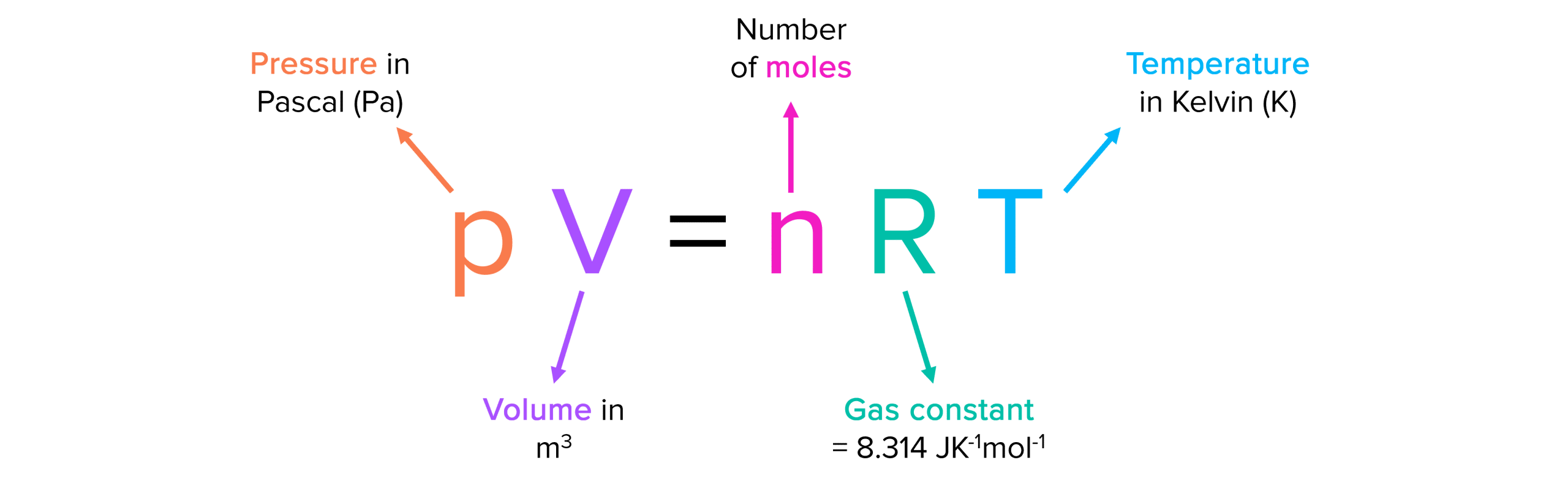What Is The Pressure Of The Gas . Gases exert pressure, which is force per unit area. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. Gases exert pressure, which is force per unit area. From this, you will learn how gases exert pressure on a. The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. This means that there are more air. Gas pressure is the force exerted by a gas on a specific area. A car tyre contains gas under pressure. The force which the substance exerts on another substance per unit area is known as pressure. The pressure of a gas may be expressed in the si unit of pascal or kilopascal, as well as in many.
from mmerevise.co.uk
From this, you will learn how gases exert pressure on a. Gases exert pressure, which is force per unit area. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. Gas pressure is the force exerted by a gas on a specific area. This means that there are more air. The pressure of a gas may be expressed in the si unit of pascal or kilopascal, as well as in many. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. The force which the substance exerts on another substance per unit area is known as pressure. Gases exert pressure, which is force per unit area.
The Ideal Gas Equation MME
What Is The Pressure Of The Gas From this, you will learn how gases exert pressure on a. A car tyre contains gas under pressure. From this, you will learn how gases exert pressure on a. The force which the substance exerts on another substance per unit area is known as pressure. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. Gases exert pressure, which is force per unit area. Gases exert pressure, which is force per unit area. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. This means that there are more air. The pressure of a gas may be expressed in the si unit of pascal or kilopascal, as well as in many. Gas pressure is the force exerted by a gas on a specific area.
From www.slideserve.com
PPT Gas Pressure PowerPoint Presentation, free download ID3551904 What Is The Pressure Of The Gas Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. The pressure of a gas may be expressed in the si unit of pascal or kilopascal, as well as in many. Gases exert pressure, which is force per unit. What Is The Pressure Of The Gas.
From flatworldknowledge.lardbucket.org
Relationships among Pressure, Temperature, Volume, and Amount What Is The Pressure Of The Gas The force which the substance exerts on another substance per unit area is known as pressure. Gases exert pressure, which is force per unit area. A car tyre contains gas under pressure. The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. Early scientists explored the relationships among the pressure of a gas (p). What Is The Pressure Of The Gas.
From philschatz.com
Gas Exchange · Anatomy and Physiology What Is The Pressure Of The Gas Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. Gases exert pressure,. What Is The Pressure Of The Gas.
