Are Aromatics Polar Or Nonpolar . There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. Obviously, it is possible to have nonpolar. four key rules for aromaticity. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. Bond polarity and molecular polarity are different (though related) concepts. In short the molecular dipole moment is the vector. understanding the distinction between the two types of molecules is crucial as it influences. when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar.
from www.wou.edu
when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar. four key rules for aromaticity. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. Obviously, it is possible to have nonpolar. In short the molecular dipole moment is the vector. Bond polarity and molecular polarity are different (though related) concepts. understanding the distinction between the two types of molecules is crucial as it influences. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use.
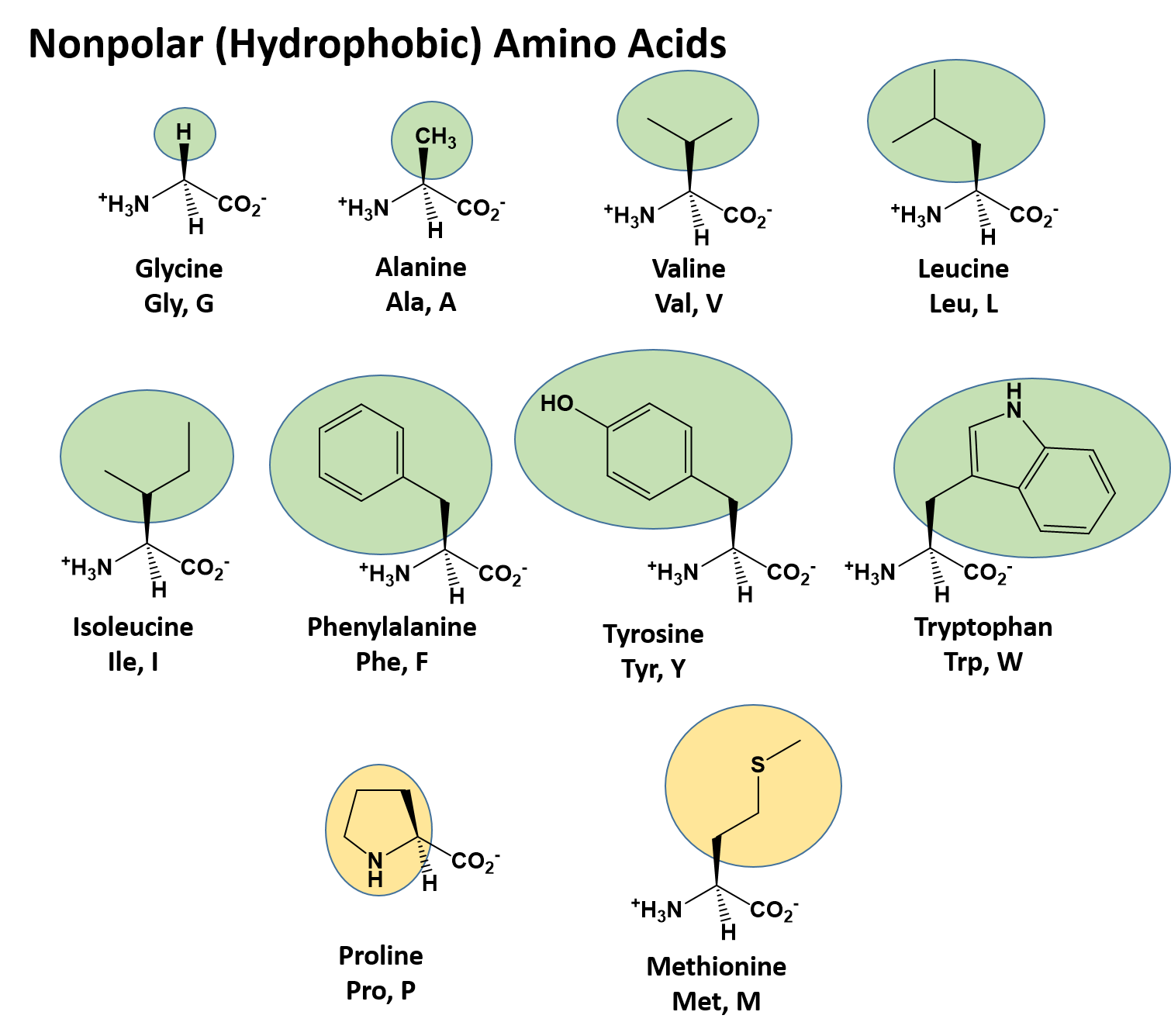
CH103 Chapter 8 The Major Macromolecules Chemistry
Are Aromatics Polar Or Nonpolar four key rules for aromaticity. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. In short the molecular dipole moment is the vector. Bond polarity and molecular polarity are different (though related) concepts. when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar. understanding the distinction between the two types of molecules is crucial as it influences. four key rules for aromaticity. Obviously, it is possible to have nonpolar.
