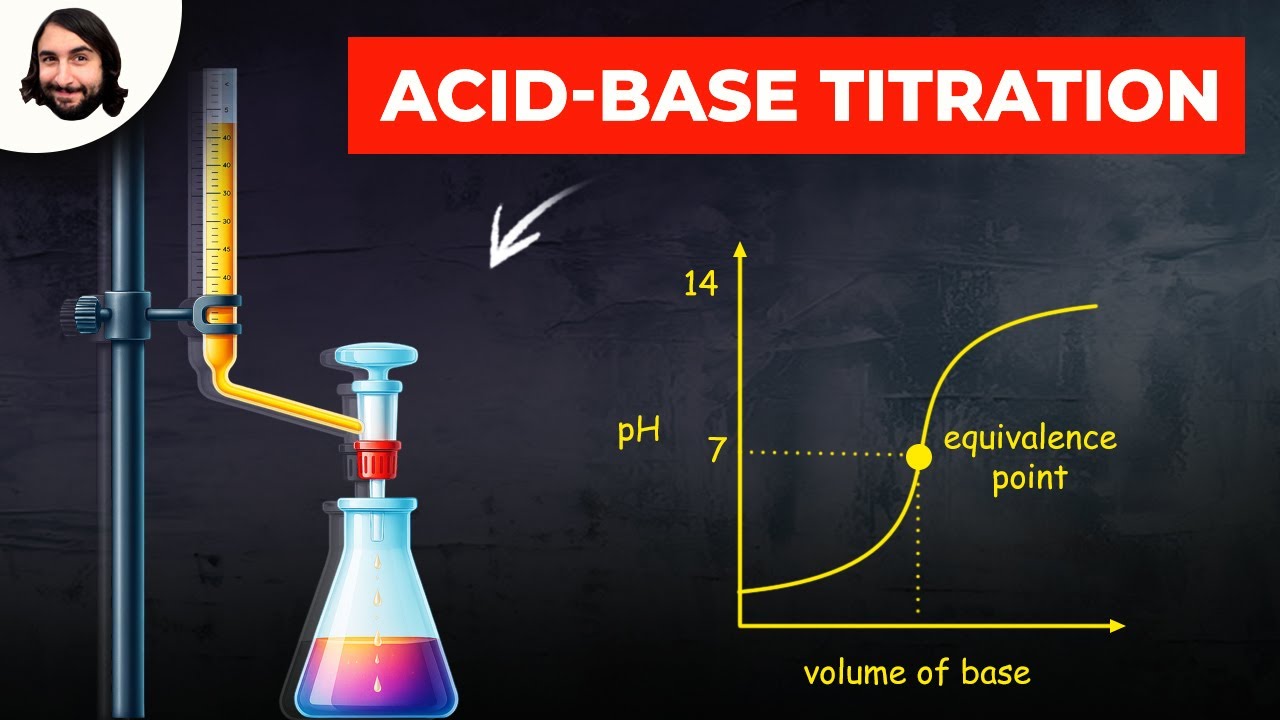
Titration Wikipedia . It is used to work out the unknown concentration of a known substance. Titration is a type of quantitative chemical analysis. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. It is a useful means of characterizing. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called.
from wikiref.net
It is used to work out the unknown concentration of a known substance. Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. It is a useful means of characterizing. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration is a type of quantitative chemical analysis. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without.
What are different types of titration? Wiki REF
Titration Wikipedia It is used to work out the unknown concentration of a known substance. Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. It is used to work out the unknown concentration of a known substance. It is a useful means of characterizing. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. Titration is a type of quantitative chemical analysis. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of.
From www.microlit.com
An Advanced Guide to Titration Microlit Titration Wikipedia A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is a type of quantitative chemical analysis. It is a. Titration Wikipedia.
From mungfali.com
Titration Steps Titration Wikipedia Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is a type of quantitative chemical analysis. Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume. Titration Wikipedia.
From mmerevise.co.uk
Titrations and Uncertainties MME Titration Wikipedia Titration is a type of quantitative chemical analysis. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. A thermometric titration is. Titration Wikipedia.
From giohqmnus.blob.core.windows.net
Titration Definition Wikipedia at James Henrich blog Titration Wikipedia Titration is a type of quantitative chemical analysis. It is used to work out the unknown concentration of a known substance. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration is. Titration Wikipedia.
From www.scribd.com
Amperometric Titration Wikipedia, The Free Encyclopedia PDF Titration Wikipedia A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. It is a useful means of characterizing. In analytical chemistry, potentiometric titration is a technique similar to direct titration. Titration Wikipedia.
From www.slideserve.com
PPT Non aqueous Titration theory and principles ppt PowerPoint Titration Wikipedia It is a useful means of characterizing. It is used to work out the unknown concentration of a known substance. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration is the process in which one solution is added to another solution such that it reacts under conditions. Titration Wikipedia.
From chem.libretexts.org
AcidBase Titrations Chemistry LibreTexts Titration Wikipedia Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration is the process in which one solution is added to another solution such that it. Titration Wikipedia.
From resource.studiaacademy.com
Acids, Alkalis and Titrations Studia Academy Resources Titration Wikipedia It is used to work out the unknown concentration of a known substance. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is a. Titration Wikipedia.
From www.slideserve.com
PPT Neutralization and Titration PowerPoint Presentation, free Titration Wikipedia It is used to work out the unknown concentration of a known substance. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is a. Titration Wikipedia.
From ar.inspiredpencil.com
Titration Titration Wikipedia It is a useful means of characterizing. It is used to work out the unknown concentration of a known substance. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is the process in which one solution is added to another. Titration Wikipedia.
