studypool.com
SOLUTION Zeroth Law of Thermodynamics Presentation Studypool State Zeroth Law Of Thermodynamics What is the zeroth law of thermodynamics? Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. If two systems, a and b, are. Energy can neither be created nor destroyed in a chemical reaction. The zeroth. State Zeroth Law Of Thermodynamics.
From www.youtube.com
Thermodynamics Equilibrium Zeroth Law of thermodynamics Steady state vs. equilibrium state State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. The zeroth law of thermodynamics is one of. State Zeroth Law Of Thermodynamics.
From slideplayer.com
The “Zeroth Law” of Thermodynamics ppt download State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in. If two systems, a and b, are. Learn. State Zeroth Law Of Thermodynamics.
From www.youtube.com
Zeroth Law of Thermodynamics YouTube State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. What is the zeroth law of thermodynamics? Thermodynamics. State Zeroth Law Of Thermodynamics.
From www.adda247.com
Zeroth Law of Thermodynamics, Equation, Formula, Notes PDF for NEET State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in. What is the zeroth law of thermodynamics? Energy can neither be created nor destroyed in a chemical reaction. If two systems, a and b, are. The zeroth law of. State Zeroth Law Of Thermodynamics.
From www.studypool.com
SOLUTION Zeroth Law of Thermodynamics Presentation Studypool State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. The zeroth law of thermodynamics is one of. State Zeroth Law Of Thermodynamics.
From ar.inspiredpencil.com
Zeroth Law Of Thermodynamics Application State Zeroth Law Of Thermodynamics Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. The zeroth law of thermodynamics is. State Zeroth Law Of Thermodynamics.
From www.slideserve.com
PPT Thermal Physics (Thermodynamics) PowerPoint Presentation ID421070 State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. If two systems, a and b, are. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in. Thermodynamics. State Zeroth Law Of Thermodynamics.
From www.slideserve.com
PPT Zeroth Law of Thermodynamics PowerPoint Presentation, free download ID4959841 State Zeroth Law Of Thermodynamics What is the zeroth law of thermodynamics? Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. If two systems, a and b, are. Energy can neither be created nor destroyed in a chemical reaction. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. The zeroth. State Zeroth Law Of Thermodynamics.
From physicsinmyview.com
Zeroth Law Of Thermodynamics Definition, History And Examples Physics In My View State Zeroth Law Of Thermodynamics Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. If two systems, a and b, are. Energy can neither be created nor destroyed in a chemical reaction. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each. State Zeroth Law Of Thermodynamics.
From www.slideserve.com
PPT TEMPERATURE & ZEROTH LAW OF THERMODYNAMICS PowerPoint Presentation ID5361849 State Zeroth Law Of Thermodynamics What is the zeroth law of thermodynamics? The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. The. State Zeroth Law Of Thermodynamics.
From www.geeksforgeeks.org
Zeroth Law of Thermodynamics Definition, Application & FAQs State Zeroth Law Of Thermodynamics Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in. The zeroth law of thermodynamics states that if. State Zeroth Law Of Thermodynamics.
From www.youtube.com
Zeroth Law of Thermodynamics Lecture 4 YouTube State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. Thermodynamics is the study of how heat moves. State Zeroth Law Of Thermodynamics.
From www.youtube.com
Physics 5.2.1 The Zeroth Law of Thermodynamics. YouTube State Zeroth Law Of Thermodynamics What is the zeroth law of thermodynamics? If two systems, a and b, are. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. Thermodynamics is the study. State Zeroth Law Of Thermodynamics.
From www.linkedin.com
Zeroth Law of Thermodynamics State Zeroth Law Of Thermodynamics Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. What is the zeroth law of thermodynamics? Energy can neither be created nor destroyed in a chemical reaction. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each. State Zeroth Law Of Thermodynamics.
From lawofthermodynamicsinfo.com
4 Laws Of Thermodynamics With Examples (Very Simple) State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. What is the zeroth law of thermodynamics? If. State Zeroth Law Of Thermodynamics.
From learnbin.net
The Zeroth Law Of Thermodynamics The Concept Of Temperature Learnbin State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. If two systems, a and b,. State Zeroth Law Of Thermodynamics.
From scienceinfo.com
Zeroth Law of Thermodynamics with Application and Examples State Zeroth Law Of Thermodynamics What is the zeroth law of thermodynamics? The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. Energy can neither be created nor destroyed in a chemical reaction. Learn the definition and implications of the zeroth law of thermodynamics,. State Zeroth Law Of Thermodynamics.
