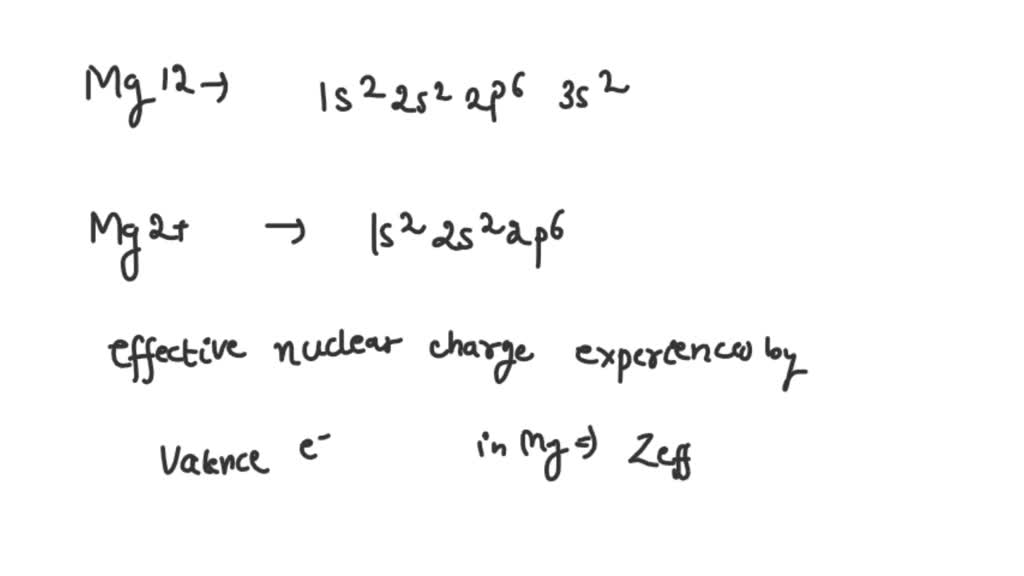Charge Of Magnesium Ion That Has 10 Electrons . When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its neutral state. There are four ways to find the charge of an element: 93 rows this table shows the most common charges for atoms of the chemical elements. The usual charge of an element is common to its group. See examples of cations and. This electric charge generated on the ion is. 93 rows ionic charge: Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms. You can use this table to predict whether an atom can bond with another atom. See examples of ion formation and composition for magnesium and nitrogen. Find out how to use the atomic number, the valence shell and the periodic table to determine the. Learn how atoms lose or gain electrons to become ions with different charges.
from www.numerade.com
See examples of cations and. The usual charge of an element is common to its group. When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. This electric charge generated on the ion is. 93 rows ionic charge: The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its neutral state. Find out how to use the atomic number, the valence shell and the periodic table to determine the. Learn how atoms lose or gain electrons to become ions with different charges. See examples of ion formation and composition for magnesium and nitrogen.
SOLVED Write the full electronic configurations of both Mg and Mg2
Charge Of Magnesium Ion That Has 10 Electrons Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. Find out how to use the atomic number, the valence shell and the periodic table to determine the. Learn how atoms lose or gain electrons to become ions with different charges. The usual charge of an element is common to its group. 93 rows ionic charge: When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). See examples of ion formation and composition for magnesium and nitrogen. There are four ways to find the charge of an element: The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its neutral state. 93 rows this table shows the most common charges for atoms of the chemical elements. This electric charge generated on the ion is. See examples of cations and. You can use this table to predict whether an atom can bond with another atom. Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms.
From www.animalia-life.club
Magnesium Electron Configuration Charge Of Magnesium Ion That Has 10 Electrons You can use this table to predict whether an atom can bond with another atom. When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). Find out how to use the atomic number, the valence shell and the periodic table to determine the. See examples of cations and. Learn how. Charge Of Magnesium Ion That Has 10 Electrons.
From slideplayer.com
What are POLYATOMIC IONS? ppt download Charge Of Magnesium Ion That Has 10 Electrons See examples of cations and. 93 rows ionic charge: You can use this table to predict whether an atom can bond with another atom. Find out how to use the atomic number, the valence shell and the periodic table to determine the. The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons. Charge Of Magnesium Ion That Has 10 Electrons.
