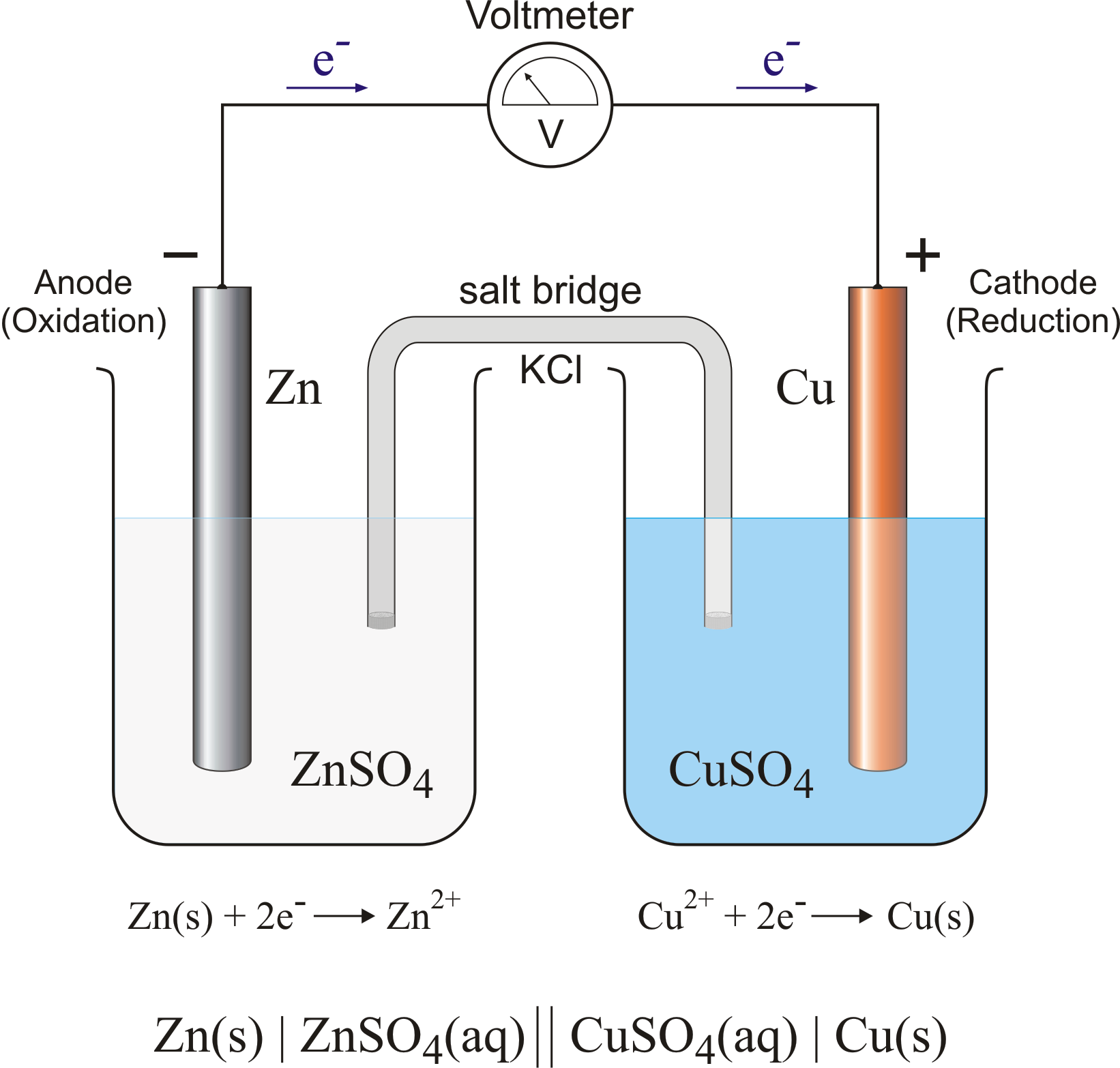
A Standard Galvanic Cell Is Constructed In Which . A galvanic cell is an example of how to use simple. Electrons flow from the anode to the cathode: A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. One beaker contains a strip of gallium metal immersed in a 1 m. Left to right in the standard galvanic cell in the figure. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. It is used to supply electrical current through a redox reaction to the transfer of electrons. The cell is comprised of two half. The electrochemical cell type is a galvanic cell.