From www.youtube.com
Zeroth Law of Thermodynamics YouTube State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in. What is the zeroth law of thermodynamics? Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. Energy. State Zeroth Law Of Thermodynamics.
From sciencenotes.org
Laws of Thermodynamics State Zeroth Law Of Thermodynamics Energy can neither be created nor destroyed in a chemical reaction. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. Learn the definition and implications of. State Zeroth Law Of Thermodynamics.
From www.eigenplus.com
Laws of Thermodynamics Definition & Examples eigenplus State Zeroth Law Of Thermodynamics What is the zeroth law of thermodynamics? The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal. State Zeroth Law Of Thermodynamics.
From www.youtube.com
ZEROTH LAW OF THERMODYNAMICS _ PART 01 YouTube State Zeroth Law Of Thermodynamics What is the zeroth law of thermodynamics? The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal. State Zeroth Law Of Thermodynamics.
From chem.libretexts.org
1.2 The First Law of Thermodynamics Chemistry LibreTexts State Zeroth Law Of Thermodynamics Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. If two systems, a and b, are. What. State Zeroth Law Of Thermodynamics.
From ar.inspiredpencil.com
Zeroth Law Of Thermodynamics State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. If two systems, a and b, are. Energy can neither be created nor destroyed in a chemical reaction. Thermodynamics is the study of how heat moves around in ‘macroscopic’. State Zeroth Law Of Thermodynamics.
From www.chegg.com
Solved 1. The zeroth law of thermodynamics states that A) State Zeroth Law Of Thermodynamics Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. Energy can neither be created nor destroyed in a chemical reaction. What is the zeroth law of thermodynamics? Learn the definition and implications of the zeroth law. State Zeroth Law Of Thermodynamics.
From www.slideserve.com
PPT Zeroth Law of Thermodynamics PowerPoint Presentation, free download ID4959841 State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with a third body c, then they are in thermal equilibrium with each other. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. If two systems, a and b, are. The. State Zeroth Law Of Thermodynamics.
From ar.inspiredpencil.com
Zeroth Law Of Thermodynamics State Zeroth Law Of Thermodynamics Energy can neither be created nor destroyed in a chemical reaction. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. If two systems, a and b, are. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same.. State Zeroth Law Of Thermodynamics.
From ar.inspiredpencil.com
Zeroth Law Of Thermodynamics State Zeroth Law Of Thermodynamics What is the zeroth law of thermodynamics? If two systems, a and b, are. Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. The zeroth law of thermodynamics is one of the four laws of thermodynamics,. State Zeroth Law Of Thermodynamics.
From www.youtube.com
Zeroth law of Thermodynamics, temperature and its measurement YouTube State Zeroth Law Of Thermodynamics The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. The zeroth law of thermodynamics states that if two bodies a and b are in thermal equilibrium with. State Zeroth Law Of Thermodynamics.
From en.ppt-online.org
Revision of Thermodynamic Concepts S online presentation State Zeroth Law Of Thermodynamics Energy can neither be created nor destroyed in a chemical reaction. Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. The zeroth law of thermodynamics states that if two bodies a and b are in thermal. State Zeroth Law Of Thermodynamics.
From ar.inspiredpencil.com
Zeroth Law Of Thermodynamics State Zeroth Law Of Thermodynamics Learn the definition and implications of the zeroth law of thermodynamics, which states that systems in thermal equilibrium have the same. The zeroth law of thermodynamics states that if thermal system a is in thermal equilibrium with thermal system b, and thermal. Energy can neither be created nor destroyed in a chemical reaction. The zeroth law of thermodynamics is one. State Zeroth Law Of Thermodynamics.
From www.studocu.com
Chapter 5 Laws of thermodynamics, their consequences, zeroth law and the equation of state for State Zeroth Law Of Thermodynamics Energy can neither be created nor destroyed in a chemical reaction. What is the zeroth law of thermodynamics? The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in. Learn the definition and implications of the zeroth law of thermodynamics,. State Zeroth Law Of Thermodynamics.
From ar.inspiredpencil.com
Zeroth Law Of Thermodynamics State Zeroth Law Of Thermodynamics Energy can neither be created nor destroyed in a chemical reaction. Thermodynamics is the study of how heat moves around in ‘macroscopic’ objects. The zeroth law of thermodynamics is one of the four laws of thermodynamics, which states that if two systems are in thermal equilibrium with a third system, then they are in. What is the zeroth law of. State Zeroth Law Of Thermodynamics.