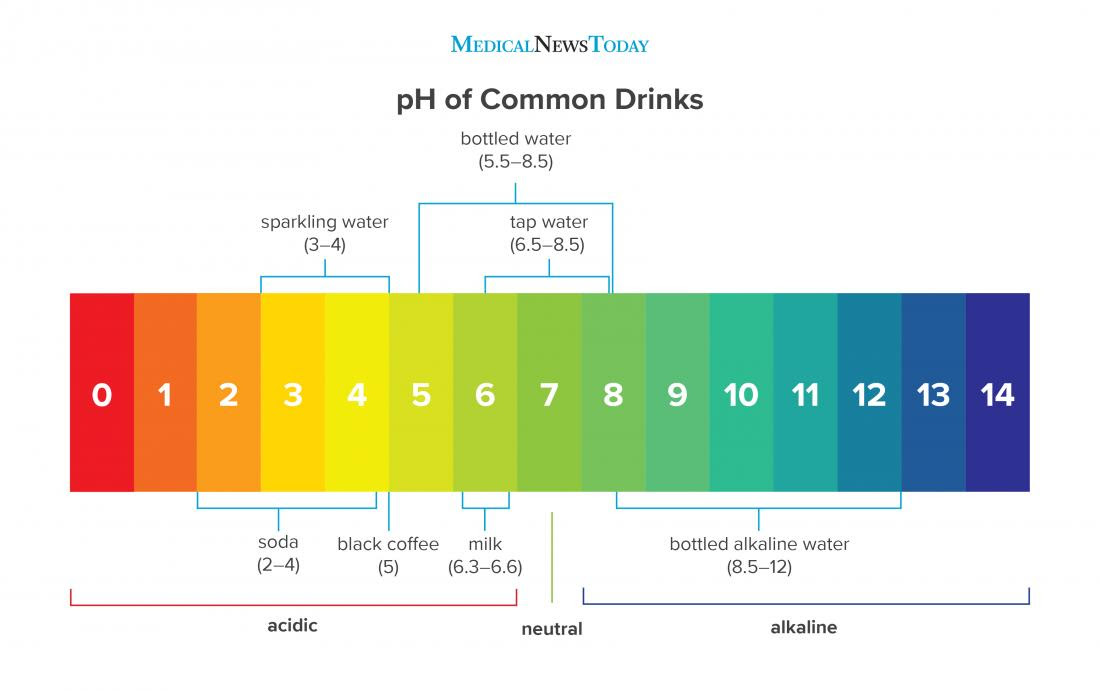
What S The Ph Of Pure Water . This is because water absorbs carbon dioxide from the air, which is then slowly converted. simply put the ph of pure water is 7. when pure water is exposed to air, it becomes mildly acidic. at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. the ph of water. However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5.
from news.regenerativemedgroup.com
However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). simply put the ph of pure water is 7. the ph of water. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. This is because water absorbs carbon dioxide from the air, which is then slowly converted. at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7.
The pH of water What to know Regenerative Medical Group
What S The Ph Of Pure Water the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. simply put the ph of pure water is 7. when pure water is exposed to air, it becomes mildly acidic. although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. the ph of water. This is because water absorbs carbon dioxide from the air, which is then slowly converted. All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink.
From docslib.org
Measuring Ph of Pure Water and Other Low Conductivity Waters DocsLib What S The Ph Of Pure Water when pure water is exposed to air, it becomes mildly acidic. This is because water absorbs carbon dioxide from the air, which is then slowly converted. at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. All acids have a ph that is lower than 7 and all. What S The Ph Of Pure Water.
From www.slideserve.com
PPT ACID RAIN PowerPoint Presentation, free download ID2170150 What S The Ph Of Pure Water although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. when pure water is exposed to air, it becomes mildly acidic. Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. When you’re measuring. What S The Ph Of Pure Water.
From opentextbc.ca
Water Hardness and pH Understanding Ingredients for the Canadian Baker What S The Ph Of Pure Water simply put the ph of pure water is 7. at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. Surface water systems typically have a ph. What S The Ph Of Pure Water.
From www.youtube.com
What is the pH value of pure water? YouTube What S The Ph Of Pure Water although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. the ph of water. when pure water is exposed to air, it becomes mildly acidic. simply put the ph of pure water is 7. Surface water systems typically have a ph range of 6.5 to. What S The Ph Of Pure Water.
