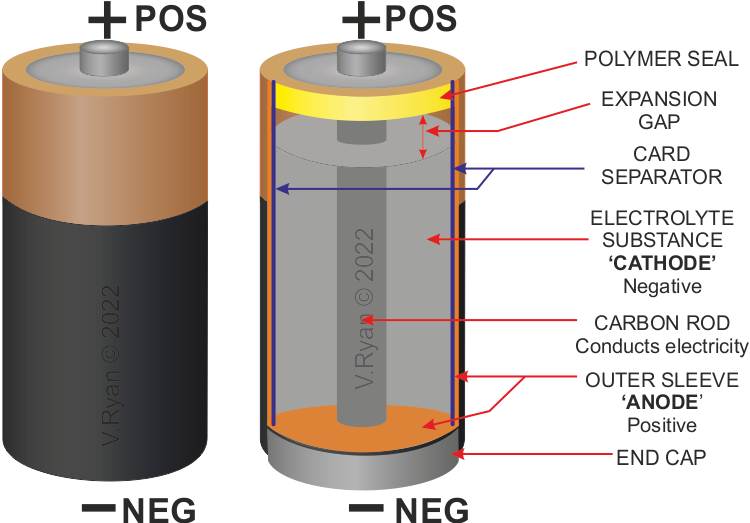
Why Can't Alkaline Batteries Be Recharged Gcse . Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. After some time, alkaline batteries no longer produce charge. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Alkaline batteries), irreversible reactions take place at the electrodes. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Chemical reactions stop when one of the reactants has been used up. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. Alkaline batteries are non rechargeable. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an.
from gioqzurub.blob.core.windows.net
Alkaline batteries), irreversible reactions take place at the electrodes. Chemical reactions stop when one of the reactants has been used up. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. After some time, alkaline batteries no longer produce charge. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Alkaline batteries are non rechargeable.
Alkaline Batteries Gcse Chemistry at Patricia Clark blog
Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Chemical reactions stop when one of the reactants has been used up. Alkaline batteries are non rechargeable. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. After some time, alkaline batteries no longer produce charge. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Alkaline batteries), irreversible reactions take place at the electrodes. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline.
From gioqzurub.blob.core.windows.net
Alkaline Batteries Gcse Chemistry at Patricia Clark blog Why Can't Alkaline Batteries Be Recharged Gcse Alkaline batteries are non rechargeable. Alkaline batteries), irreversible reactions take place at the electrodes. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Chemical reactions stop when one of. Why Can't Alkaline Batteries Be Recharged Gcse.
From powerclues.com
Why are Alkaline Batteries Not Always Recyclable? Power Clues Why Can't Alkaline Batteries Be Recharged Gcse The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. After some time, alkaline batteries no longer produce charge. Alkaline batteries are non rechargeable. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Chemical reactions stop when one of. Why Can't Alkaline Batteries Be Recharged Gcse.
From gioqzurub.blob.core.windows.net
Alkaline Batteries Gcse Chemistry at Patricia Clark blog Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. Chemical reactions stop when one of the reactants has been used up. Study with quizlet and memorize flashcards containing. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.croma.com
Buy Duracell Recharge Plus 750 mAh Alkaline AAA Rechargeable Battery Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. After some time, alkaline batteries no longer produce charge. The most common aa and aaa batteries are called alkaline batteries, and these have. Why Can't Alkaline Batteries Be Recharged Gcse.
From powerclues.com
Can a 1.5 V Battery Be Recharged? (Charging Voltage for 1.5V Battery Why Can't Alkaline Batteries Be Recharged Gcse Alkaline batteries are non rechargeable. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why. Why Can't Alkaline Batteries Be Recharged Gcse.