From www.semanticscholar.org
Table 1 from Sorption of polar and nonpolar aromatic organic contaminants by plant cuticular Are Aromatics Polar Or Nonpolar understanding the distinction between the two types of molecules is crucial as it influences. Bond polarity and molecular polarity are different (though related) concepts. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. here are examples of polar and nonpolar molecules, a look at how polarity. Are Aromatics Polar Or Nonpolar.
From www.numerade.com
SOLVED Draw the structures of the following amino acids at physiological pH and classify them Are Aromatics Polar Or Nonpolar here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. Obviously, it is possible to have nonpolar. understanding the distinction between the two types of molecules is crucial as it influences. Bond polarity and molecular polarity are different (though related) concepts. aromatic hydrocarbons. Are Aromatics Polar Or Nonpolar.
From nursehub.com
Understanding Types of Chemical Bonds NurseHub Are Aromatics Polar Or Nonpolar In short the molecular dipole moment is the vector. understanding the distinction between the two types of molecules is crucial as it influences. when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar. Bond polarity and molecular polarity are different (though related) concepts. aromatic hydrocarbons have a multiple, conjugated. Are Aromatics Polar Or Nonpolar.
From www.slideshare.net
Bioc 523 Are Aromatics Polar Or Nonpolar here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to. Are Aromatics Polar Or Nonpolar.
From www.slideserve.com
PPT Amino Acids and Proteins PowerPoint Presentation, free download ID3570650 Are Aromatics Polar Or Nonpolar Bond polarity and molecular polarity are different (though related) concepts. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. four key rules for aromaticity. understanding the distinction between. Are Aromatics Polar Or Nonpolar.
From www.wou.edu
CH103 Chapter 8 The Major Macromolecules Chemistry Are Aromatics Polar Or Nonpolar In short the molecular dipole moment is the vector. four key rules for aromaticity. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. Obviously, it is possible to have. Are Aromatics Polar Or Nonpolar.
From www.coscinecreative.com
How Do You Teach Polar Vs. Nonpolar Molecules? — CoScine Creative Are Aromatics Polar Or Nonpolar understanding the distinction between the two types of molecules is crucial as it influences. In short the molecular dipole moment is the vector. four key rules for aromaticity. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. There turn out to be. Are Aromatics Polar Or Nonpolar.
From askfilo.com
Aromatic hydrocarbons are non polar hydrogen atom molecules and are usua.. Are Aromatics Polar Or Nonpolar here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. In short the molecular dipole moment is the vector. Obviously, it is possible to have nonpolar. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient. Are Aromatics Polar Or Nonpolar.
From www.masterorganicchemistry.com
Rules for Aromaticity The 4 Key Factors Master Organic Chemistry Are Aromatics Polar Or Nonpolar four key rules for aromaticity. Bond polarity and molecular polarity are different (though related) concepts. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. understanding the distinction between the two types of molecules is crucial as it influences. Obviously, it is possible to have nonpolar. . Are Aromatics Polar Or Nonpolar.
From www.chegg.com
Solved Indicate whether each of the following amino acids is Are Aromatics Polar Or Nonpolar aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. understanding the distinction between the two types of molecules is crucial as it influences. here are examples of polar. Are Aromatics Polar Or Nonpolar.
From www.researchgate.net
Structural formulae of representative polar polycyclic aromatic... Download Scientific Diagram Are Aromatics Polar Or Nonpolar here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. Bond polarity and molecular polarity are different (though related) concepts. four key rules for aromaticity. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient. Are Aromatics Polar Or Nonpolar.
