Product Between Hydrochloric Acid And Zinc Metal . Zinc also reacts with hcl, releasing small bubbles of. Let’s examine the example of the interaction between zinc and hydrochloric acid. Neutralisation is the reaction between an acid and a base. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! Zinc + hydrogen chloride = zinc chloride + dihydrogen. Acids will react with reactive metals, such as magnesium. Acids react with metals, bases and carbonates to produce salts. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. The bubbles are hydrogen gas (right side of. The reaction between zinc metal and the. Zn(s) + 2hcl(aq) → zncl2(aq).
from www.alamy.com
The reaction between zinc metal and the. Acids will react with reactive metals, such as magnesium. Neutralisation is the reaction between an acid and a base. Let’s examine the example of the interaction between zinc and hydrochloric acid. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. This chemistry video tutorial explains how to predict the products of the single replacement. It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. The bubbles are hydrogen gas (right side of. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals.
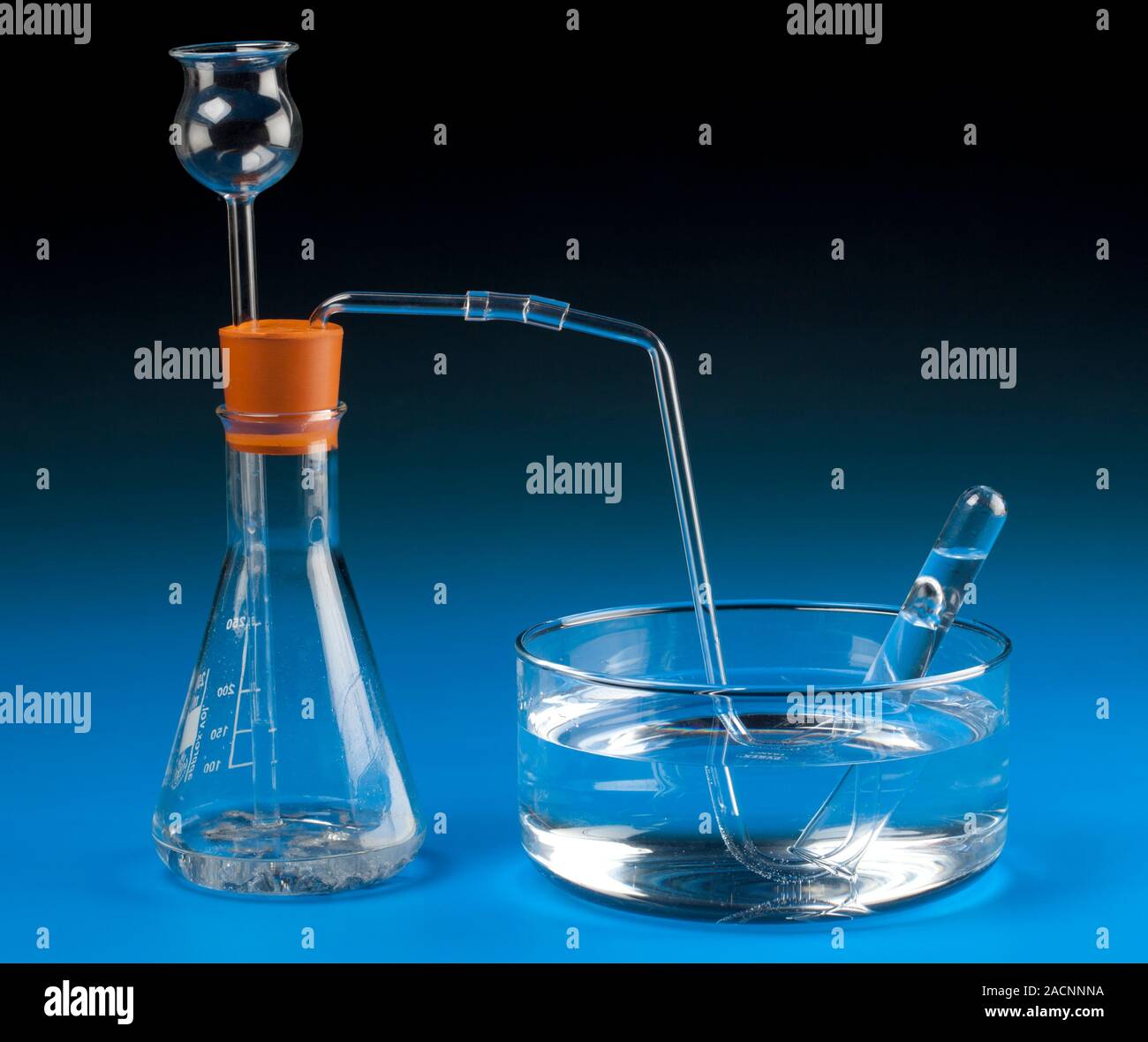
Metalacid reaction hydrogen collection. Apparatus for collecting hydrogen from the reaction
Product Between Hydrochloric Acid And Zinc Metal The reaction between zinc metal and the. This chemistry video tutorial explains how to predict the products of the single replacement. The bubbles are hydrogen gas (right side of. Zinc also reacts with hcl, releasing small bubbles of. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. Neutralisation is the reaction between an acid and a base. Let’s examine the example of the interaction between zinc and hydrochloric acid. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. Zn(s) + 2hcl(aq) → zncl2(aq). Zinc + hydrogen chloride = zinc chloride + dihydrogen. Acids react with metals, bases and carbonates to produce salts. Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. Acids will react with reactive metals, such as magnesium.
From www.sciencephoto.com
Zinc reacts with hydrochloric acid Stock Image C052/7641 Science Photo Library Product Between Hydrochloric Acid And Zinc Metal Zn(s) + 2hcl(aq) → zncl2(aq). Neutralisation is the reaction between an acid and a base. Zinc + hydrogen chloride = zinc chloride + dihydrogen. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. Acids will react with reactive metals, such as magnesium. This experiment aims to study the displacement reaction between hydrochloric acid with. Product Between Hydrochloric Acid And Zinc Metal.