From powerclues.com
Can a Completely Dead Deep Cycle Battery Be Recharged? (What Happens If Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. After some time, alkaline batteries no longer produce charge. The most common aa and aaa batteries are called alkaline batteries, and these have zinc. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.electricity-magnetism.org
Characteristics of Alkaline Batteries Cell voltage, Capacity & Self Why Can't Alkaline Batteries Be Recharged Gcse After some time, alkaline batteries no longer produce charge. Alkaline batteries are non rechargeable. Chemical reactions stop when one of the reactants has been used up. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.desertcart.com.om
Buy Amazon Basics 48 Count (Pack of 1) AA HighPerformance Alkaline Why Can't Alkaline Batteries Be Recharged Gcse Alkaline batteries are non rechargeable. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. Chemical reactions stop when one of the reactants has been used up. After some time, alkaline batteries no longer produce charge. Alkaline batteries), irreversible reactions take place at the electrodes. Study with quizlet and memorize flashcards. Why Can't Alkaline Batteries Be Recharged Gcse.
From batterygroup.co.uk
Rechargeable vs Single Use Batteries Battery Group Battery Group Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. Chemical reactions stop when one of the. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.which.co.uk
Why you should be using rechargeable batteries Which? News Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. The most. Why Can't Alkaline Batteries Be Recharged Gcse.
From economiasustentable.com
Qué hacer con las pilas usadas y cómo se pueden reciclar Economía Why Can't Alkaline Batteries Be Recharged Gcse After some time, alkaline batteries no longer produce charge. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Alkaline batteries), irreversible reactions take place at the electrodes. In rechargeable cells and batteries, like. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.furniturezstore.com
Rayovac Fusion Premium Alkaline, AA Batteries, 8 Count with SD Card Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. After some time, alkaline batteries no longer produce charge. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Study with quizlet and memorize flashcards containing terms like transition. Why Can't Alkaline Batteries Be Recharged Gcse.
From lifeboat.com
Why can’t you recharge batteries instantly? Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Chemical reactions stop when one of the reactants has been used up. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Study with quizlet and memorize flashcards containing terms like why do. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.columbian.com
The complicated process of recycling batteries in Washington The Why Can't Alkaline Batteries Be Recharged Gcse Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an.. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.linkedin.com
Why can't my car battery be charged? Why Can't Alkaline Batteries Be Recharged Gcse Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Alkaline batteries), irreversible reactions take place at the electrodes. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Alkaline batteries are non rechargeable. Chemical reactions stop when one of. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.duracell.co.uk
Rechargeable AAA Batteries 750mAh Duracell Plus Batteries Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Alkaline batteries), irreversible reactions take place at the electrodes. Chemical reactions stop when one of the reactants has been used up. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.mustangbatterystock.com
price of 9 volt aa/aaa batteries alkaline Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Alkaline batteries are non rechargeable. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when. Why Can't Alkaline Batteries Be Recharged Gcse.
From thorntons-recycling.ie
News Thorntons Recycling Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. After some time, alkaline batteries no longer produce charge. Alkaline batteries are non rechargeable. Alkaline batteries), irreversible reactions take place at the electrodes. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an.. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.victorpro.net
Why Are Alkaline Batteries Better Than Other Types? VictorPro Why Can't Alkaline Batteries Be Recharged Gcse The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. Alkaline batteries are non rechargeable. Chemical reactions stop when one of the reactants has been used up. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. In rechargeable cells and batteries, like the one used. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.gamestop.com
GameStop AA Alkaline Batteries 24 Pack GameStop Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. After some. Why Can't Alkaline Batteries Be Recharged Gcse.
From pickboon.com
Can Lithium Batteries Be Recharged Why Yes or No? Pickboon Why Can't Alkaline Batteries Be Recharged Gcse Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Alkaline batteries), irreversible reactions take place at the electrodes. After some time, alkaline batteries no longer produce charge. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when. Why Can't Alkaline Batteries Be Recharged Gcse.
