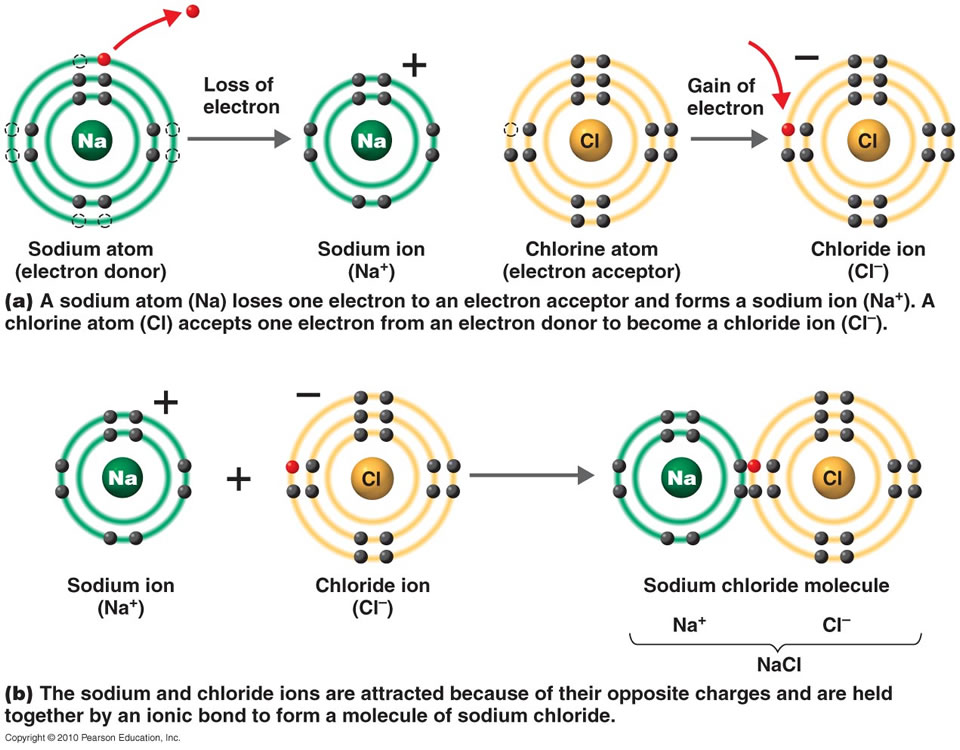
Ionic Bonds Metallic Atoms Tend To . By definition, a metal is relatively stable if it loses. They also tend to have high melting and boiling points,. These elements lie to the left in a. A sheet of aluminum foil and a copper wire are both. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. A simplified model to describe metallic bonding has been developed by paul drüde called the. They also tend to have high melting. many metallic elements have relatively low ionization potentials and lose electrons easily. metallic bonds are formed between two metal atoms. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; ionic solids exhibit a crystalline structure and tend to be rigid and brittle;
from derekcarrsavvy-chemist.blogspot.com
These elements lie to the left in a. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; They also tend to have high melting. They also tend to have high melting and boiling points,. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; many metallic elements have relatively low ionization potentials and lose electrons easily. By definition, a metal is relatively stable if it loses. A sheet of aluminum foil and a copper wire are both. metallic bonds are formed between two metal atoms. A simplified model to describe metallic bonding has been developed by paul drüde called the.
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures
Ionic Bonds Metallic Atoms Tend To These elements lie to the left in a. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. A simplified model to describe metallic bonding has been developed by paul drüde called the. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; They also tend to have high melting. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; many metallic elements have relatively low ionization potentials and lose electrons easily. metallic bonds are formed between two metal atoms. By definition, a metal is relatively stable if it loses. They also tend to have high melting and boiling points,. These elements lie to the left in a. A sheet of aluminum foil and a copper wire are both.
From slideplayer.com
Chapter 9 Chemical Bonds ppt download Ionic Bonds Metallic Atoms Tend To A sheet of aluminum foil and a copper wire are both. many metallic elements have relatively low ionization potentials and lose electrons easily. These elements lie to the left in a. By definition, a metal is relatively stable if it loses. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; A simplified model to. Ionic Bonds Metallic Atoms Tend To.