From www.pinterest.co.uk
Magnesium, atomic structure Stock Image C018/3693 Science Photo Charge Of Magnesium Ion That Has 10 Electrons Learn how atoms lose or gain electrons to become ions with different charges. Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms. Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. The charge on a magnesium ion with ten electrons would be. Charge Of Magnesium Ion That Has 10 Electrons.
From www.slideserve.com
PPT KS4 Chemistry PowerPoint Presentation, free download ID1787201 Charge Of Magnesium Ion That Has 10 Electrons Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. The usual charge of an element is common to its group. This electric charge generated on the ion is. There are four ways to find the charge of an element: See examples of ion formation and composition for magnesium and nitrogen. Find. Charge Of Magnesium Ion That Has 10 Electrons.
From slideplayer.com
Chemistry Lesson 8 Ions ppt download Charge Of Magnesium Ion That Has 10 Electrons Learn how atoms lose or gain electrons to become ions with different charges. 93 rows ionic charge: When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). The usual charge of an element is common to its group. 93 rows this table shows the most common charges for atoms of. Charge Of Magnesium Ion That Has 10 Electrons.
From valenceelectrons.com
How Many Protons,Neutrons and Electrons Does Magnesium Have? Charge Of Magnesium Ion That Has 10 Electrons Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms. This electric charge generated on the ion is. 93 rows ionic charge: See examples of ion formation and composition for magnesium and nitrogen. There are four ways to find the charge of an element: Find out how to use the atomic number,. Charge Of Magnesium Ion That Has 10 Electrons.
From www.dreamstime.com
Model of magnesium atom stock vector. Illustration of mass 164475021 Charge Of Magnesium Ion That Has 10 Electrons Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). Find out how to use the atomic number, the valence shell and the periodic table to determine the. See examples of ion. Charge Of Magnesium Ion That Has 10 Electrons.
From www.animalia-life.club
Magnesium Electron Configuration Charge Of Magnesium Ion That Has 10 Electrons Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms. Find out how to use the atomic number, the valence shell and the periodic table to determine the. There are four ways to find the charge of an element: See examples of cations and. This electric charge generated on the ion is.. Charge Of Magnesium Ion That Has 10 Electrons.
From www.slideserve.com
PPT How do atoms form ions? PowerPoint Presentation, free download Charge Of Magnesium Ion That Has 10 Electrons Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. See examples of ion formation and composition for magnesium and nitrogen. The usual charge of an element is common to its group. The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its. Charge Of Magnesium Ion That Has 10 Electrons.
From socratic.org
Question 6b48e + Example Charge Of Magnesium Ion That Has 10 Electrons Find out how to use the atomic number, the valence shell and the periodic table to determine the. There are four ways to find the charge of an element: The usual charge of an element is common to its group. The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its. Charge Of Magnesium Ion That Has 10 Electrons.
From www.slideshare.net
The periodic table Charge Of Magnesium Ion That Has 10 Electrons There are four ways to find the charge of an element: The usual charge of an element is common to its group. The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its neutral state. 93 rows this table shows the most common charges for atoms of the chemical elements. Learn. Charge Of Magnesium Ion That Has 10 Electrons.
From www.alamy.com
Symbol and electron diagram for Magnesium illustration Stock Vector Charge Of Magnesium Ion That Has 10 Electrons The usual charge of an element is common to its group. You can use this table to predict whether an atom can bond with another atom. When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). 93 rows ionic charge: Learn how to name and write the charge of monatomic. Charge Of Magnesium Ion That Has 10 Electrons.
From brainly.com
What is the charge on a magnesium ion? How do they get that charge Charge Of Magnesium Ion That Has 10 Electrons Find out how to use the atomic number, the valence shell and the periodic table to determine the. See examples of cations and. 93 rows ionic charge: The usual charge of an element is common to its group. 93 rows this table shows the most common charges for atoms of the chemical elements. Learn how to use the periodic table. Charge Of Magnesium Ion That Has 10 Electrons.
From commons.wikimedia.org
FileElectron shell 012 magnesium.png Wikimedia Commons Charge Of Magnesium Ion That Has 10 Electrons There are four ways to find the charge of an element: When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). See examples of ion formation and composition for magnesium and nitrogen. The usual charge of an element is common to its group. Learn how to use the periodic table. Charge Of Magnesium Ion That Has 10 Electrons.