From fyouusdzh.blob.core.windows.net
Mossy Zinc And Hydrochloric Acid Balanced Equation at Nell Everett blog Product Between Hydrochloric Acid And Zinc Metal Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. The bubbles are hydrogen gas (right side of. Acids react with metals, bases and carbonates to produce salts. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. The reaction between zinc and hydrochloric acid can be explained by the activity. Product Between Hydrochloric Acid And Zinc Metal.
From www.coursehero.com
[Solved] Consider the reaction of zinc metal with hydrochloric acid Zn +... Course Hero Product Between Hydrochloric Acid And Zinc Metal The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. Zn(s) + 2hcl(aq) → zncl2(aq). It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. Let’s examine the example of the interaction between zinc and hydrochloric acid. Zn. Product Between Hydrochloric Acid And Zinc Metal.
From www.youtube.com
Reaction Of Zinc with Hydrochloric acid Chemistry demonstration YouTube Product Between Hydrochloric Acid And Zinc Metal The reaction between zinc metal and the. Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Zinc also reacts with hcl, releasing small bubbles of. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. It. Product Between Hydrochloric Acid And Zinc Metal.
From www.chegg.com
Solved 1. The reaction of zinc metal and hydrochloric acid Product Between Hydrochloric Acid And Zinc Metal Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. Zinc also reacts with hcl, releasing small bubbles of. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. The reaction between zinc metal and. Product Between Hydrochloric Acid And Zinc Metal.
From www.alamy.com
Metalacid reaction hydrogen collection. Apparatus for collecting hydrogen from the reaction Product Between Hydrochloric Acid And Zinc Metal Zinc also reacts with hcl, releasing small bubbles of. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. Acids will react with reactive metals, such as magnesium. Let’s examine the example of the interaction between zinc and hydrochloric acid. It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! Zinc is more. Product Between Hydrochloric Acid And Zinc Metal.
From www.slideshare.net
Metals Reactivity Series Product Between Hydrochloric Acid And Zinc Metal The bubbles are hydrogen gas (right side of. This chemistry video tutorial explains how to predict the products of the single replacement. Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. Zn(s) + 2hcl(aq) → zncl2(aq). This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. Let’s examine the example of. Product Between Hydrochloric Acid And Zinc Metal.