from glossary.periodni.com
Electrons flow from the anode to the cathode: A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell is an example of how to use simple. The cell is comprised of two half. One beaker contains a strip of gallium metal immersed in a 1 m. Left to right in the standard galvanic cell in the figure. The electrochemical cell type is a galvanic cell. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below.
Galvanic Chemistry Dictionary & Glossary
A Standard Galvanic Cell Is Constructed In Which One beaker contains a strip of gallium metal immersed in a 1 m. One beaker contains a strip of gallium metal immersed in a 1 m. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. The cell is comprised of two half. It is used to supply electrical current through a redox reaction to the transfer of electrons. The electrochemical cell type is a galvanic cell. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. A galvanic cell is an example of how to use simple. Left to right in the standard galvanic cell in the figure. Electrons flow from the anode to the cathode:
From www.chegg.com
Solved A standard galvanic cell is constructed with Co²+ A Standard Galvanic Cell Is Constructed In Which The electrochemical cell type is a galvanic cell. Left to right in the standard galvanic cell in the figure. Electrons flow from the anode to the cathode: A galvanic cell is an example of how to use simple. The cell is comprised of two half. It is used to supply electrical current through a redox reaction to the transfer of. A Standard Galvanic Cell Is Constructed In Which.
From www.geeksforgeeks.org
Galvanic Cell Definition, Construction, Working Principle A Standard Galvanic Cell Is Constructed In Which A galvanic cell is an example of how to use simple. The electrochemical cell type is a galvanic cell. It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. One beaker contains. A Standard Galvanic Cell Is Constructed In Which.
From chem.libretexts.org
Chapter 19.1 Describing Electrochemical Cells Chemistry LibreTexts A Standard Galvanic Cell Is Constructed In Which Electrons flow from the anode to the cathode: A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. One beaker contains a strip of gallium metal immersed in a 1. A Standard Galvanic Cell Is Constructed In Which.
From glossary.periodni.com
Galvanic Chemistry Dictionary & Glossary A Standard Galvanic Cell Is Constructed In Which Electrons flow from the anode to the cathode: A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. Left to right in the standard galvanic cell in the figure. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. One. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed with Cu2+ Cu A Standard Galvanic Cell Is Constructed In Which One beaker contains a strip of gallium metal immersed in a 1 m. The electrochemical cell type is a galvanic cell. The cell is comprised of two half. Electrons flow from the anode to the cathode: A galvanic cell is an example of how to use simple. Left to right in the standard galvanic cell in the figure. A galvanic. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which It is used to supply electrical current through a redox reaction to the transfer of electrons. Electrons flow from the anode to the cathode: The cell is comprised of two half. The electrochemical cell type is a galvanic cell. A galvanic cell is an example of how to use simple. A galvanic cell with a measured standard cell potential of. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved question. A standard galvanic cell is constructed A Standard Galvanic Cell Is Constructed In Which A galvanic cell is an example of how to use simple. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. The electrochemical cell type is a galvanic cell. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. Electrons. A Standard Galvanic Cell Is Constructed In Which.
From courses.lumenlearning.com
Galvanic Cells General Chemistry A Standard Galvanic Cell Is Constructed In Which It is used to supply electrical current through a redox reaction to the transfer of electrons. The electrochemical cell type is a galvanic cell. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. One beaker contains a strip of gallium metal immersed in a 1 m. A. A Standard Galvanic Cell Is Constructed In Which.
