Chlorine Chemical Reaction In Water . By comparison, the density of air is 1.29 grams per liter. Chlorine + water → hypochlorous acid + hydrochloric acid. The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. When chlorine gas enters the water, the following reaction occurs: When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: Chlorine can be used to clean water and make it drinkable; Cl2 + h2o → hocl +. The reaction of chlorine in water is a disproportionation reaction in which the. When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. Chlorine is a dense gas with a density of 3.21 grams per liter. Reaction of chlorine with water. When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo.
from www.alamy.com
The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. Cl2 + h2o → hocl +. The reaction of chlorine in water is a disproportionation reaction in which the. When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. Reaction of chlorine with water. By comparison, the density of air is 1.29 grams per liter. Chlorine is a dense gas with a density of 3.21 grams per liter. Chlorine can be used to clean water and make it drinkable; When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. When chlorine gas enters the water, the following reaction occurs:
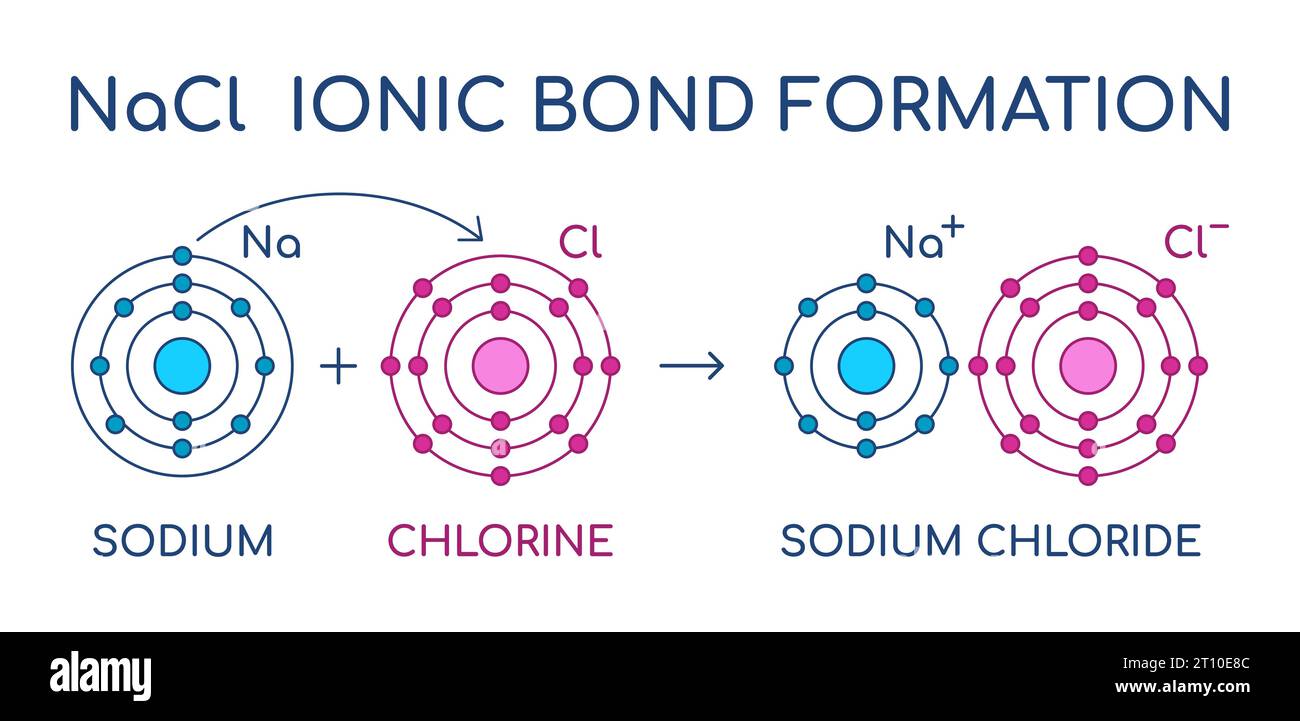
Sodium Chloride ionic bond formation. NaCl structure. Sodium and
Chlorine Chemical Reaction In Water When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. Chlorine + water → hypochlorous acid + hydrochloric acid. The reaction of chlorine in water is a disproportionation reaction in which the. Chlorine can be used to clean water and make it drinkable; By comparison, the density of air is 1.29 grams per liter. When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. When chlorine gas enters the water, the following reaction occurs: Chlorine is a dense gas with a density of 3.21 grams per liter. The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. Reaction of chlorine with water. Cl2 + h2o → hocl +. When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo.
