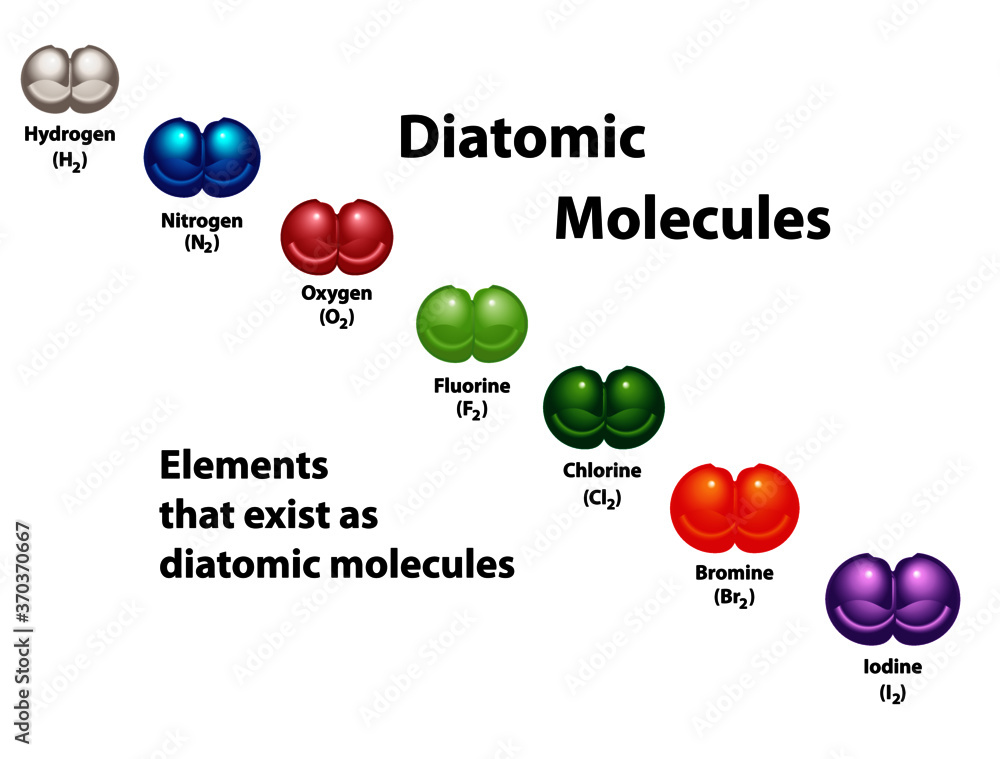
Chlorine Bromine And Iodine Are Alike In That All The Three . Learn about the halogens, a group of five elements with similar chemical properties and applications. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Find out how they are obtained from seawater,. They are all oxidizing agents and form. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Find out their properties, names, and where they are located on the. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity.
from stock.adobe.com
Find out their properties, names, and where they are located on the. Find out how they are obtained from seawater,. They are all oxidizing agents and form. Learn about the halogens, a group of five elements with similar chemical properties and applications. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens.
Vecteur Stock Diatomic molecules diagram shows elements that exist as
Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Find out how they are obtained from seawater,. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Find out their properties, names, and where they are located on the. Learn about the halogens, a group of five elements with similar chemical properties and applications. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. They are all oxidizing agents and form.
From www.sciencephoto.com
Bromine and Iodine Stock Image C002/8098 Science Photo Library Chlorine Bromine And Iodine Are Alike In That All The Three Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. They are all oxidizing agents and form. Find out their properties, names, and where they. Chlorine Bromine And Iodine Are Alike In That All The Three.
From slidetodoc.com
HALOGENS Chlorine Fluorine GROUP 7 A Bromine Iodine Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the halogens, a group of five elements with similar chemical properties and applications. Find out their properties, names, and where they are located on the. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Halogen is. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.slideserve.com
PPT Chemistry UNIT 3 PowerPoint Presentation, free download ID5906142 Chlorine Bromine And Iodine Are Alike In That All The Three Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. They are all oxidizing agents and form. Find out how they are obtained from seawater,. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine,. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.slideserve.com
PPT Chapter Twenty PowerPoint Presentation, free download ID6301153 Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. They are all oxidizing agents and form. Halogens are highly reactive nonmetallic elements in group 17 of the periodic. Chlorine Bromine And Iodine Are Alike In That All The Three.
