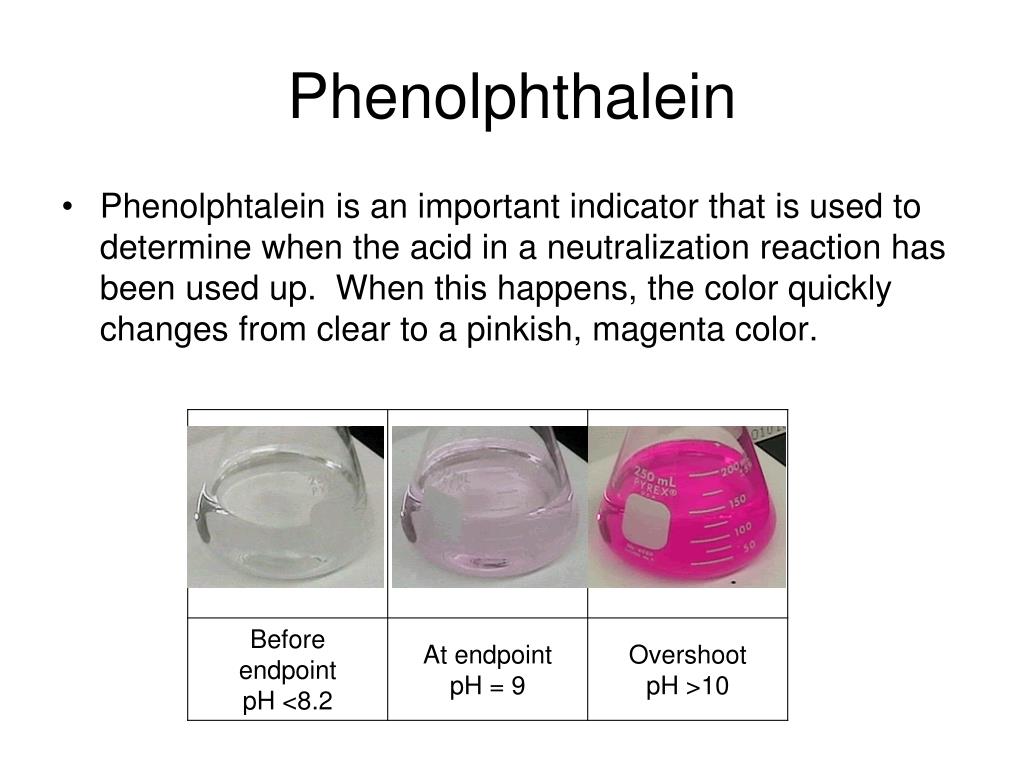
What Is Acid-Base Indicator Phenolphthalein . The weak acid form (hin) will have one. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. For this application, it turns colorless in acidic solutions and. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. In this case, the weak acid is colourless. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. Phenolphthalein is another commonly used indicator for titrations, and is another. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values.
from www.slideserve.com
For this application, it turns colorless in acidic solutions and. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. The weak acid form (hin) will have one. Phenolphthalein is another commonly used indicator for titrations, and is another. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. In this case, the weak acid is colourless.
PPT The pH scale PowerPoint Presentation, free download ID4827855
What Is Acid-Base Indicator Phenolphthalein Phenolphthalein is another commonly used indicator for titrations, and is another. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. The weak acid form (hin) will have one. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. In this case, the weak acid is colourless. Phenolphthalein is another commonly used indicator for titrations, and is another. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. For this application, it turns colorless in acidic solutions and.
From www.dreamstime.com
Phenolphthalein Indicator in Acidbase Titration Stock Vector What Is Acid-Base Indicator Phenolphthalein in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. an indicator is usually some. What Is Acid-Base Indicator Phenolphthalein.
From www.numerade.com
SOLVED Colours of phenolphthalein indicator Alkaline Endpoint True What Is Acid-Base Indicator Phenolphthalein in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. In this case, the weak acid is colourless. Phenolphthalein is another commonly used indicator for titrations, and. What Is Acid-Base Indicator Phenolphthalein.
From www.youtube.com
Acid Base Indicator Phenolphthalein explained with experiment What Is Acid-Base Indicator Phenolphthalein The weak acid form (hin) will have one. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. In this case, the weak acid is colourless. For this application, it turns colorless in acidic solutions and. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. an indicator. What Is Acid-Base Indicator Phenolphthalein.
From www.priyamstudycentre.com
Phenolphthalein Indicator, Solution, Uses What Is Acid-Base Indicator Phenolphthalein The weak acid form (hin) will have one. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. an indicator is usually some weak organic acid or base dye that changes colors at definite. What Is Acid-Base Indicator Phenolphthalein.
From www.alamy.com
Phenolphthalein is used as a single indicator in acidbase titrations What Is Acid-Base Indicator Phenolphthalein an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. For this application, it turns colorless in acidic solutions and. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. in acids, the phenolphthalein indicator remains colorless but starts to turn pink. What Is Acid-Base Indicator Phenolphthalein.