From www.slideserve.com
PPT Chlorine Chemistry PowerPoint Presentation ID1910807 Chlorine Chemical Reaction In Water The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. Cl2 + h2o → hocl +. Chlorine is a dense gas with a density of 3.21 grams per liter. Chlorine can be used to clean water and make it drinkable; When chlorine dissolves in water, it reacts to form the strong. Chlorine Chemical Reaction In Water.
From www.chemistryviews.org
The Chemistry of Pools ChemistryViews Chlorine Chemical Reaction In Water When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. Cl2 + h2o → hocl +. Chlorine is a dense gas with a density of 3.21 grams per liter. When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: When chlorine dissolves in. Chlorine Chemical Reaction In Water.
From slidetodoc.com
REACTION OF CHLORINE WITH WATER Chlorine dissolves in Chlorine Chemical Reaction In Water When chlorine gas enters the water, the following reaction occurs: When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. By comparison, the density of air is 1.29 grams per liter. Chlorine can be used to clean water and make it drinkable;. Chlorine Chemical Reaction In Water.
From elchoroukhost.net
Write The Chemical Formulas Of Two Ions When Table Salt Dissolved In Chlorine Chemical Reaction In Water Chlorine + water → hypochlorous acid + hydrochloric acid. By comparison, the density of air is 1.29 grams per liter. When chlorine gas enters the water, the following reaction occurs: The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. The reaction of chlorine in water is a disproportionation reaction in. Chlorine Chemical Reaction In Water.
From www.sciencephoto.com
Ironchlorine reaction Stock Image C011/4535 Science Photo Library Chlorine Chemical Reaction In Water By comparison, the density of air is 1.29 grams per liter. When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. The reaction of chlorine in water is a disproportionation reaction in which the. Chlorine is a dense gas with a density. Chlorine Chemical Reaction In Water.
From www.alamy.com
Synthesis reaction sodium chloride formation of sodium metal and Chlorine Chemical Reaction In Water When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. The reaction of chlorine in water is a disproportionation reaction in which the. The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. Reaction of chlorine with water. When chlorine gas enters the water,. Chlorine Chemical Reaction In Water.
From www.numerade.com
SOLVED The reaction of chlorine gas with water results in the Chlorine Chemical Reaction In Water Cl2 + h2o → hocl +. When chlorine gas enters the water, the following reaction occurs: When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: Chlorine + water → hypochlorous acid +. Chlorine Chemical Reaction In Water.
From general.chemistrysteps.com
Electrolysis of Water Chemistry Steps Chlorine Chemical Reaction In Water When chlorine gas enters the water, the following reaction occurs: Chlorine can be used to clean water and make it drinkable; By comparison, the density of air is 1.29 grams per liter. When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: The reaction of chlorine in water is a. Chlorine Chemical Reaction In Water.
From chem.libretexts.org
4.3 The Reaction of Sodium with Chlorine Chemistry LibreTexts Chlorine Chemical Reaction In Water The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. Chlorine is a dense gas with a density of 3.21 grams per liter. Reaction of chlorine with water. By comparison, the density of air is 1.29 grams per liter. When chlorine gas is dissolved in water, it also reacts with the. Chlorine Chemical Reaction In Water.
From www.youtube.com
Reaction of Water with Aluminium Chloride YouTube Chlorine Chemical Reaction In Water Chlorine is a dense gas with a density of 3.21 grams per liter. Chlorine + water → hypochlorous acid + hydrochloric acid. Reaction of chlorine with water. The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and. Chlorine Chemical Reaction In Water.
From elchoroukhost.net
Chemical Equation For Table Salt And Water Elcho Table Chlorine Chemical Reaction In Water Cl2 + h2o → hocl +. Reaction of chlorine with water. By comparison, the density of air is 1.29 grams per liter. Chlorine is a dense gas with a density of 3.21 grams per liter. When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for. Chlorine Chemical Reaction In Water.
From www.youtube.com
How to Balance Cl2 + H2O = HCl + O2 (Chlorine gas + Water) YouTube Chlorine Chemical Reaction In Water Reaction of chlorine with water. By comparison, the density of air is 1.29 grams per liter. When chlorine gas enters the water, the following reaction occurs: When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. The reaction of chlorine in water. Chlorine Chemical Reaction In Water.