From fdocuments.in
BONDINGPREVIEW. Bonds Between Atoms Covalent Ionic Polyatomic Ions Ionic Bonds Metallic Atoms Tend To They also tend to have high melting. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. A sheet of aluminum foil and a copper wire are both. A simplified model to describe metallic bonding has. Ionic Bonds Metallic Atoms Tend To.
From www.slideserve.com
PPT Chemical Bonds PowerPoint Presentation, free download ID2962570 Ionic Bonds Metallic Atoms Tend To ionic solids exhibit a crystalline structure and tend to be rigid and brittle; They also tend to have high melting. These elements lie to the left in a. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; A simplified model to describe metallic bonding has been developed by paul drüde called the. They also. Ionic Bonds Metallic Atoms Tend To.
From www.pinterest.com
Metallic Bond A bond formed when metal atoms share electrons. school Ionic Bonds Metallic Atoms Tend To ionic solids exhibit a crystalline structure and tend to be rigid and brittle; They also tend to have high melting and boiling points,. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; By definition, a metal is relatively stable if it loses. many metallic elements have relatively low ionization potentials and lose electrons. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
Chapter 8 Chemical Bonding ppt download Ionic Bonds Metallic Atoms Tend To ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. By definition, a metal is relatively stable if it loses. A simplified model to describe metallic bonding has been developed by paul drüde called the. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; . Ionic Bonds Metallic Atoms Tend To.
From courses.lumenlearning.com
The Building Blocks of Molecules Biology I Ionic Bonds Metallic Atoms Tend To ionic solids exhibit a crystalline structure and tend to be rigid and brittle; many metallic elements have relatively low ionization potentials and lose electrons easily. These elements lie to the left in a. A sheet of aluminum foil and a copper wire are both. By definition, a metal is relatively stable if it loses. metallic bonds are. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
Chapter 7 Ionic and Metallic Bonding 7.1 Ions 7.2 Ionic Bonds and ppt Ionic Bonds Metallic Atoms Tend To They also tend to have high melting and boiling points,. many metallic elements have relatively low ionization potentials and lose electrons easily. By definition, a metal is relatively stable if it loses. These elements lie to the left in a. A simplified model to describe metallic bonding has been developed by paul drüde called the. metallic bonds are. Ionic Bonds Metallic Atoms Tend To.
From revisechemistry.uk
Chemical Bonds, Ionic, Covalent and Metallic AQA C2 revisechemistry.uk Ionic Bonds Metallic Atoms Tend To ionic solids exhibit a crystalline structure and tend to be rigid and brittle; A sheet of aluminum foil and a copper wire are both. These elements lie to the left in a. They also tend to have high melting and boiling points,. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; metallic bonds. Ionic Bonds Metallic Atoms Tend To.
From derekcarrsavvy-chemist.blogspot.com
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures Ionic Bonds Metallic Atoms Tend To metallic bonds are formed between two metal atoms. They also tend to have high melting. They also tend to have high melting and boiling points,. A simplified model to describe metallic bonding has been developed by paul drüde called the. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds. Ionic Bonds Metallic Atoms Tend To.
From chem.libretexts.org
Ionic Solids Chemistry LibreTexts Ionic Bonds Metallic Atoms Tend To A sheet of aluminum foil and a copper wire are both. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; These elements lie to the left in a. metallic bonds are formed between two. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
Ionic/Covalent/Metallic Bonding Notes ppt download Ionic Bonds Metallic Atoms Tend To ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. metallic bonds are formed between two metal atoms. A simplified model to describe metallic bonding has been developed by paul drüde called the. By definition, a metal is relatively stable if it loses. They also tend to have high. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
Ionic Bonding Ch 8 CVHS Chemistry ppt download Ionic Bonds Metallic Atoms Tend To metallic bonds are formed between two metal atoms. They also tend to have high melting. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; These elements lie to the left in a. They also tend to have high melting and boiling points,. By definition, a metal is relatively stable if it loses. A simplified. Ionic Bonds Metallic Atoms Tend To.
