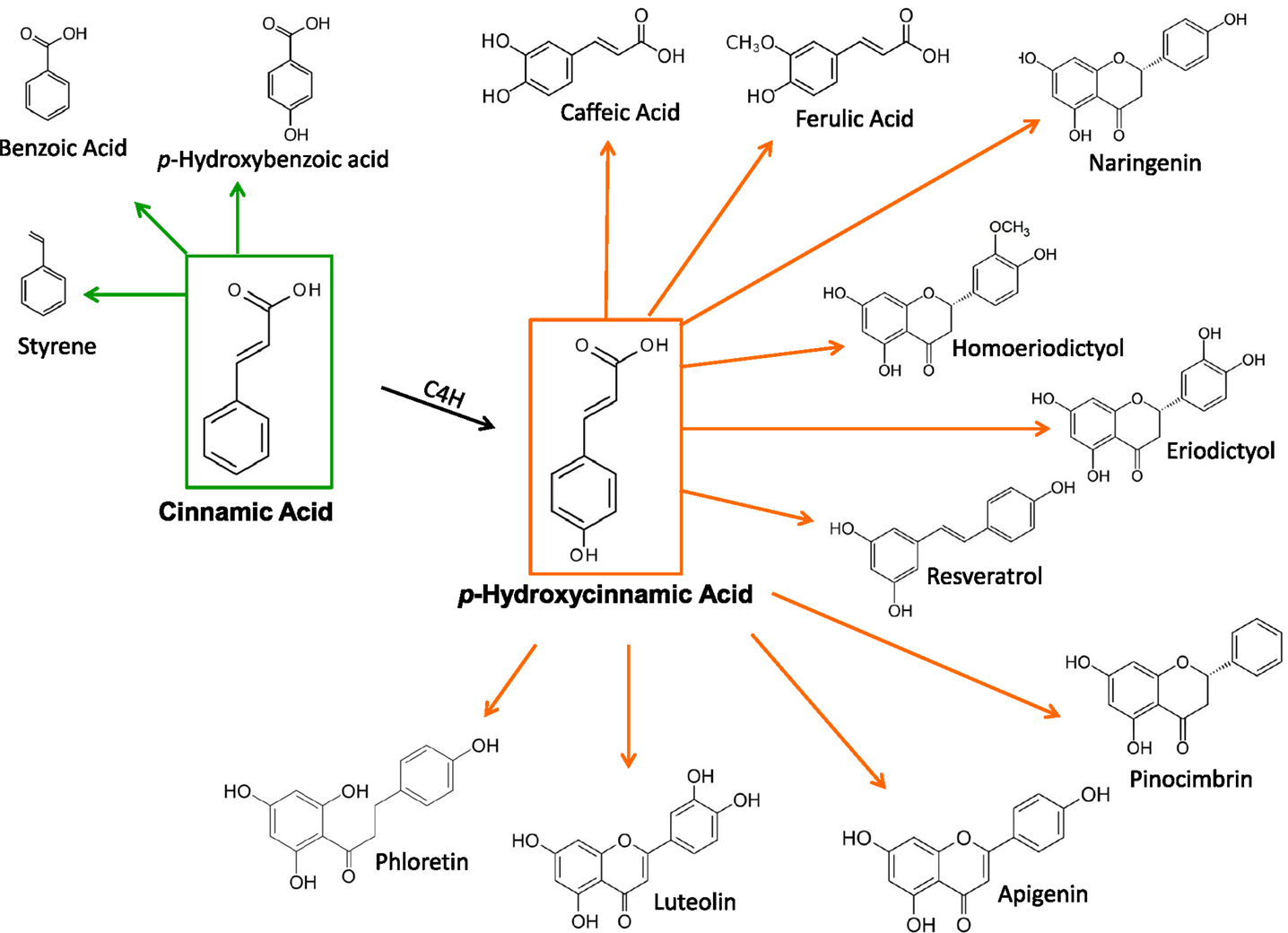
Laboratory Preparation Of Cinnamic Acid . This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted in their multiple functional groups.
from www.frontiersin.org
The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%).
Frontiers Production of Cinnamic and pHydroxycinnamic Acids in
Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and.
From www.chegg.com
Solved Lab Notes for the Synthesis of Cinnamic Acids The Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. The great success of cinnamic acids was rooted in their multiple functional groups. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to. Laboratory Preparation Of Cinnamic Acid.
From www.numerade.com
SOLVED transcinnamic acid Bromination of transcinnamic acid can Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted in their multiple functional groups. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g. Laboratory Preparation Of Cinnamic Acid.
From studylib.net
Synthesis and Bioactivity Evaluation of Cinnamic Acid Esters from Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From file.scirp.org
Organocatalyzed Decarboxylation of Naturally Occurring Cinnamic Acids Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
Construction of cinnamylamine biosynthetic pathway using cinnamic acid Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From docslib.org
Organic Chemistry II Laboratory Stereochemistry of the Addition of Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.youtube.com
Synthesis of Cinnamic Acid From Benzaldehyde YouTube Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted in their multiple functional groups. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition,. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
Synthesis of 3carboxycoumarins, cinnamic acid derivatives via Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted in their multiple functional groups. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g. Laboratory Preparation Of Cinnamic Acid.