From studymind.co.uk
Group 1 Reactions (GCSE Chemistry) Study Mind Chlorine Chemical Reaction In Water When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: Chlorine can be used to clean water and make it drinkable; The reaction of chlorine in water is a disproportionation reaction in which the. Chlorine + water → hypochlorous acid + hydrochloric acid. Chlorine is a dense gas with a. Chlorine Chemical Reaction In Water.
From socratic.org
What is the chemical equation for HCl dissolving into water and Chlorine Chemical Reaction In Water Chlorine + water → hypochlorous acid + hydrochloric acid. Chlorine is a dense gas with a density of 3.21 grams per liter. When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. The reaction of chlorine in water is a disproportionation reaction. Chlorine Chemical Reaction In Water.
From www.slideserve.com
PPT Water Treatment Disinfection Processes PowerPoint Presentation Chlorine Chemical Reaction In Water The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: The reaction of chlorine in water is a disproportionation reaction in which the. Chlorine can be used to clean water and. Chlorine Chemical Reaction In Water.
From virttodo.weebly.com
Chlorines reactivity virttodo Chlorine Chemical Reaction In Water Chlorine is a dense gas with a density of 3.21 grams per liter. By comparison, the density of air is 1.29 grams per liter. The reaction of chlorine in water is a disproportionation reaction in which the. Cl2 + h2o → hocl +. Chlorine + water → hypochlorous acid + hydrochloric acid. The reaction of chlorine in water is a. Chlorine Chemical Reaction In Water.
From openoregon.pressbooks.pub
4.3 AcidBase Reactions Introduction to Chemistry Chlorine Chemical Reaction In Water Chlorine + water → hypochlorous acid + hydrochloric acid. Reaction of chlorine with water. When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. Chlorine is a dense gas with a density of 3.21 grams per liter. By comparison, the density of. Chlorine Chemical Reaction In Water.
From spmchemistry.blog.onlinetuition.com.my
Reaction of Alkali Metals with Chlorine SPM Chemistry Chlorine Chemical Reaction In Water When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. Chlorine + water → hypochlorous acid + hydrochloric acid. Chlorine can be. Chlorine Chemical Reaction In Water.
From www.istockphoto.com
Chlorine Chemical Illustrations, RoyaltyFree Vector Graphics & Clip Chlorine Chemical Reaction In Water By comparison, the density of air is 1.29 grams per liter. When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. When chlorine dissolves in water, it reacts to form the. Chlorine Chemical Reaction In Water.
From www.youtube.com
Bleach and Hydrogen Peroxide Reaction + Balanced Equation YouTube Chlorine Chemical Reaction In Water When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. The reaction of chlorine in water is a disproportionation reaction in which the. When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. Reaction of. Chlorine Chemical Reaction In Water.
From www.youtube.com
Balancing and Writing the Equation for Sodium + Chlorine gas YouTube Chlorine Chemical Reaction In Water Chlorine can be used to clean water and make it drinkable; By comparison, the density of air is 1.29 grams per liter. The reaction of chlorine in water is a disproportionation reaction in which the. When chlorine gas enters the water, the following reaction occurs: When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the. Chlorine Chemical Reaction In Water.
From sciencenotes.org
Does Boiling Remove Chlorine? How to Dechlorinate Water Chlorine Chemical Reaction In Water Chlorine is a dense gas with a density of 3.21 grams per liter. By comparison, the density of air is 1.29 grams per liter. Reaction of chlorine with water. When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. When chlorine gas is dissolved in water, it also reacts with the water to. Chlorine Chemical Reaction In Water.
From www.techquintal.com
10 Advantages and Disadvantages of Chlorination of Water to Know Tech Chlorine Chemical Reaction In Water By comparison, the density of air is 1.29 grams per liter. Reaction of chlorine with water. Cl2 + h2o → hocl +. When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. Chlorine + water → hypochlorous acid + hydrochloric acid. The reaction of chlorine in water is a disproportionation reaction in which. Chlorine Chemical Reaction In Water.
From www.slideserve.com
PPT Residual Chlorine & Chlorine Demand PowerPoint Presentation ID Chlorine Chemical Reaction In Water When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: When chlorine gas enters the water, the following reaction occurs: When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. Chlorine is. Chlorine Chemical Reaction In Water.
From www.youtube.com
13.1.2 Describe the reactions of chlorine and the chlorides referred to Chlorine Chemical Reaction In Water Chlorine is a dense gas with a density of 3.21 grams per liter. The reaction of chlorine in water is a disproportionation reaction in which the. Reaction of chlorine with water. By comparison, the density of air is 1.29 grams per liter. Cl2 + h2o → hocl +. The reaction of chlorine in water is a disproportionation reaction in which. Chlorine Chemical Reaction In Water.