From en.wikipedia.org
Ionic bonding Wikipedia Ionic Bonds Metallic Atoms Tend To ionic solids exhibit a crystalline structure and tend to be rigid and brittle; A simplified model to describe metallic bonding has been developed by paul drüde called the. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; metallic bonds are formed between two metal atoms. A sheet of aluminum foil and a copper. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
Ionic/Covalent/Metallic Bonding Notes ppt download Ionic Bonds Metallic Atoms Tend To By definition, a metal is relatively stable if it loses. A sheet of aluminum foil and a copper wire are both. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. many metallic elements have relatively low ionization potentials and lose electrons easily. They also tend to have high. Ionic Bonds Metallic Atoms Tend To.
From slidetodoc.com
Chemical Bonding Chemical Bonds When atoms of different Ionic Bonds Metallic Atoms Tend To ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; metallic bonds are formed between two metal atoms. A sheet of aluminum foil and a copper wire are both. A simplified model to describe metallic. Ionic Bonds Metallic Atoms Tend To.
From www.britannica.com
chemical bonding Ionic and covalent compounds Britannica Ionic Bonds Metallic Atoms Tend To A sheet of aluminum foil and a copper wire are both. A simplified model to describe metallic bonding has been developed by paul drüde called the. They also tend to have high melting and boiling points,. metallic bonds are formed between two metal atoms. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best. Ionic Bonds Metallic Atoms Tend To.
From www2.victoriacollege.edu
Chapter 2 Atoms, Molecules and Life Chemistry) Ionic Bonds Metallic Atoms Tend To ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. many metallic elements have relatively low ionization potentials and lose electrons easily. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; A simplified model to describe metallic bonding has been developed by paul drüde. Ionic Bonds Metallic Atoms Tend To.
From derekcarrsavvy-chemist.blogspot.co.uk
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures Ionic Bonds Metallic Atoms Tend To By definition, a metal is relatively stable if it loses. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. A sheet of aluminum foil and a copper wire are both. many metallic elements have relatively low ionization potentials and lose electrons easily. ionic solids exhibit a crystalline. Ionic Bonds Metallic Atoms Tend To.
From www.youtube.com
Examples of Ionic Compoiunds YouTube Ionic Bonds Metallic Atoms Tend To A simplified model to describe metallic bonding has been developed by paul drüde called the. metallic bonds are formed between two metal atoms. By definition, a metal is relatively stable if it loses. many metallic elements have relatively low ionization potentials and lose electrons easily. ionic solids exhibit a crystalline structure and tend to be rigid and. Ionic Bonds Metallic Atoms Tend To.
From www.tec-science.com
Ionic bonding tecscience Ionic Bonds Metallic Atoms Tend To many metallic elements have relatively low ionization potentials and lose electrons easily. A sheet of aluminum foil and a copper wire are both. They also tend to have high melting. A simplified model to describe metallic bonding has been developed by paul drüde called the. metallic bonds are formed between two metal atoms. ionic solids exhibit a. Ionic Bonds Metallic Atoms Tend To.
From slidetodoc.com
IONIC BONDING THE IONIC BOND Ionic bonds tend Ionic Bonds Metallic Atoms Tend To A sheet of aluminum foil and a copper wire are both. A simplified model to describe metallic bonding has been developed by paul drüde called the. By definition, a metal is relatively stable if it loses. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. They also tend to. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
Structure of Atoms. ppt download Ionic Bonds Metallic Atoms Tend To A sheet of aluminum foil and a copper wire are both. metallic bonds are formed between two metal atoms. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. These elements lie to the left. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
When a chemical bond is broken, energy is ppt download Ionic Bonds Metallic Atoms Tend To A simplified model to describe metallic bonding has been developed by paul drüde called the. They also tend to have high melting. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. These elements lie to the left in a. A sheet of aluminum foil and a copper wire are. Ionic Bonds Metallic Atoms Tend To.
From slidetodoc.com
BONDING CONTENTS Introduction Chemical and physical bonding Ionic Ionic Bonds Metallic Atoms Tend To ionic solids exhibit a crystalline structure and tend to be rigid and brittle; A simplified model to describe metallic bonding has been developed by paul drüde called the. A sheet of aluminum foil and a copper wire are both. They also tend to have high melting and boiling points,. many metallic elements have relatively low ionization potentials and. Ionic Bonds Metallic Atoms Tend To.