From www.nagwa.com
Question Video Describing the Correct Symbol Equation for the Reaction between Zinc Metal and Product Between Hydrochloric Acid And Zinc Metal The reaction between zinc metal and the. The bubbles are hydrogen gas (right side of. Zinc + hydrogen chloride = zinc chloride + dihydrogen. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Zn(s) + 2hcl(aq) → zncl2(aq). Zn. Product Between Hydrochloric Acid And Zinc Metal.
From readingandwritingprojectcom.web.fc2.com
zinc metal and hydrochloric acid Product Between Hydrochloric Acid And Zinc Metal Acids react with metals, bases and carbonates to produce salts. Zinc + hydrogen chloride = zinc chloride + dihydrogen. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. The reaction between zinc metal and the. The bubbles are hydrogen. Product Between Hydrochloric Acid And Zinc Metal.
From melscience.com
The reaction between hydrochloric acid and zinc MEL Chemistry Product Between Hydrochloric Acid And Zinc Metal Let’s examine the example of the interaction between zinc and hydrochloric acid. Zn(s) + 2hcl(aq) → zncl2(aq). The bubbles are hydrogen gas (right side of. Zinc also reacts with hcl, releasing small bubbles of. Acids will react with reactive metals, such as magnesium. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. This chemistry. Product Between Hydrochloric Acid And Zinc Metal.
From spark.adobe.com
Zinc and Hydrochloric Acid Product Between Hydrochloric Acid And Zinc Metal Acids will react with reactive metals, such as magnesium. This chemistry video tutorial explains how to predict the products of the single replacement. Let’s examine the example of the interaction between zinc and hydrochloric acid. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. Zinc + hydrogen chloride = zinc chloride + dihydrogen. The. Product Between Hydrochloric Acid And Zinc Metal.
From fphoto.photoshelter.com
science chemistry redox reaction zinc hydrochloric acid Fundamental Photographs The Art of Product Between Hydrochloric Acid And Zinc Metal Acids will react with reactive metals, such as magnesium. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. Let’s examine the example of the interaction between zinc and hydrochloric acid. The reaction between zinc metal and the. Zinc also reacts with hcl, releasing small bubbles of. It is fairly obvious that zinc metal reacts with. Product Between Hydrochloric Acid And Zinc Metal.
From www.pdffiller.com
The reaction between hydrochloric acid and zinc MEL Chemistry Doc Template pdfFiller Product Between Hydrochloric Acid And Zinc Metal Acids react with metals, bases and carbonates to produce salts. Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. The bubbles are hydrogen gas (right side of. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. Let’s examine the example of the interaction between zinc and hydrochloric acid.. Product Between Hydrochloric Acid And Zinc Metal.
From www.youtube.com
Zn + HCl Reaction Zinc + Hydrochloric Acid YouTube Product Between Hydrochloric Acid And Zinc Metal The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Let’s examine the example of the interaction between zinc and hydrochloric acid. Neutralisation is the reaction between an acid and a base. This experiment aims to study the displacement reaction. Product Between Hydrochloric Acid And Zinc Metal.
From warreninstitute.org
Unlock The POWER Of Chemical Reaction Zn + HCl To Zinc + Hydrochloric Acid Product Between Hydrochloric Acid And Zinc Metal This chemistry video tutorial explains how to predict the products of the single replacement. Zn(s) + 2hcl(aq) → zncl2(aq). Let’s examine the example of the interaction between zinc and hydrochloric acid. The reaction between zinc metal and the. It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! Neutralisation is the reaction between an acid and a base.. Product Between Hydrochloric Acid And Zinc Metal.
From www.sciencephoto.com
Zinc reacting with hydrochloric acid Stock Image A500/0662 Science Photo Library Product Between Hydrochloric Acid And Zinc Metal Zinc + hydrogen chloride = zinc chloride + dihydrogen. Acids react with metals, bases and carbonates to produce salts. Neutralisation is the reaction between an acid and a base. The reaction between zinc metal and the. The bubbles are hydrogen gas (right side of. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals.. Product Between Hydrochloric Acid And Zinc Metal.