From www.slideserve.com
PPT Analyze Immediately PowerPoint Presentation ID375903 Chlorine Chemical Reaction In Water Chlorine is a dense gas with a density of 3.21 grams per liter. Chlorine can be used to clean water and make it drinkable; Reaction of chlorine with water. When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. When chlorine gas enters the water, the following reaction occurs: By comparison, the density. Chlorine Chemical Reaction In Water.
From www.compoundchem.com
Chlorination & Pee in the Pool The Chemistry of Swimming Pools Chlorine Chemical Reaction In Water The reaction of chlorine in water is a disproportionation reaction in which the. Chlorine is a dense gas with a density of 3.21 grams per liter. Chlorine can be used to clean water and make it drinkable; When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. By comparison, the density of air. Chlorine Chemical Reaction In Water.
From kids.britannica.com
chemical reaction Students Britannica Kids Homework Help Chlorine Chemical Reaction In Water When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. When chlorine gas enters the water, the following reaction occurs: By comparison, the density of air is 1.29 grams per liter. Chlorine can be used to clean water and make it drinkable;. Chlorine Chemical Reaction In Water.
From byjus.com
31. How to write a balanced chemical equation when phosphorus reacts Chlorine Chemical Reaction In Water The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. Chlorine is a dense gas with a density of 3.21 grams per liter. Reaction of chlorine with water. When chlorine gas enters the water, the following reaction occurs: Chlorine + water → hypochlorous acid + hydrochloric acid. By comparison, the density. Chlorine Chemical Reaction In Water.
From blog.orendatech.com
Understanding Breakpoint Chlorination Chlorine Chemical Reaction In Water When chlorine gas is dissolved in water, it also reacts with the water to form hypochlorous acid and hydrochloric acid: By comparison, the density of air is 1.29 grams per liter. Chlorine + water → hypochlorous acid + hydrochloric acid. The reaction of chlorine in water is a disproportionation reaction in which the. When chlorine dissolves in water, it reacts. Chlorine Chemical Reaction In Water.
From www.tessshebaylo.com
Salt Water Electrolysis Equation Tessshebaylo Chlorine Chemical Reaction In Water The reaction of chlorine in water is a disproportionation reaction in which the chlorine gets both oxidised and reduced. Cl2 + h2o → hocl +. Chlorine can be used to clean water and make it drinkable; Chlorine is a dense gas with a density of 3.21 grams per liter. When chlorine dissolves in water, it reacts to form the strong. Chlorine Chemical Reaction In Water.
From www.youtube.com
Synthesizing and Condensing Chlorine + Liquid Chlorine and Sodium Chlorine Chemical Reaction In Water By comparison, the density of air is 1.29 grams per liter. The reaction of chlorine in water is a disproportionation reaction in which the. Chlorine is a dense gas with a density of 3.21 grams per liter. When chlorine gas enters the water, the following reaction occurs: When chlorine dissolves in water, it reacts to form the strong acid, hcl,. Chlorine Chemical Reaction In Water.
From hydrogenchloridemekaiga.blogspot.com
Hydrogen Chloride Hydrogen Chloride In Water Equation Chlorine Chemical Reaction In Water Reaction of chlorine with water. Chlorine can be used to clean water and make it drinkable; By comparison, the density of air is 1.29 grams per liter. When chlorine gas enters the water, the following reaction occurs: Cl2 + h2o → hocl +. When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but. Chlorine Chemical Reaction In Water.
From www.alamy.com
Sodium Chloride ionic bond formation. NaCl structure. Sodium and Chlorine Chemical Reaction In Water When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the bleaching properties. The reaction of chlorine in water is a disproportionation reaction in which the. Reaction of chlorine with water. When chlorine gas enters the water, the following reaction occurs: The reaction of chlorine. Chlorine Chemical Reaction In Water.
From www.tessshebaylo.com
Chemical Equation For Water Purification Tessshebaylo Chlorine Chemical Reaction In Water Cl2 + h2o → hocl +. Chlorine can be used to clean water and make it drinkable; When chlorine reacts with water, it forms a mixture of hydrochloric acid, hcl, and chloric acid, hclo. When chlorine dissolves in water, it reacts to form the strong acid, hcl, and the weak but strongly oxidising acid, hocl, which is responsible for the. Chlorine Chemical Reaction In Water.