From www.mdpi.com
Molecules Free FullText Hydration Thermodynamics of NonPolar Aromatic Hydrocarbons Are Aromatics Polar Or Nonpolar here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. In short the molecular dipole moment is the vector. understanding the distinction between the two types. Are Aromatics Polar Or Nonpolar.
From school.careers360.com
difference between polar and non polar Overview, Structure, Properties & Uses Are Aromatics Polar Or Nonpolar four key rules for aromaticity. Bond polarity and molecular polarity are different (though related) concepts. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. understanding the distinction between. Are Aromatics Polar Or Nonpolar.
From www.chegg.com
Solved pick one amino acid from each chemical Are Aromatics Polar Or Nonpolar aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. In short the molecular dipole moment is the vector. understanding the distinction between the two types of molecules is crucial as it influences. when its value is zero, the molecule is said to be nonpolar, otherwise it’s. Are Aromatics Polar Or Nonpolar.
From chemwiki.ucdavis.edu
24.1 Functional Groups and Classes of Organic Compounds Chemwiki Are Aromatics Polar Or Nonpolar Bond polarity and molecular polarity are different (though related) concepts. when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. Obviously, it is possible to have. Are Aromatics Polar Or Nonpolar.
From www.numerade.com
SOLVED e. Determine whether the side chain of the amino acid is a nonpolar aliphatic; aromatic Are Aromatics Polar Or Nonpolar Obviously, it is possible to have nonpolar. four key rules for aromaticity. when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. understanding the. Are Aromatics Polar Or Nonpolar.
From www.slideserve.com
PPT Polar or Nonpolar PowerPoint Presentation, free download ID3483667 Are Aromatics Polar Or Nonpolar here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. Obviously, it is possible to have nonpolar. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. four key rules for aromaticity. In. Are Aromatics Polar Or Nonpolar.
From www.slideserve.com
PPT MEDI12042011SU PowerPoint Presentation, free download ID2031289 Are Aromatics Polar Or Nonpolar Bond polarity and molecular polarity are different (though related) concepts. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. Obviously, it is possible to have nonpolar. In short the molecular dipole moment is the vector. here are examples of polar and nonpolar molecules, a look at how. Are Aromatics Polar Or Nonpolar.
From www.numerade.com
SOLVED Text Select the most appropriate classification for the amino acid that is shown. H 1 Are Aromatics Polar Or Nonpolar There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and. Are Aromatics Polar Or Nonpolar.
From www.researchgate.net
1D NMR spectra of MK1 in polar and nonpolar solvents. Lines through... Download Scientific Are Aromatics Polar Or Nonpolar understanding the distinction between the two types of molecules is crucial as it influences. four key rules for aromaticity. Obviously, it is possible to have nonpolar. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. aromatic hydrocarbons have a multiple, conjugated. Are Aromatics Polar Or Nonpolar.
From mcatmastery.net
Amino Acids Structure on the MCAT MCAT Mastery Are Aromatics Polar Or Nonpolar Obviously, it is possible to have nonpolar. Bond polarity and molecular polarity are different (though related) concepts. four key rules for aromaticity. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent. Are Aromatics Polar Or Nonpolar.
From nznano.blogspot.com
NZ Nano Polar aromatic molecules Are Aromatics Polar Or Nonpolar There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. understanding the distinction between the two types of molecules is crucial as it influences. when. Are Aromatics Polar Or Nonpolar.
From byjus.com
Aromatic Compounds Questions Practice Questions of Aromatic Compounds with Answer & Explanations Are Aromatics Polar Or Nonpolar understanding the distinction between the two types of molecules is crucial as it influences. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. four key rules for aromaticity. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this. Are Aromatics Polar Or Nonpolar.
From dxorlaiph.blob.core.windows.net
Amino Acids Draw Structures at Violet Lane blog Are Aromatics Polar Or Nonpolar when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. Obviously, it is possible to have nonpolar. four key rules for aromaticity. aromatic hydrocarbons. Are Aromatics Polar Or Nonpolar.