From www.sciencephoto.com
Zinc reacting with hydrochloric acid Stock Image A500/0309 Science Photo Library Product Between Hydrochloric Acid And Zinc Metal Neutralisation is the reaction between an acid and a base. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. Let’s examine the example of the interaction between. Product Between Hydrochloric Acid And Zinc Metal.
From polizneuro.weebly.com
Reactivity series of metals polizneuro Product Between Hydrochloric Acid And Zinc Metal The reaction between zinc metal and the. Zinc + hydrogen chloride = zinc chloride + dihydrogen. Zinc also reacts with hcl, releasing small bubbles of. Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. It is fairly obvious that zinc. Product Between Hydrochloric Acid And Zinc Metal.
From www.slideserve.com
PPT KS3 Chemistry PowerPoint Presentation, free download ID639260 Product Between Hydrochloric Acid And Zinc Metal This chemistry video tutorial explains how to predict the products of the single replacement. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Zn(s) + 2hcl(aq) → zncl2(aq). This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. The bubbles are hydrogen gas (right side of. The reaction between zinc metal. Product Between Hydrochloric Acid And Zinc Metal.
From fphoto.photoshelter.com
science chemistry redox reaction zinc hydrochloric acid Fundamental Photographs The Art of Product Between Hydrochloric Acid And Zinc Metal Zinc + hydrogen chloride = zinc chloride + dihydrogen. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. Let’s examine the example of the interaction between zinc and hydrochloric acid. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Neutralisation is the reaction between an acid and a base. Zinc. Product Between Hydrochloric Acid And Zinc Metal.
From www.chegg.com
Solved 5. Zinc metal reacts with hydrochloric acid to Product Between Hydrochloric Acid And Zinc Metal Zinc also reacts with hcl, releasing small bubbles of. Zinc + hydrogen chloride = zinc chloride + dihydrogen. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! Neutralisation is the reaction between an acid and a base. The reaction between zinc metal. Product Between Hydrochloric Acid And Zinc Metal.
From slideplayer.com
Types of Chemical Reactions Ch ppt download Product Between Hydrochloric Acid And Zinc Metal Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. The reaction between zinc metal and the. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Let’s examine the example of the interaction between zinc and hydrochloric acid. Zinc is more reactive than hydrogen, so it can displace hydrogen from. Product Between Hydrochloric Acid And Zinc Metal.
From www.numerade.com
In this problem, zinc reacts with hydrochloric acid to make zinc chloride and hydrogen gas Product Between Hydrochloric Acid And Zinc Metal Acids will react with reactive metals, such as magnesium. It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! The bubbles are hydrogen gas (right side of. The reaction between zinc metal and the. Zinc also reacts with hcl, releasing small bubbles of. Let’s examine the example of the interaction between zinc and hydrochloric acid. Zn(s) + 2hcl(aq). Product Between Hydrochloric Acid And Zinc Metal.
From www.teachoo.com
Assertion (A) When zinc is added to dilute hydrochloric acid, hydro Product Between Hydrochloric Acid And Zinc Metal Acids react with metals, bases and carbonates to produce salts. The reaction between zinc metal and the. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. Acids will react with reactive metals, such as magnesium. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. For example, zinc. Product Between Hydrochloric Acid And Zinc Metal.
From express.adobe.com
Zinc and Hydrochloric Acid Product Between Hydrochloric Acid And Zinc Metal Zinc also reacts with hcl, releasing small bubbles of. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Acids react with metals, bases and carbonates to produce salts. Let’s examine the example of the interaction between zinc and hydrochloric acid. Zinc + hydrogen chloride = zinc chloride + dihydrogen. Acids will react with reactive metals,. Product Between Hydrochloric Acid And Zinc Metal.
From askfilo.com
Zinc metal reacts with hydrochloric acid by the following reaction Zn(s).. Product Between Hydrochloric Acid And Zinc Metal Neutralisation is the reaction between an acid and a base. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. Acids will react with reactive metals, such as magnesium. Zinc + hydrogen chloride = zinc chloride + dihydrogen. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. It is. Product Between Hydrochloric Acid And Zinc Metal.