From drowwater.com
Best pH Level for Drinking Water? DrowWater What S The Ph Of Pure Water simply put the ph of pure water is 7. when pure water is exposed to air, it becomes mildly acidic. All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. This is because water absorbs carbon dioxide from the air, which is then slowly converted. although. What S The Ph Of Pure Water.
From v9306.1blu.de
PH Scale Indicators Mini Chemistry Learn Chemistry Online What S The Ph Of Pure Water the ph of water. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. When you’re measuring the ph of water, a 7.0 reading. What S The Ph Of Pure Water.
From byjus.com
At 100 degree C kw =10^ 12.the pH of pure water at 100 degree Celsius What S The Ph Of Pure Water When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. This is because water absorbs carbon dioxide from the air, which is then slowly converted. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. simply put the ph. What S The Ph Of Pure Water.
From www.toppr.com
The pH of pure water at 50^oC is( pKw = 13.26 at 50^oC) What S The Ph Of Pure Water However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). This is because water absorbs carbon dioxide from the air, which is then slowly converted. although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. the ph of water.. What S The Ph Of Pure Water.
From www.youtube.com
IS the pH of PURE WATER always 7? pH of water calculation A Level What S The Ph Of Pure Water This is because water absorbs carbon dioxide from the air, which is then slowly converted. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. when pure water is exposed to air, it becomes mildly acidic. although the ph of pure water is 7,. What S The Ph Of Pure Water.
From perfect-hydration.com
Drinking Water pH Explained What Should Be the Ideal pH? Perfect What S The Ph Of Pure Water although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. This is because water absorbs carbon dioxide from the air, which is then slowly converted.. What S The Ph Of Pure Water.
From www.springwellwater.com
PH Explained Understanding pH in Your Home's Drinking Water What S The Ph Of Pure Water Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. the ph of water. when pure water is exposed to air, it becomes mildly acidic. All acids have a ph that is lower than 7 and all bases have a ph that is higher than. What S The Ph Of Pure Water.
From www.scienceabc.com
Why Does The pH Scale Range From 0 To 14? Can It Go Beyond That Range? What S The Ph Of Pure Water at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. simply put the ph of pure water is 7. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. This is because water absorbs carbon dioxide from the air, which is then. What S The Ph Of Pure Water.
From watertechadvice.com
pH of Water Everything You Need To Know What S The Ph Of Pure Water the ph of water. However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. although the ph of pure water is 7, drinking water and natural water. What S The Ph Of Pure Water.
From upberi.com
What to Know about the pH of Drinking Water (2022) What S The Ph Of Pure Water at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. This is because water absorbs carbon dioxide from the air, which is then slowly converted. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink.. What S The Ph Of Pure Water.
From chemguru.sg
How to Calculate pH of Water What S The Ph Of Pure Water All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. simply put the ph of pure water is 7. the ph of water. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink.. What S The Ph Of Pure Water.
From www.tardish2o.co.uk
PH water Levels pH level in Drinking Water What is a safe pH value? What S The Ph Of Pure Water at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. when. What S The Ph Of Pure Water.
From www.sciencephoto.com
PH of Pure Water Stock Image C002/8113 Science Photo Library What S The Ph Of Pure Water although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. simply put the ph of pure water is 7. at 100°c, the ph. What S The Ph Of Pure Water.
From netsolwater.com
What is pH in water, and what is pH in wastewater? Netsol Water What S The Ph Of Pure Water Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7.. What S The Ph Of Pure Water.
From sciencenotes.org
The pH Scale of Common Chemicals What S The Ph Of Pure Water at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. This is because water absorbs carbon dioxide from the air, which is then slowly converted. . What S The Ph Of Pure Water.
From upberi.com
What to Know about the pH of Drinking Water (2022) What S The Ph Of Pure Water although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. This is because water absorbs carbon dioxide from the air, which is then slowly converted. the ph of water. All acids have a ph that is lower than 7 and all bases have a ph that is. What S The Ph Of Pure Water.
From skfelixer.com
The pH Value Of Purified Water — All You Need To Know SKF Elixer What S The Ph Of Pure Water simply put the ph of pure water is 7. All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. when pure water is exposed to air, it becomes mildly acidic. Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a. What S The Ph Of Pure Water.