From ecurrencythailand.com
Which Is The Least Reactive Fluorine Chlorine Bromine Iodine? 22 Most Chlorine Bromine And Iodine Are Alike In That All The Three They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Find out their properties, names, and. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.differencebetween.com
Difference Between Bromine and Chlorine Compare the Difference Chlorine Bromine And Iodine Are Alike In That All The Three Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Learn about the properties and uses of chlorine, bromine and iodine,. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.researchgate.net
Recoveries of bromine (), chlorine (), fluorine (), iodine (), and Chlorine Bromine And Iodine Are Alike In That All The Three Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Learn about the halogens, a group of five elements with similar chemical properties and applications. They are all oxidizing agents and form. Find out their properties, names, and where they are located on the. Halogens are highly reactive nonmetallic elements. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.alamy.com
halogens in gas jars Fl Cl Br I fluorine chlorine bromine iodine Stock Chlorine Bromine And Iodine Are Alike In That All The Three Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Find out how they are obtained from seawater,. Find out their properties, names, and where they are located on the. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Learn about the halogens, a group of nonmetal elements with 7 valence. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.researchgate.net
(a) Electronic properties of iodine, bromine, and chlorine; (b Chlorine Bromine And Iodine Are Alike In That All The Three They are all oxidizing agents and form. Learn about the halogens, a group of five elements with similar chemical properties and applications. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. They include. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.slideserve.com
PPT Winery Sanitation PowerPoint Presentation, free download ID6714725 Chlorine Bromine And Iodine Are Alike In That All The Three Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Find out their properties, names, and where they are located on the. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Learn about the halogens, a group of five elements with similar chemical properties and applications. They include fluorine, chlorine, bromine,. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.scribd.com
11.3 Group 7 Halogens Chlorine Bromine Iodine 2c Edexcel Igcse 9 Chlorine Bromine And Iodine Are Alike In That All The Three Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Find out how they are obtained from seawater,. Find out their properties, names, and where they are located on the. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine,. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.numerade.com
SOLVED Three acids with similar structures of the form HOX (H is Chlorine Bromine And Iodine Are Alike In That All The Three Find out how they are obtained from seawater,. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Find out their. Chlorine Bromine And Iodine Are Alike In That All The Three.
From nap.nationalacademies.org
Table of Isotopes of Fluorine, Chlorine, Bromine and Iodine The Chlorine Bromine And Iodine Are Alike In That All The Three Find out their properties, names, and where they are located on the. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.desertcart.ae
Buy Chlorine, Bromine and Iodine NMR Physico and Biological Chlorine Bromine And Iodine Are Alike In That All The Three Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Find out how they are obtained from seawater,. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. They are all oxidizing agents and form. Learn about the halogens, a group of. Chlorine Bromine And Iodine Are Alike In That All The Three.
From resource.studiaacademy.com
2.2 Group 7 (Halogens) Chlorine, Bromine and Iodine Studia Academy Chlorine Bromine And Iodine Are Alike In That All The Three Find out their properties, names, and where they are located on the. They are all oxidizing agents and form. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine,. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.alamy.com
Three conical flasks, each containing a different halogen. They are Chlorine Bromine And Iodine Are Alike In That All The Three Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Find out their properties, names, and where they are located on the. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine.. Chlorine Bromine And Iodine Are Alike In That All The Three.
From stock.adobe.com
Vecteur Stock Diatomic molecules diagram shows elements that exist as Chlorine Bromine And Iodine Are Alike In That All The Three Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Find out how they are obtained from seawater,. Find out their properties, names, and where they are located on the. They include fluorine, chlorine, bromine, iodine, astatine,. Chlorine Bromine And Iodine Are Alike In That All The Three.
From fphoto.photoshelter.com
halogens chemistry bromine chlorine iodine Fundamental Photographs Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Learn about the halogens, a group of five elements with similar chemical properties and applications. They are all oxidizing agents and form. Find out their properties, names, and where they are located on the. Halogens. Chlorine Bromine And Iodine Are Alike In That All The Three.
From joelgordon.photoshelter.com
Chlorine Bromine Iodine Joel Gordon Photography Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Find out how they are obtained from seawater,. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Find out their properties, names, and where they are located on. Chlorine Bromine And Iodine Are Alike In That All The Three.
From slideplayer.com
Nonmetals, Nobel Gases, and Metalloids ppt download Chlorine Bromine And Iodine Are Alike In That All The Three Find out their properties, names, and where they are located on the. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Learn about the halogens, a group of five elements with similar chemical properties and applications. Halogen is. Chlorine Bromine And Iodine Are Alike In That All The Three.