From www.masterorganicchemistry.com
Rules for Aromaticity The 4 Key Factors Master Organic Chemistry Are Aromatics Polar Or Nonpolar There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. In short the molecular dipole moment is the vector. when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar. here are examples of polar and nonpolar molecules, a look at how polarity. Are Aromatics Polar Or Nonpolar.
From chemistry.stackexchange.com
biochemistry Why are Tyrosine and Tryptophan considered hydrophobic? Chemistry Stack Exchange Are Aromatics Polar Or Nonpolar Bond polarity and molecular polarity are different (though related) concepts. Obviously, it is possible to have nonpolar. In short the molecular dipole moment is the vector. when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar. here are examples of polar and nonpolar molecules, a look at how polarity relates. Are Aromatics Polar Or Nonpolar.
From testpubschina.acs.org
Solubility of Polar and Nonpolar Aromatic Molecules in Subcritical Water The Role of the Are Aromatics Polar Or Nonpolar Obviously, it is possible to have nonpolar. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. Bond polarity and molecular polarity are different (though related) concepts. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic.. Are Aromatics Polar Or Nonpolar.
From www.lecturio.com
Basics of Amino Acids Concise Medical Knowledge Are Aromatics Polar Or Nonpolar In short the molecular dipole moment is the vector. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. four key rules for aromaticity. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. when its value is zero,. Are Aromatics Polar Or Nonpolar.
From www.difference101.com
Aromatic vs. Aliphatic Compounds 5 Key Differences, Pros & Cons, Examples Difference 101 Are Aromatics Polar Or Nonpolar In short the molecular dipole moment is the vector. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. when its value is zero, the molecule is said to be nonpolar, otherwise it’s said to be polar. Obviously, it is possible to have nonpolar. here are examples. Are Aromatics Polar Or Nonpolar.
From www.differencebetween.com
Difference Between Polar and Nonpolar Amino Acids Compare the Difference Between Similar Terms Are Aromatics Polar Or Nonpolar here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. understanding the distinction between the two types of molecules is crucial as it influences. Bond polarity. Are Aromatics Polar Or Nonpolar.
From www.slideserve.com
PPT Amino Acids and Peptides PowerPoint Presentation, free download ID6333061 Are Aromatics Polar Or Nonpolar aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. Bond polarity and molecular polarity are different (though related) concepts. In short the molecular dipole moment is the vector. understanding the distinction between the two types of molecules is crucial as it influences. here are examples of. Are Aromatics Polar Or Nonpolar.
From cwsimons.com
Structure of Amino Acids and Proteins Are Aromatics Polar Or Nonpolar understanding the distinction between the two types of molecules is crucial as it influences. Bond polarity and molecular polarity are different (though related) concepts. There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. four key rules for aromaticity. when its value is zero, the molecule is said to. Are Aromatics Polar Or Nonpolar.
From www.semanticscholar.org
Figure 3 from Sorption of polar and nonpolar aromatic compounds to four surface soils of eastern Are Aromatics Polar Or Nonpolar In short the molecular dipole moment is the vector. here are examples of polar and nonpolar molecules, a look at how polarity relates to ionic and covalent bonds, and how you can use. Bond polarity and molecular polarity are different (though related) concepts. understanding the distinction between the two types of molecules is crucial as it influences. . Are Aromatics Polar Or Nonpolar.
From www.slideserve.com
PPT Lecture 2 Eukaryotic cells, amino acids PowerPoint Presentation ID448531 Are Aromatics Polar Or Nonpolar There turn out to be 4 conditions a molecule must meet in order for it to be aromatic. Obviously, it is possible to have nonpolar. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. understanding the distinction between the two types of molecules is crucial as it. Are Aromatics Polar Or Nonpolar.
From sciencenotes.org
Polar and Nonpolar Molecules Are Aromatics Polar Or Nonpolar In short the molecular dipole moment is the vector. aromatic hydrocarbons have a multiple, conjugated c=c with pi electron system, but this does not create a sufficient dipole for. understanding the distinction between the two types of molecules is crucial as it influences. four key rules for aromaticity. when its value is zero, the molecule is. Are Aromatics Polar Or Nonpolar.