From ar.inspiredpencil.com
Energizer Rechargeable Batteries Why Can't Alkaline Batteries Be Recharged Gcse Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Chemical reactions stop when one of the reactants has been used up. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. In rechargeable batteries the reactions are reversed by. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.uetechnologies.com
How to Recharge a Rechargeable Battery? (6 Steps & Types) Why Can't Alkaline Batteries Be Recharged Gcse Chemical reactions stop when one of the reactants has been used up. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. The most common aa and aaa batteries. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.literoflightusa.org
Are Regular Batteries Okay for Solar Lights? Why Can't Alkaline Batteries Be Recharged Gcse After some time, alkaline batteries no longer produce charge. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Chemical reactions stop when one of the reactants has been used up. Alkaline batteries are non rechargeable. Study with quizlet and memorize flashcards containing terms like why do alkaline. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.shtfdad.com
How To Recharge Alkaline Batteries Safely And Without Leaks Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Chemical reactions stop when one of the reactants has been used up. After some time, alkaline batteries no longer produce charge. Alkaline batteries), irreversible reactions take. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.youtube.com
Alkaline Batteries Recharging YouTube Why Can't Alkaline Batteries Be Recharged Gcse Alkaline batteries), irreversible reactions take place at the electrodes. After some time, alkaline batteries no longer produce charge. Chemical reactions stop when one of the reactants has been used up. Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Alkaline batteries are non rechargeable. The most common aa and aaa. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.youtube.com
Can you recharge alkaline batteries ? YouTube Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Alkaline batteries are non rechargeable. Alkaline batteries), irreversible reactions take place at the electrodes. Chemical reactions stop when one of the reactants has. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.electricity-magnetism.org
Why are alkaline batteries (AAA or AA) made to be 1.5V while Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Chemical reactions stop when one of the reactants has been used up. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Alkaline batteries are non rechargeable. Alkaline batteries), irreversible reactions take place. Why Can't Alkaline Batteries Be Recharged Gcse.
From discover.hubpages.com
Can Car Battery Be Recharged? HubPages Why Can't Alkaline Batteries Be Recharged Gcse Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Alkaline batteries are non rechargeable. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. After some time, alkaline batteries no longer produce charge. The most common aa and aaa. Why Can't Alkaline Batteries Be Recharged Gcse.
From thepowerfacts.com
Does a Car Battery Recharge Itself While off? How Long Will It Take Why Can't Alkaline Batteries Be Recharged Gcse The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Alkaline batteries), irreversible reactions take place at the electrodes. Alkaline batteries are non rechargeable. After some time, alkaline batteries no. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.pinterest.com
How To Recharge Alkaline Batteries Safely and Without Any Leaks Why Can't Alkaline Batteries Be Recharged Gcse Chemical reactions stop when one of the reactants has been used up. Alkaline batteries), irreversible reactions take place at the electrodes. After some time, alkaline batteries no longer produce charge. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. Alkaline batteries are non rechargeable. The most common aa. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.howtogeek.com
Why Do Alkaline Batteries Leak? Why Can't Alkaline Batteries Be Recharged Gcse In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why can carbon extract nickel?, why do alkaline. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.instructables.com
Easily Recharge Alkaline Batteries! 6 Steps Instructables Why Can't Alkaline Batteries Be Recharged Gcse Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Chemical reactions stop when one of the reactants has been used up. Study with quizlet and memorize flashcards containing terms like transition vs alkali metals, why. Why Can't Alkaline Batteries Be Recharged Gcse.
From circuitbebsonir.z13.web.core.windows.net
Aa Battery Charger And Batteries Why Can't Alkaline Batteries Be Recharged Gcse Alkaline batteries), irreversible reactions take place at the electrodes. In rechargeable cells and batteries, like the one used to power your mobile phone, the chemical reactions can be reversed when an. The most common aa and aaa batteries are called alkaline batteries, and these have zinc metal and manganese dioxide. Chemical reactions stop when one of the reactants has been. Why Can't Alkaline Batteries Be Recharged Gcse.
From www.duracell.in
Duracell Ultra Alkaline C Batteries Why Can't Alkaline Batteries Be Recharged Gcse Study with quizlet and memorize flashcards containing terms like why do alkaline batteries eventually stop working?, why can alkaline. Chemical reactions stop when one of the reactants has been used up. In rechargeable batteries the reactions are reversed by connecting the cells to an external electrical. Alkaline batteries are non rechargeable. Study with quizlet and memorize flashcards containing terms like. Why Can't Alkaline Batteries Be Recharged Gcse.