From masterconceptsinchemistry.com
What’s the relationship between pressure and volume of gas? Core What Is The Pressure Of The Gas Gases exert pressure, which is force per unit area. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. This means that there are more air. The pressure of the gas equals the hydrostatic pressure due to a column. What Is The Pressure Of The Gas.
From www.youtube.com
Dalton's Law of Partial Pressure Problems, Mole Fraction, Chemistry Gas What Is The Pressure Of The Gas The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. The force which the substance exerts on another substance per unit area is known as pressure. Early scientists explored the relationships among the. What Is The Pressure Of The Gas.
From www.britannica.com
Pressure Definition, Measurement, & Types Britannica What Is The Pressure Of The Gas The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. This means that there are more air. Gas pressure is the force exerted by a gas on a. What Is The Pressure Of The Gas.
From courses.lumenlearning.com
Relating Pressure, Volume, Amount, and Temperature The Ideal Gas Law What Is The Pressure Of The Gas The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. This means that there are more air. Gas pressure is the force exerted by a gas on a specific area. From this, you will learn how gases exert pressure on a. A car tyre. What Is The Pressure Of The Gas.
From www.shalom-education.com
Volume and Pressure in Gases GCSE Physics Revision What Is The Pressure Of The Gas A car tyre contains gas under pressure. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. Gases exert pressure, which is force per unit area. From this, you will learn how gases exert pressure on a. This means that there are more air. The pressure of a gas. What Is The Pressure Of The Gas.
From www.youtube.com
Calculating Gas Pressure Pressure in the Gas Laws CLEAR & SIMPLE What Is The Pressure Of The Gas In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. From this, you will learn how gases exert pressure on a. Gases exert. What Is The Pressure Of The Gas.
From courses.lumenlearning.com
8.3 Gases and Pressure The Basics of General, Organic, and Biological What Is The Pressure Of The Gas This means that there are more air. The pressure of a gas may be expressed in the si unit of pascal or kilopascal, as well as in many. Gases exert pressure, which is force per unit area. Gases exert pressure, which is force per unit area. The force which the substance exerts on another substance per unit area is known. What Is The Pressure Of The Gas.
From driverlayer.com
what is the definition of atmospheric pressure DriverLayer Search Engine What Is The Pressure Of The Gas Gas pressure is the force exerted by a gas on a specific area. This means that there are more air. Gases exert pressure, which is force per unit area. The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. The pressure of a gas may be expressed in the si unit of pascal or. What Is The Pressure Of The Gas.
From chem.libretexts.org
6.2 The Simple Gas Laws Chemistry LibreTexts What Is The Pressure Of The Gas The force which the substance exerts on another substance per unit area is known as pressure. This means that there are more air. Gas pressure is the force exerted by a gas on a specific area. From this, you will learn how gases exert pressure on a. Gases exert pressure, which is force per unit area. The pressure of a. What Is The Pressure Of The Gas.
From www.slideserve.com
PPT Gas Partial Pressures in Atmospheric Air ( Total Pressure = 760 What Is The Pressure Of The Gas A car tyre contains gas under pressure. Gases exert pressure, which is force per unit area. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. This means that there are more air. From this, you will learn how. What Is The Pressure Of The Gas.
From mmerevise.co.uk
The Ideal Gas Equation MME What Is The Pressure Of The Gas The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. The pressure of a gas may be expressed in the si unit of. What Is The Pressure Of The Gas.
From www.slideserve.com
PPT Chemistry 14.1 PowerPoint Presentation, free download ID4874125 What Is The Pressure Of The Gas A car tyre contains gas under pressure. This means that there are more air. Gas pressure is the force exerted by a gas on a specific area. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. The pressure. What Is The Pressure Of The Gas.
From www.embibe.com
Sketch a graph to show the relationship between the pressure of a gas What Is The Pressure Of The Gas The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. From this, you will learn how gases exert pressure on a. The pressure of the gas equals the hydrostatic pressure due to a. What Is The Pressure Of The Gas.
From www.sciencephoto.com
Gay Lussac's pressuretemperature gas law, illustration Stock Image What Is The Pressure Of The Gas A car tyre contains gas under pressure. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. From this, you will learn how. What Is The Pressure Of The Gas.
From www.askiitians.com
Gas Laws And Properties Of Gases Study Material for IIT JEE askIITians What Is The Pressure Of The Gas The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. Gas pressure is the force exerted by a gas on a specific area. A car tyre contains gas. What Is The Pressure Of The Gas.
From www.youtube.com
Gas Pressure and Volume GCSE Physics Revision YouTube What Is The Pressure Of The Gas From this, you will learn how gases exert pressure on a. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. The pressure. What Is The Pressure Of The Gas.
From www.slideserve.com
PPT Dalton’s Law The total pressure of a mixture of gases equals the What Is The Pressure Of The Gas In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. This means that there are more air. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. From this, you will learn how. What Is The Pressure Of The Gas.