From www.youtube.com
Types of Bonding (Ionic, Covalent, Metallic) GCSE Chemistry Revision Ionic Bonds Metallic Atoms Tend To These elements lie to the left in a. A sheet of aluminum foil and a copper wire are both. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. A simplified model to describe metallic bonding has been developed by paul drüde called the. metallic bonds are formed between. Ionic Bonds Metallic Atoms Tend To.
From www.chemistrylearner.com
Ionic, Covalent, and Metallic Bonds Differences and Similarities Ionic Bonds Metallic Atoms Tend To ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; By definition, a metal is relatively stable if it loses. They also tend to have high melting and boiling points,. many metallic elements have relatively. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
Comparison of Properties Ionic Compounds Covalent Compounds Metals Ionic Bonds Metallic Atoms Tend To They also tend to have high melting. These elements lie to the left in a. By definition, a metal is relatively stable if it loses. many metallic elements have relatively low ionization potentials and lose electrons easily. A sheet of aluminum foil and a copper wire are both. ionic solids exhibit a crystalline structure and tend to be. Ionic Bonds Metallic Atoms Tend To.
From www.sliderbase.com
Ionic Bonding Presentation Chemistry Ionic Bonds Metallic Atoms Tend To ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. By definition, a metal is relatively stable if it loses. A sheet of aluminum foil and a copper wire are both. They also tend to have high melting and boiling points,. ionic solids exhibit a crystalline structure and tend. Ionic Bonds Metallic Atoms Tend To.
From slidetodoc.com
Metallic Bonds 2 a Students know atoms combine Ionic Bonds Metallic Atoms Tend To These elements lie to the left in a. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. many metallic elements have relatively low ionization potentials and lose electrons easily. They also tend to have. Ionic Bonds Metallic Atoms Tend To.
From www.britannica.com
Metallic bond Properties, Examples, & Explanation Britannica Ionic Bonds Metallic Atoms Tend To ionic solids exhibit a crystalline structure and tend to be rigid and brittle; A sheet of aluminum foil and a copper wire are both. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; metallic bonds are formed between two metal atoms. By definition, a metal is relatively stable if it loses. They also. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
Section 2 Ionic and Covalent Bonding ppt download Ionic Bonds Metallic Atoms Tend To metallic bonds are formed between two metal atoms. These elements lie to the left in a. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; A sheet of aluminum foil and a copper wire are both. They also tend to have high melting and boiling points,. many metallic elements have relatively low ionization. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
Ionic & Metallic Bonding ppt download Ionic Bonds Metallic Atoms Tend To They also tend to have high melting and boiling points,. metallic bonds are formed between two metal atoms. These elements lie to the left in a. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. A simplified model to describe metallic bonding has been developed by paul drüde. Ionic Bonds Metallic Atoms Tend To.
From slidetodoc.com
Chemical Bonds Three basic types Ionic Electrostatic attraction Ionic Bonds Metallic Atoms Tend To They also tend to have high melting and boiling points,. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. many metallic elements have relatively low ionization potentials and lose electrons easily. They also tend to have high melting. ionic solids exhibit a crystalline structure and tend to. Ionic Bonds Metallic Atoms Tend To.
From stewart-switch.com
Ionic Compound Diagram Ionic Bonds Metallic Atoms Tend To ionic solids exhibit a crystalline structure and tend to be rigid and brittle; These elements lie to the left in a. ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed. ionic solids exhibit a crystalline structure and tend to be rigid and brittle; A sheet of aluminum. Ionic Bonds Metallic Atoms Tend To.
From slideplayer.com
CHAPTER 6 THE CHEMISTRY IN BIOLOGY ppt download Ionic Bonds Metallic Atoms Tend To They also tend to have high melting. A simplified model to describe metallic bonding has been developed by paul drüde called the. They also tend to have high melting and boiling points,. These elements lie to the left in a. A sheet of aluminum foil and a copper wire are both. ionic solids exhibit a crystalline structure and tend. Ionic Bonds Metallic Atoms Tend To.