From psu.pb.unizin.org
Galvanic Cells (17.2) Chemistry 110 A Standard Galvanic Cell Is Constructed In Which It is used to supply electrical current through a redox reaction to the transfer of electrons. Left to right in the standard galvanic cell in the figure. Electrons flow from the anode to the cathode: One beaker contains a strip of gallium metal immersed in a 1 m. A galvanic cell with a measured standard cell potential of 0.27 v. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed with Cr3+ A Standard Galvanic Cell Is Constructed In Which A galvanic cell is an example of how to use simple. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. The electrochemical cell type is a galvanic cell. The cell is comprised of two half. Electrons flow from the anode to the cathode: One beaker contains a. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which One beaker contains a strip of gallium metal immersed in a 1 m. Electrons flow from the anode to the cathode: It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. Left to right in the. A Standard Galvanic Cell Is Constructed In Which.
From www.answersarena.com
[Solved] A standard galvanic cell is constructed in which A Standard Galvanic Cell Is Constructed In Which A galvanic cell is an example of how to use simple. Electrons flow from the anode to the cathode: A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. Left to right in the standard galvanic cell in the figure. One beaker contains a strip of gallium metal. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which The cell is comprised of two half. One beaker contains a strip of gallium metal immersed in a 1 m. Electrons flow from the anode to the cathode: A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. A galvanic cell based on the spontaneous reaction between copper. A Standard Galvanic Cell Is Constructed In Which.
From www.scienceabc.com
Galvanic Cell Definition, Diagram And Working A Standard Galvanic Cell Is Constructed In Which A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. A galvanic cell is an example of how to use simple. Left to right in the standard galvanic cell in the figure. Electrons flow from the anode to the cathode: It is used to supply electrical current through. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. The electrochemical cell type is a galvanic cell. It is used to supply electrical current through a redox reaction to the transfer of electrons. One beaker contains a strip of gallium metal immersed in a 1 m. A. A Standard Galvanic Cell Is Constructed In Which.
From www.scienceabc.com
Galvanic Cell Definition, Diagram And Working A Standard Galvanic Cell Is Constructed In Which The electrochemical cell type is a galvanic cell. Electrons flow from the anode to the cathode: A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. One beaker contains a strip of gallium metal immersed in a 1 m. It is used to supply electrical current through a redox reaction to the. A Standard Galvanic Cell Is Constructed In Which.
From www.numerade.com
SOLVED A standard galvanic cell is constructed in which a Ni2+ Ni A Standard Galvanic Cell Is Constructed In Which Electrons flow from the anode to the cathode: A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. One beaker contains a strip of gallium metal immersed in a 1 m. The electrochemical cell type is a galvanic cell. It is used to supply electrical current through a redox reaction to the. A Standard Galvanic Cell Is Constructed In Which.
From solvedlib.com
A standard galvanic cell is constructed with Mg2+ Mg … SolvedLib A Standard Galvanic Cell Is Constructed In Which One beaker contains a strip of gallium metal immersed in a 1 m. Left to right in the standard galvanic cell in the figure. The cell is comprised of two half. It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell with a measured standard cell potential of 0.27 v is. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed with A Standard Galvanic Cell Is Constructed In Which The electrochemical cell type is a galvanic cell. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. The cell is comprised of two half. A galvanic cell is an example of how to use simple. One beaker contains a strip of gallium metal immersed in a 1. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which One beaker contains a strip of gallium metal immersed in a 1 m. The electrochemical cell type is a galvanic cell. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which A galvanic cell is an example of how to use simple. It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. Left to right in the standard galvanic cell in the figure. Electrons flow from the. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed with 12]I and A Standard Galvanic Cell Is Constructed In Which A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. It is used to supply electrical current through a redox reaction to the transfer of electrons. Electrons flow from the anode to the cathode: A galvanic cell is an example of how to use simple. A galvanic cell with a measured standard. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed from zinc A Standard Galvanic Cell Is Constructed In Which A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. The cell is comprised of two half. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. The electrochemical cell type is a galvanic cell. Electrons flow from the anode. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which The cell is comprised of two half. A galvanic cell is an example of how to use simple. It is used to supply electrical current through a redox reaction to the transfer of electrons. One beaker contains a strip of gallium metal immersed in a 1 m. A galvanic cell based on the spontaneous reaction between copper and silver(i) is. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. Left to right in. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which The electrochemical cell type is a galvanic cell. The cell is comprised of two half. Left to right in the standard galvanic cell in the figure. It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell is an example of how to use simple. A galvanic cell based on the spontaneous. A Standard Galvanic Cell Is Constructed In Which.