From www.teachoo.com
Reaction of Metals and NonMetals with Acids Teachoo Concepts Product Between Hydrochloric Acid And Zinc Metal The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. This chemistry video tutorial explains how to predict the products of the single replacement. Zinc also reacts with hcl, releasing small bubbles of. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. Zinc + hydrogen chloride = zinc. Product Between Hydrochloric Acid And Zinc Metal.
From www.slideshare.net
Metals Product Between Hydrochloric Acid And Zinc Metal Neutralisation is the reaction between an acid and a base. Zinc + hydrogen chloride = zinc chloride + dihydrogen. Let’s examine the example of the interaction between zinc and hydrochloric acid. Zn(s) + 2hcl(aq) → zncl2(aq). It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and. Product Between Hydrochloric Acid And Zinc Metal.
From www.slideserve.com
PPT KS3 Chemistry PowerPoint Presentation, free download ID639260 Product Between Hydrochloric Acid And Zinc Metal For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. This chemistry video tutorial explains how to predict the products of the single replacement. The reaction between zinc metal and the. Zinc is more reactive than hydrogen, so it can displace hydrogen from the acid. Zn(s) + 2hcl(aq) → zncl2(aq). Zinc + hydrogen chloride = zinc. Product Between Hydrochloric Acid And Zinc Metal.
From www.youtube.com
The reaction between hydrochloric acid and zinc metal + pop test YouTube Product Between Hydrochloric Acid And Zinc Metal Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. It is fairly obvious that zinc metal reacts with aqueous hydrochloric acid! Zn(s) + 2hcl(aq) → zncl2(aq). This chemistry video tutorial explains how to predict the products of the. Product Between Hydrochloric Acid And Zinc Metal.
From www.teachoo.com
How do Acids and Bases react with Metals? [with Examples] Teachoo Product Between Hydrochloric Acid And Zinc Metal Zinc + hydrogen chloride = zinc chloride + dihydrogen. Neutralisation is the reaction between an acid and a base. The bubbles are hydrogen gas (right side of. The reaction between zinc metal and the. Zinc also reacts with hcl, releasing small bubbles of. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. It is fairly. Product Between Hydrochloric Acid And Zinc Metal.
From www.youtube.com
Reaction of Zinc and Hydrochloric acid YouTube Product Between Hydrochloric Acid And Zinc Metal Acids react with metals, bases and carbonates to produce salts. Zinc also reacts with hcl, releasing small bubbles of. The bubbles are hydrogen gas (right side of. Zinc + hydrogen chloride = zinc chloride + dihydrogen. Neutralisation is the reaction between an acid and a base. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal.. Product Between Hydrochloric Acid And Zinc Metal.
From www.numerade.com
SOLVED Consider the reaction between zinc metal and hydrochloric acid. Which of the following Product Between Hydrochloric Acid And Zinc Metal Let’s examine the example of the interaction between zinc and hydrochloric acid. The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. Zinc is more reactive. Product Between Hydrochloric Acid And Zinc Metal.
From www.chegg.com
Solved Zinc metal reacts with hydrochloric acid to produce Product Between Hydrochloric Acid And Zinc Metal Neutralisation is the reaction between an acid and a base. Acids will react with reactive metals, such as magnesium. For example, zinc metal reacts with hydrochloric acid, producing zinc chloride and hydrogen gas. Acids react with metals, bases and carbonates to produce salts. Zn + hcl = zncl2 + h2 is a single displacement (substitution) reaction where one. It is. Product Between Hydrochloric Acid And Zinc Metal.
From www.teachoo.com
Reactions of Acids and Bases Full list (with Examples) Teachoo Product Between Hydrochloric Acid And Zinc Metal The reaction between zinc and hydrochloric acid can be explained by the activity series of metals. Acids will react with reactive metals, such as magnesium. Acids react with metals, bases and carbonates to produce salts. Zinc + hydrogen chloride = zinc chloride + dihydrogen. This experiment aims to study the displacement reaction between hydrochloric acid with zinc metal. Zinc is. Product Between Hydrochloric Acid And Zinc Metal.