From www.youtube.com
Combined Gas Law Pressure, Volume and Temperature Straight Science What Is The Pressure Of The Gas The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. Gases exert pressure, which is force per unit area. The pressure of the gas equals the hydrostatic pressure due to a column of. What Is The Pressure Of The Gas.
From study.com
GayLussac's Law Gas Pressure and Temperature Relationship Video What Is The Pressure Of The Gas Gases exert pressure, which is force per unit area. From this, you will learn how gases exert pressure on a. Gas pressure is the force exerted by a gas on a specific area. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. In. What Is The Pressure Of The Gas.
From www.slideserve.com
PPT GAS EXCHANGE AND GAS TRANSFER PowerPoint Presentation, free What Is The Pressure Of The Gas The pressure of a gas may be expressed in the si unit of pascal or kilopascal, as well as in many. Gases exert pressure, which is force per unit area. This means that there are more air. The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. Gases exert pressure, which is force per. What Is The Pressure Of The Gas.
From www.slideserve.com
PPT 1. The volume of a gas at 99.0 kPa is 300.0 mL. If the pressure What Is The Pressure Of The Gas Gases exert pressure, which is force per unit area. Gases exert pressure, which is force per unit area. Gas pressure is the force exerted by a gas on a specific area. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant. What Is The Pressure Of The Gas.
From socratic.org
A sample of a gas has a volume of 2.0 liters at a pressure of 1.0 What Is The Pressure Of The Gas A car tyre contains gas under pressure. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure. What Is The Pressure Of The Gas.
From saylordotorg.github.io
Gases What Is The Pressure Of The Gas Gas pressure is the force exerted by a gas on a specific area. This means that there are more air. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. Gases exert pressure, which is force per unit area. From this, you will learn how gases exert pressure on. What Is The Pressure Of The Gas.
From www.visionlearning.com
Properties of Gases Chemistry Visionlearning What Is The Pressure Of The Gas This means that there are more air. Gases exert pressure, which is force per unit area. The force which the substance exerts on another substance per unit area is known as pressure. Gas pressure is the force exerted by a gas on a specific area. The pressure of the gas equals the hydrostatic pressure due to a column of mercury. What Is The Pressure Of The Gas.
From www.slideserve.com
PPT Gases PowerPoint Presentation, free download ID4144868 What Is The Pressure Of The Gas This means that there are more air. Gases exert pressure, which is force per unit area. The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. The pressure. What Is The Pressure Of The Gas.
From www.slideserve.com
PPT Chapter 5 Gases PowerPoint Presentation, free download ID6918783 What Is The Pressure Of The Gas Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm plus the pressure of the atmosphere at. The force which. What Is The Pressure Of The Gas.
From chem.libretexts.org
7.2 The Gas Laws Chemistry LibreTexts What Is The Pressure Of The Gas From this, you will learn how gases exert pressure on a. A car tyre contains gas under pressure. Gases exert pressure, which is force per unit area. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. The pressure. What Is The Pressure Of The Gas.
From drcalef.com
The Combined Gas Law Pressure, Volume, and Temperature What Is The Pressure Of The Gas The pressure of a gas may be expressed in the si unit of pascal or kilopascal,. The force which the substance exerts on another substance per unit area is known as pressure. Gases exert pressure, which is force per unit area. The pressure of the gas equals the hydrostatic pressure due to a column of mercury of height 13.7 cm. What Is The Pressure Of The Gas.
From owlcation.com
The Theories and Behavior of Gas Owlcation What Is The Pressure Of The Gas This means that there are more air. In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. Gas pressure is the force exerted by a gas on a specific area. The pressure of a gas may be expressed in the si unit of pascal or kilopascal, as well as. What Is The Pressure Of The Gas.
From www.expii.com
Combined Gas Law — Overview & Calculations Expii What Is The Pressure Of The Gas In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. From this, you will learn how gases exert pressure on a. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables. What Is The Pressure Of The Gas.
From www.youtube.com
11.8 The Ideal Gas Law Pressure, Volume, Temperature, & Moles YouTube What Is The Pressure Of The Gas Gases exert pressure, which is force per unit area. Early scientists explored the relationships among the pressure of a gas (p) and its temperature (t), volume (v), and amount (n) by holding two of the four variables constant (amount and. A car tyre contains gas under pressure. The pressure of the gas equals the hydrostatic pressure due to a column. What Is The Pressure Of The Gas.
From www.edplace.com
Understand Gas Pressure Worksheet EdPlace What Is The Pressure Of The Gas In this article, you will learn the definition of pressure, and how to apply this definition to a sample of gas. The pressure of a gas may be expressed in the si unit of pascal or kilopascal, as well as in many. A car tyre contains gas under pressure. The pressure of a gas may be expressed in the si. What Is The Pressure Of The Gas.