From www.scribd.com
Titration Wikipedia PDF Titration Chemistry Titration Wikipedia Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. It is a useful means of characterizing. Titration is a type. Titration Wikipedia.
From www.science-revision.co.uk
Titrations Titration Wikipedia It is used to work out the unknown concentration of a known substance. It is a useful means of characterizing. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely. Titration Wikipedia.
From www.priyamstudycentre.com
Acid Base Titration Principle, Types, Process, Indicators Titration Wikipedia Titration is a type of quantitative chemical analysis. It is a useful means of characterizing. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an. Titration Wikipedia.
From www.pinterest.com
School level titration demonstration Titration Wikipedia in 2021 Titration Wikipedia Titration is a type of quantitative chemical analysis. It is used to work out the unknown concentration of a known substance. Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. It is a useful means of characterizing. Titration, process of chemical. Titration Wikipedia.
From www.pinterest.com
ITC1 Isothermal titration calorimetry Wikipedia in 2021 Titration Wikipedia Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration is the controlled mixing of a solution with known concentration. Titration Wikipedia.
From www.chemicals.co.uk
Titration Experiments In Chemistry The Chemistry Blog Titration Wikipedia It is a useful means of characterizing. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration, process of chemical analysis in which the quantity of some constituent. Titration Wikipedia.
From monomole.com
Titration overview Mono Mole Titration Wikipedia Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. It is a useful means of characterizing. Titration is a type of quantitative chemical analysis. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located. Titration Wikipedia.
From azwikihow.blogspot.com
PREPARATION OF STANDARD SOLUTIONS (TITRATION) AZ WIKI HOW Titration Wikipedia In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is the process in which one solution is added to another solution such that it. Titration Wikipedia.
From www.chegg.com
Solved If you search acid base titration on Wikipedia, the Titration Wikipedia A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. It is a. Titration Wikipedia.
From www.slideserve.com
PPT 09 Titrations & their CALCULATIONS PowerPoint Presentation ID Titration Wikipedia Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration is the controlled mixing of a solution with known concentration. Titration Wikipedia.
From wikiref.net
What are different types of titration? Wiki REF Titration Wikipedia It is a useful means of characterizing. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. In analytical chemistry, potentiometric titration is a technique similar to direct titration. Titration Wikipedia.
From mmerevise.co.uk
Acids and Bases Questions and Revision MME Titration Wikipedia Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. It is used to work out the unknown concentration of a known. Titration Wikipedia.
From www.chemicals.co.uk
What is Titration in Chemistry? The Chemistry Blog Titration Wikipedia It is used to work out the unknown concentration of a known substance. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration is a type of quantitative chemical analysis. Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added. Titration Wikipedia.
From www.slideserve.com
PPT Titrations PowerPoint Presentation, free download ID2145164 Titration Wikipedia Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration is a type of quantitative chemical analysis. Titration is the controlled mixing of a solution. Titration Wikipedia.
From chemistry-acidsandbases.weebly.com
Titrations Titration Wikipedia A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration is a type of quantitative chemical analysis. In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. Titration is the controlled mixing of a solution with known concentration (the standard solution or. Titration Wikipedia.
From www.rdworldonline.com
What are titration instruments? Research & Development World Titration Wikipedia It is a useful means of characterizing. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. Titration is a type of. Titration Wikipedia.
From edu.rsc.org
Vintage titrations sulfur dioxide concentrations in wine Resource Titration Wikipedia A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. In analytical chemistry, potentiometric titration is a technique similar to direct. Titration Wikipedia.
From general.chemistrysteps.com
Titration of a Polyprotic Acids Chemistry Steps Titration Wikipedia Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. It is a useful means of characterizing. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration, process of chemical. Titration Wikipedia.
From www.chemistryscl.com
Titrimetry, Titration Classifications, Standard solutions, Equivalence Titration Wikipedia A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. It is used to work out the unknown concentration of a known substance. Titration is the process in which. Titration Wikipedia.
From www.youtube.com
Titration of a monoprotic strong acid (HCl) and monoprotic strong base Titration Wikipedia Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. It is used to work out the unknown concentration of a known substance. A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without. In analytical chemistry, potentiometric titration is. Titration Wikipedia.
From www.ebbecke-verfahrenstechnik.de
Coulometrische KarlFischerTitration Lexikon Ebbecke Titration Wikipedia Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is the controlled mixing of a solution with known concentration (the standard solution or titrant) to another solution (called. In analytical chemistry, potentiometric titration is a technique similar to direct titration. Titration Wikipedia.
From keystagewiki.com
Titration Key Stage Wiki Titration Wikipedia In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. It is used to work out the unknown concentration of a known substance. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. Titration is the. Titration Wikipedia.
From theedge.com.hk
Chemistry How To Titration The Edge Titration Wikipedia Titration is a type of quantitative chemical analysis. It is used to work out the unknown concentration of a known substance. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. It is a useful means of characterizing. Titration is the process. Titration Wikipedia.
From www.wikiwand.com
Titration Wikiwand Titration Wikipedia In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. It is used to work out the unknown concentration of a known substance. Titration is a type of quantitative chemical analysis. Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added. Titration Wikipedia.
From www.gbu-presnenskij.ru
Drug Titration Wikipedia, 41 OFF www.gbupresnenskij.ru Titration Wikipedia Titration is the process in which one solution is added to another solution such that it reacts under conditions in which the added volume may be accurately measured. Titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of. In analytical chemistry, potentiometric. Titration Wikipedia.