From valenceelectrons.com
How to Write the Electron Configuration for Magnesium (Mg)? Charge Of Magnesium Ion That Has 10 Electrons Find out how to use the atomic number, the valence shell and the periodic table to determine the. This electric charge generated on the ion is. When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). You can use this table to predict whether an atom can bond with another. Charge Of Magnesium Ion That Has 10 Electrons.
From www.newtondesk.com
magnesium electron configuration Newton Desk Charge Of Magnesium Ion That Has 10 Electrons This electric charge generated on the ion is. Find out how to use the atomic number, the valence shell and the periodic table to determine the. Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. 93 rows ionic charge: Learn how atoms lose or gain electrons to become ions with different. Charge Of Magnesium Ion That Has 10 Electrons.
From material-properties.org
Magnesium Periodic Table and Atomic Properties Charge Of Magnesium Ion That Has 10 Electrons Learn how atoms lose or gain electrons to become ions with different charges. The usual charge of an element is common to its group. See examples of cations and. There are four ways to find the charge of an element: You can use this table to predict whether an atom can bond with another atom. The charge on a magnesium. Charge Of Magnesium Ion That Has 10 Electrons.
From www.youtube.com
How to Find the Ionic Charge for Magnesium (Mg) YouTube Charge Of Magnesium Ion That Has 10 Electrons There are four ways to find the charge of an element: Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its neutral state. This electric charge generated on the ion is. See. Charge Of Magnesium Ion That Has 10 Electrons.
From slideplayer.com
“My name is Bond, Ionic Bond; taken, not shared!” ppt download Charge Of Magnesium Ion That Has 10 Electrons 93 rows ionic charge: This electric charge generated on the ion is. 93 rows this table shows the most common charges for atoms of the chemical elements. When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). Learn how atoms lose or gain electrons to become ions with different charges.. Charge Of Magnesium Ion That Has 10 Electrons.
From slideplayer.com
“My name is Bond, Ionic Bond; taken, not shared!” ppt download Charge Of Magnesium Ion That Has 10 Electrons The usual charge of an element is common to its group. 93 rows ionic charge: There are four ways to find the charge of an element: This electric charge generated on the ion is. 93 rows this table shows the most common charges for atoms of the chemical elements. Find out how to use the atomic number, the valence shell. Charge Of Magnesium Ion That Has 10 Electrons.
From slidetodoc.com
Metal ions Nonmetal ions Positive ion Gain electrons Charge Of Magnesium Ion That Has 10 Electrons See examples of cations and. See examples of ion formation and composition for magnesium and nitrogen. 93 rows this table shows the most common charges for atoms of the chemical elements. You can use this table to predict whether an atom can bond with another atom. Learn how atoms lose or gain electrons to become ions with different charges. The. Charge Of Magnesium Ion That Has 10 Electrons.
From periodictable.me
Magnesium Electron Configuration (Mg) with Orbital Diagram Charge Of Magnesium Ion That Has 10 Electrons Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms. There are four ways to find the charge of an element: This electric charge generated on the ion is. See examples of cations and.. Charge Of Magnesium Ion That Has 10 Electrons.
From quizlet.com
Magnesium Oxide Diagram Quizlet Charge Of Magnesium Ion That Has 10 Electrons The usual charge of an element is common to its group. See examples of ion formation and composition for magnesium and nitrogen. Find out how to use the atomic number, the valence shell and the periodic table to determine the. Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. See examples. Charge Of Magnesium Ion That Has 10 Electrons.
From ar.inspiredpencil.com
Magnesium Atom Structure Charge Of Magnesium Ion That Has 10 Electrons See examples of ion formation and composition for magnesium and nitrogen. This electric charge generated on the ion is. See examples of cations and. Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. Find out how to use the atomic number, the valence shell and the periodic table to determine the.. Charge Of Magnesium Ion That Has 10 Electrons.
From slidetodoc.com
Ions Alchemy Lesson V Chemistry Opener Chem Survey Charge Of Magnesium Ion That Has 10 Electrons Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms. Find out how to use the atomic number, the valence shell and the periodic table to determine the. You can use this table to predict whether an atom can bond with another atom. The usual charge of an element is common to. Charge Of Magnesium Ion That Has 10 Electrons.