From studiousguy.com
Bromine (Br) Properties & Uses StudiousGuy Chlorine Bromine And Iodine Are Alike In That All The Three Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Find out their properties, names, and where they are located on the. Learn about the halogens, a group of five elements with similar chemical properties and applications. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Find out how. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.masterorganicchemistry.com
Introduction to Free Radical Substitution Reactions Master Organic Chlorine Bromine And Iodine Are Alike In That All The Three Find out their properties, names, and where they are located on the. Find out how they are obtained from seawater,. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. They are all oxidizing agents and form. Learn about the halogens, a. Chlorine Bromine And Iodine Are Alike In That All The Three.
From slideplayer.com
Intermolecular Forces ppt download Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. They are all oxidizing agents and form. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Learn about the properties and uses of. Chlorine Bromine And Iodine Are Alike In That All The Three.
From slideplayer.com
Atomic structure. ppt download Chlorine Bromine And Iodine Are Alike In That All The Three They are all oxidizing agents and form. Find out their properties, names, and where they are located on the. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Find out how they are obtained from seawater,. Learn about the halogens, a group of five elements with similar chemical properties. Chlorine Bromine And Iodine Are Alike In That All The Three.
From fphoto.photoshelter.com
science element halogen iodine bromine chlorine Fundamental Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. They are all oxidizing agents and form. Find out their properties, names, and where they are located on the. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Learn about. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.researchgate.net
Analytical scheme for radiochemical purification of chlorine, bromine Chlorine Bromine And Iodine Are Alike In That All The Three Find out how they are obtained from seawater,. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Learn about the halogens, a group of five elements with similar chemical properties and applications. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. They are all oxidizing. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.researchgate.net
(a) Electronic properties of iodine, bromine, and chlorine; (b Chlorine Bromine And Iodine Are Alike In That All The Three Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. They are all oxidizing agents and form. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Learn about the halogens, a group of five elements with similar chemical properties and applications. Halogens are highly. Chlorine Bromine And Iodine Are Alike In That All The Three.
From material-properties.org
Chlorine and Iodine Comparison Properties Material Properties Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the halogens, a group of five elements with similar chemical properties and applications. Find out how they are obtained from seawater,. They are all oxidizing agents and form. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. They. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.slideshare.net
Lecture 6.1 The Periodic Table Chlorine Bromine And Iodine Are Alike In That All The Three Find out how they are obtained from seawater,. Find out their properties, names, and where they are located on the. They are all oxidizing agents and form. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Halogens are highly reactive nonmetallic elements in group 17 of the periodic table. Learn about the halogens, a. Chlorine Bromine And Iodine Are Alike In That All The Three.
From studylib.net
Chlorine, bromine, and iodine have very similar chemical properties Chlorine Bromine And Iodine Are Alike In That All The Three Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Learn about the halogens, a group of five elements with similar chemical properties and applications. Find out how they are obtained from seawater,. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Halogens. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.numerade.com
SOLVED Fluorine, chlorine, bromine, iodine, and astatine make up group Chlorine Bromine And Iodine Are Alike In That All The Three They are all oxidizing agents and form. Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Learn about the halogens, a group of five elements with similar chemical properties and applications. Find out their properties, names, and where they. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.differencebetween.com
Difference Between Bromine and Iodine Compare the Difference Between Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. They are all oxidizing agents and form. Learn about the halogens, a group of five elements with similar chemical properties and applications. Halogen is a group of six nonmetallic elements in the periodic table, including. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.diffzy.com
Bromine vs. Chlorine What's The Difference In Tabular Form, Points Chlorine Bromine And Iodine Are Alike In That All The Three Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. Learn about the halogens, a group of five elements with similar chemical properties and applications. Find out their properties, names, and where they are located on the. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. Halogens are. Chlorine Bromine And Iodine Are Alike In That All The Three.
From www.horiba.com
Analysis of Chlorine, Bromine and Iodine in water using ICPOES HORIBA Chlorine Bromine And Iodine Are Alike In That All The Three Find out how they are obtained from seawater,. Learn about the halogens, a group of five elements with similar chemical properties and applications. Learn about the properties and uses of chlorine, bromine and iodine, the three common halogens. They are all oxidizing agents and form. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high. Chlorine Bromine And Iodine Are Alike In That All The Three.
From resource.studiaacademy.com
2.2 Group 7 (Halogens) Chlorine, Bromine and Iodine Studia Academy Chlorine Bromine And Iodine Are Alike In That All The Three Halogen is a group of six nonmetallic elements in the periodic table, including fluorine, chlorine, bromine, iodine, astatine, and tennessine. Learn about the halogens, a group of nonmetal elements with 7 valence electrons and high reactivity. They include fluorine, chlorine, bromine, iodine, astatine, and possibly tennessine. Learn about the properties and uses of chlorine, bromine and iodine, the three common. Chlorine Bromine And Iodine Are Alike In That All The Three.