From www.slideserve.com
PPT Voltaic/Galvanic Cells PowerPoint Presentation, free download A Standard Galvanic Cell Is Constructed In Which One beaker contains a strip of gallium metal immersed in a 1 m. The electrochemical cell type is a galvanic cell. A galvanic cell is an example of how to use simple. Electrons flow from the anode to the cathode: It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell based. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed in which a A Standard Galvanic Cell Is Constructed In Which The electrochemical cell type is a galvanic cell. The cell is comprised of two half. It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. Electrons flow from the anode to the. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed with \\( A Standard Galvanic Cell Is Constructed In Which It is used to supply electrical current through a redox reaction to the transfer of electrons. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. Left to right in the standard galvanic cell in the figure. Electrons flow from the anode to the cathode: The cell is comprised of two half.. A Standard Galvanic Cell Is Constructed In Which.
From www.numerade.com
SOLVED (8 points) Write the balanced equation for the galvanic cell A Standard Galvanic Cell Is Constructed In Which The electrochemical cell type is a galvanic cell. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. Electrons flow from the anode to the cathode: It is used to supply electrical current through a redox reaction to the transfer of electrons. Left to right in the standard galvanic cell in the. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved A standard galvanic cell is constructed so that the A Standard Galvanic Cell Is Constructed In Which A galvanic cell is an example of how to use simple. Electrons flow from the anode to the cathode: One beaker contains a strip of gallium metal immersed in a 1 m. Left to right in the standard galvanic cell in the figure. The electrochemical cell type is a galvanic cell. It is used to supply electrical current through a. A Standard Galvanic Cell Is Constructed In Which.
From www.numerade.com
SOLVED A standard galvanic cell is constructed with Pb2+ Pb and Cu2 A Standard Galvanic Cell Is Constructed In Which Electrons flow from the anode to the cathode: The cell is comprised of two half. Left to right in the standard galvanic cell in the figure. One beaker contains a strip of gallium metal immersed in a 1 m. A galvanic cell is an example of how to use simple. The electrochemical cell type is a galvanic cell. It is. A Standard Galvanic Cell Is Constructed In Which.
From www.numerade.com
SOLVED A standard galvanic cell is constructed with Feztp FFe and A Standard Galvanic Cell Is Constructed In Which The electrochemical cell type is a galvanic cell. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. Left to right in the standard galvanic cell in the figure. One. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
Solved The cell potential for the standard galvanic cell A Standard Galvanic Cell Is Constructed In Which The cell is comprised of two half. A galvanic cell based on the spontaneous reaction between copper and silver(i) is depicted in the figure below. A galvanic cell is an example of how to use simple. A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. Electrons flow. A Standard Galvanic Cell Is Constructed In Which.
From www.chegg.com
B1. Construction of Galvanic Cells a. For each A Standard Galvanic Cell Is Constructed In Which A galvanic cell with a measured standard cell potential of 0.27 v is constructed using two beakers connected by a salt bridge. Left to right in the standard galvanic cell in the figure. A galvanic cell is an example of how to use simple. It is used to supply electrical current through a redox reaction to the transfer of electrons.. A Standard Galvanic Cell Is Constructed In Which.