From www.animalia-life.club
Magnesium Electron Configuration Charge Of Magnesium Ion That Has 10 Electrons You can use this table to predict whether an atom can bond with another atom. 93 rows this table shows the most common charges for atoms of the chemical elements. Find out how to use the atomic number, the valence shell and the periodic table to determine the. See examples of cations and. When the atom loses or gains one. Charge Of Magnesium Ion That Has 10 Electrons.
From www.pinterest.se
GensonScience Magnesium Atom model project, Atom model, Atoms and Charge Of Magnesium Ion That Has 10 Electrons There are four ways to find the charge of an element: See examples of cations and. When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed). You can use this table to predict whether an atom can bond with another atom. This electric charge generated on the ion is. The. Charge Of Magnesium Ion That Has 10 Electrons.
From gageferscase.blogspot.com
9 Protons 10 Neutrons 10 Electrons Total Charge Charge Of Magnesium Ion That Has 10 Electrons Learn how atoms lose or gain electrons to become ions with different charges. Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. You can use this table to predict whether an atom can bond with another atom. See examples of ion formation and composition for magnesium and nitrogen. Learn how to. Charge Of Magnesium Ion That Has 10 Electrons.
From www.nemoquiz.com
Ion Charge from Periodic Table NemoQuiz Charge Of Magnesium Ion That Has 10 Electrons Learn how to use the periodic table to predict the charge of ions formed by metals and nonmetals. This electric charge generated on the ion is. The usual charge of an element is common to its group. Find out how to use the atomic number, the valence shell and the periodic table to determine the. The charge on a magnesium. Charge Of Magnesium Ion That Has 10 Electrons.
From slideplayer.com
AS Chemistry Homework Periodicity ppt download Charge Of Magnesium Ion That Has 10 Electrons Find out how to use the atomic number, the valence shell and the periodic table to determine the. You can use this table to predict whether an atom can bond with another atom. The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its neutral state. Learn how to name and. Charge Of Magnesium Ion That Has 10 Electrons.
From enginedatanichered.z21.web.core.windows.net
Atomic Diagram Of Magnesium Charge Of Magnesium Ion That Has 10 Electrons 93 rows this table shows the most common charges for atoms of the chemical elements. This electric charge generated on the ion is. You can use this table to predict whether an atom can bond with another atom. The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its neutral state.. Charge Of Magnesium Ion That Has 10 Electrons.
From guidelibstoitering.z21.web.core.windows.net
Magnesium Electron Dot Diagram Charge Of Magnesium Ion That Has 10 Electrons See examples of ion formation and composition for magnesium and nitrogen. There are four ways to find the charge of an element: This electric charge generated on the ion is. 93 rows ionic charge: Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms. Find out how to use the atomic number,. Charge Of Magnesium Ion That Has 10 Electrons.
From www.animalia-life.club
Magnesium Electron Configuration Charge Of Magnesium Ion That Has 10 Electrons Learn how atoms lose or gain electrons to become ions with different charges. There are four ways to find the charge of an element: This electric charge generated on the ion is. 93 rows this table shows the most common charges for atoms of the chemical elements. The charge on a magnesium ion with ten electrons would be +2, since. Charge Of Magnesium Ion That Has 10 Electrons.
From www.numerade.com
SOLVED Write the full electronic configurations of both Mg and Mg2 Charge Of Magnesium Ion That Has 10 Electrons Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms. This electric charge generated on the ion is. The charge on a magnesium ion with ten electrons would be +2, since magnesium typically has 12 electrons in its neutral state. 93 rows this table shows the most common charges for atoms of. Charge Of Magnesium Ion That Has 10 Electrons.
From www.slideserve.com
PPT The Octet Rule PowerPoint Presentation, free download ID2654700 Charge Of Magnesium Ion That Has 10 Electrons 93 rows this table shows the most common charges for atoms of the chemical elements. Learn how to name and write the charge of monatomic ions, such as mg 2+, from neutral atoms. This electric charge generated on the ion is. The usual charge of an element is common to its group. The charge on a magnesium ion with ten. Charge Of Magnesium Ion That Has 10 Electrons.