From www.medicalnewstoday.com
What to know about the pH of water What S The Ph Of Pure Water This is because water absorbs carbon dioxide from the air, which is then slowly converted. However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. when pure water is exposed. What S The Ph Of Pure Water.
From www.toppr.com
The pH of pure water {25}^{o}C and {35}^{o}C are 7 and 6 respectively What S The Ph Of Pure Water although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. when pure water is exposed to air, it becomes mildly acidic. simply. What S The Ph Of Pure Water.
From mywaterearth.com
Safe pH Level for Drinking Water MyWaterEarth&Sky What S The Ph Of Pure Water when pure water is exposed to air, it becomes mildly acidic. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. This is because water absorbs carbon dioxide from the air, which. What S The Ph Of Pure Water.
From www.sliderbase.com
The pH Scale Presentation Chemistry What S The Ph Of Pure Water simply put the ph of pure water is 7. when pure water is exposed to air, it becomes mildly acidic. All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. . What S The Ph Of Pure Water.
From www.slideserve.com
PPT 9.3 Acid Rain PowerPoint Presentation, free download ID2573696 What S The Ph Of Pure Water at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral.. What S The Ph Of Pure Water.
From destrixel.blogspot.com
Ph Level Of Water Alkaline Water 5 Popular Health Benefits of What S The Ph Of Pure Water although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. simply put the ph of pure water is 7. However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). the ph of water. when pure water is. What S The Ph Of Pure Water.
From blog.havells.com
Right pH Level in Drinking Water How Essential? Havells India Blog What S The Ph Of Pure Water This is because water absorbs carbon dioxide from the air, which is then slowly converted. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. simply put the ph of pure water is 7. the ph of water. although the ph of pure water is 7, drinking water and natural water. What S The Ph Of Pure Water.
From www.plumbingsupply.com
Water pH and Your Plumbing What S The Ph Of Pure Water when pure water is exposed to air, it becomes mildly acidic. This is because water absorbs carbon dioxide from the air, which is then slowly converted. Surface water systems typically have a ph range of 6.5 to 8.5, whereas groundwater systems have a ph range of 6 to 8.5. However, most water includes particles that can raise the ph. What S The Ph Of Pure Water.
From news.regenerativemedgroup.com
The pH of water What to know Regenerative Medical Group What S The Ph Of Pure Water However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). when pure water is exposed to air, it becomes mildly acidic. When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. All acids have a ph that is lower than 7 and all bases have. What S The Ph Of Pure Water.
From www.youtube.com
pH Measurement of Pure Water Application Video YouTube What S The Ph Of Pure Water although the ph of pure water is 7, drinking water and natural water exhibits a ph range because it contains dissolved. the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. When you’re measuring the ph of water, a 7.0 reading means that the water. What S The Ph Of Pure Water.
From www.coursehero.com
[Solved] What is the pH of pure water at 40.0°C if the Kw at this What S The Ph Of Pure Water the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. when pure water is exposed to air, it becomes mildly acidic. at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. However, most water. What S The Ph Of Pure Water.
From waterseer.org
What is pH in Water? Definition, Importance & Level Chart What S The Ph Of Pure Water However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). All acids have a ph that is lower than 7 and all bases have a ph that is higher than 7. This is because water absorbs carbon dioxide from the air, which is then slowly converted. at 100°c, the ph of. What S The Ph Of Pure Water.
From contempo3.blogspot.com
Purified Water Di Water Ph What S The Ph Of Pure Water This is because water absorbs carbon dioxide from the air, which is then slowly converted. simply put the ph of pure water is 7. However, most water includes particles that can raise the ph from 6.5 (acidic) to 8.5 (basic or alkaline). When you’re measuring the ph of water, a 7.0 reading means that the water is neutral. Surface. What S The Ph Of Pure Water.
From www.intec-america.com
Things You Must Know About pH Control and Drinking Water Treatment What S The Ph Of Pure Water the ph level of completely pure water is 7, which is exactly in the center of the scale, making it a neutral drink. at 100°c, the ph of pure water is 6.14, which is neutral on the ph scale at this higher temperature. when pure water is exposed to air, it becomes mildly acidic. This is because. What S The Ph Of Pure Water.