From www.youtube.com
PARKIN REACTION(Preparation of Cinnamic acid)BySunitakumawat YouTube Laboratory Preparation Of Cinnamic Acid The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to. Laboratory Preparation Of Cinnamic Acid.
From www.youtube.com
Cinnamic Acid Organic Synthesis ( Perkin Reaction ) YouTube Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
Structure of cinnamic acid (1) (A). Synthesis of cinnamic acid analogs Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition,. Laboratory Preparation Of Cinnamic Acid.
From www.semanticscholar.org
Synthesis of Cinnamic Acid Based on Perkin Reaction Using Sonochemical Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
(PDF) An Efficient Perkin Synthesis of 13C‐Labelled Cinnamic Acids From Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
Structure of cinnamic acid and the substituents of the aromatic ring of Laboratory Preparation Of Cinnamic Acid The great success of cinnamic acids was rooted in their multiple functional groups. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g. Laboratory Preparation Of Cinnamic Acid.
From www.chegg.com
Solved Lab Bromination of Trans Cinnamic AcidPlease Hep me Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.studocu.com
Chem 343 Lab 11 Report Bromination of Cinnamic Acid and 1 H NMR Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition,. Laboratory Preparation Of Cinnamic Acid.
From www.chegg.com
Lab Notes for the Synthesis of Cinnamic Acids The Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. The great success of cinnamic acids was rooted in their multiple functional groups. This novel approach allows the preparation of various cinnamic acids in good to. Laboratory Preparation Of Cinnamic Acid.
From www.chegg.com
Solved In this experiment you will react transcinnamic acid Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition,. Laboratory Preparation Of Cinnamic Acid.
From www.frontiersin.org
Frontiers Production of Cinnamic and pHydroxycinnamic Acids in Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.chegg.com
Solved propose a mechanism of the Synthesis of cinnamic acid Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition,. Laboratory Preparation Of Cinnamic Acid.
From www.semanticscholar.org
Table 1 from A NOVEL DIRECT BORONMEDIATED SYNTHESIS OF CINNAMIC ACIDS Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. The great success of cinnamic acids was rooted in their multiple functional groups. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
General reaction scheme for pyridinefree synthesis of cinnamic acids Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.studocu.com
Chem 361 lab 4 Daood Tanvir Gill (2011214) Experiment 4 Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
Scheme 1. Synthesis of cinnamic acid derivatives (a) ROH, H2SO4 Laboratory Preparation Of Cinnamic Acid The great success of cinnamic acids was rooted in their multiple functional groups. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition,. Laboratory Preparation Of Cinnamic Acid.
From ar.inspiredpencil.com
Trans Cinnamic Acid Structure Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.chegg.com
Solved Synthesis Comment OH H2SO4 OH Cinnamic acid Ethanol Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition,. Laboratory Preparation Of Cinnamic Acid.
From www.mdpi.com
Molecules Free FullText A Novel Approach in Cinnamic Acid Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted in their multiple functional groups. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. Herein, reactions triggered by the radical addition,. Laboratory Preparation Of Cinnamic Acid.
From www.chemistry-online.com
Synthesis of cinnamic acid Chemistry Online Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. This novel approach allows the preparation of various cinnamic acids in good to. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
Phenylpropanoid biosynthesis through the shikimic acid and cinnamic Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.youtube.com
Perkin's Reaction Preparation of Cinnamic Acid Carbonyl Compounds Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted in their multiple functional groups. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g. Laboratory Preparation Of Cinnamic Acid.
From www.chegg.com
Lab Notes for the Synthesis of Cinnamic Acids The Laboratory Preparation Of Cinnamic Acid The great success of cinnamic acids was rooted in their multiple functional groups. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition,. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
(PDF) Synthesis and characterization of new derivatives of cinnamic acid Laboratory Preparation Of Cinnamic Acid This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.mdpi.com
Molecules Free FullText Multifunctional Cinnamic Acid Derivatives Laboratory Preparation Of Cinnamic Acid About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. This novel approach allows the preparation of various cinnamic acids in good to. Laboratory Preparation Of Cinnamic Acid.
From www.chegg.com
Lab Notes for the Synthesis of Cinnamic Acids The Laboratory Preparation Of Cinnamic Acid Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to high yields (up to 81%). The great success of cinnamic acids was rooted. Laboratory Preparation Of Cinnamic Acid.
From www.researchgate.net
Enzymatic synthesis of pcoumaric acid/transcinnamic acid by TAL and Laboratory Preparation Of Cinnamic Acid The great success of cinnamic acids was rooted in their multiple functional groups. Herein, reactions triggered by the radical addition, electrophilic addition, michael addition and. About 10.5 g (10 ml) of benzaldehyde, 15 g (14 ml) of acetic anhydride and 6 g of finely powdered potassium acetate. This novel approach allows the preparation of various cinnamic acids in good to. Laboratory Preparation Of Cinnamic Acid.