From www.vrogue.co
Acid Base Indicator Phenolphthalein Explained With Ex vrogue.co What Is Acid-Base Indicator Phenolphthalein Phenolphthalein is another commonly used indicator for titrations, and is another. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. In this case, the weak acid is colourless. The weak acid form (hin) will have one. . What Is Acid-Base Indicator Phenolphthalein.
From www.youtube.com
Using Phenolphthalein as an AcidBase Indicator / pH Indicator YouTube What Is Acid-Base Indicator Phenolphthalein phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. For this application, it turns colorless in acidic solutions and. an indicator is usually some weak organic acid or base dye that changes colors. What Is Acid-Base Indicator Phenolphthalein.
From labbyag.es
Phenolphthalein Color Chart Labb by AG What Is Acid-Base Indicator Phenolphthalein an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. In this case, the weak acid is colourless. Phenolphthalein is another commonly used indicator for titrations, and is another. The weak acid form (hin) will have one. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. . What Is Acid-Base Indicator Phenolphthalein.
From www.teachoo.com
Acid Base Indicators All types [List with Examples] Teachoo What Is Acid-Base Indicator Phenolphthalein phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. Phenolphthalein is another commonly used indicator for titrations, and is another. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at. What Is Acid-Base Indicator Phenolphthalein.
From www.slideserve.com
PPT Unit 6 Chpt 15 Acid/Base Equilibria PowerPoint Presentation What Is Acid-Base Indicator Phenolphthalein Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. In this case, the weak acid is colourless. For this application, it turns colorless in acidic solutions and. Phenolphthalein is another commonly used indicator for titrations, and is another.. What Is Acid-Base Indicator Phenolphthalein.
From www.youtube.com
Make phenolphthalein A Acid Base Indicator YouTube What Is Acid-Base Indicator Phenolphthalein phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. Phenolphthalein is another commonly used indicator for titrations, and is another. In this case, the weak acid is colourless. substances such as phenolphthalein,. What Is Acid-Base Indicator Phenolphthalein.
From www.vrogue.co
Acid Base Indicator Phenolphthalein Explained With Ex vrogue.co What Is Acid-Base Indicator Phenolphthalein substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. In this case, the weak acid is colourless. For this application, it turns colorless in acidic solutions and. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. in acids, the phenolphthalein indicator remains. What Is Acid-Base Indicator Phenolphthalein.
From www.slideserve.com
PPT The pH scale PowerPoint Presentation, free download ID4827855 What Is Acid-Base Indicator Phenolphthalein In this case, the weak acid is colourless. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. For this application, it turns colorless in acidic solutions and. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. The weak acid form (hin) will have. What Is Acid-Base Indicator Phenolphthalein.
From www.nagwa.com
Question Video Identifying the Color of a Solution Containing the Acid What Is Acid-Base Indicator Phenolphthalein For this application, it turns colorless in acidic solutions and. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely. What Is Acid-Base Indicator Phenolphthalein.
From www.alamy.com
Phenolphthalein in a labelled laboratory bottle. This colourless What Is Acid-Base Indicator Phenolphthalein Phenolphthalein is another commonly used indicator for titrations, and is another. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. For this application, it turns colorless in acidic solutions and. phenolphthalein,. What Is Acid-Base Indicator Phenolphthalein.
From www.alamy.com
Phenolphthalein acidbase indicator. Phenolphthalein indicator changes What Is Acid-Base Indicator Phenolphthalein Phenolphthalein is another commonly used indicator for titrations, and is another. In this case, the weak acid is colourless. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. Phenolphthalein is another commonly used. What Is Acid-Base Indicator Phenolphthalein.
From fphoto.photoshelter.com
science chemistry titration phenolphthalein Fundamental Photographs What Is Acid-Base Indicator Phenolphthalein an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong. What Is Acid-Base Indicator Phenolphthalein.
From www.teachoo.com
Acid Base Indicators All types [List with Examples] Teachoo What Is Acid-Base Indicator Phenolphthalein Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. For this application, it turns. What Is Acid-Base Indicator Phenolphthalein.
From brainly.in
what happens when we add acid and base on phenolphthalein?? Brainly.in What Is Acid-Base Indicator Phenolphthalein an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. Phenolphthalein is another commonly used indicator for titrations, and is another. For this application, it turns colorless. What Is Acid-Base Indicator Phenolphthalein.
From www.shutterstock.com
Acidbase Titration Setup Phenolphthalein Indicator Vector Stock Vector What Is Acid-Base Indicator Phenolphthalein The weak acid form (hin) will have one. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. In this case, the weak acid is colourless. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. in acids, the phenolphthalein indicator remains colorless but starts to turn. What Is Acid-Base Indicator Phenolphthalein.
From www.dreamstime.com
Phenolphthalein Indicator Molecule. Used in Acid Base Titrations and As What Is Acid-Base Indicator Phenolphthalein phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. The weak acid form (hin) will have one. In this case, the weak acid is colourless. Phenolphthalein is another commonly used indicator for titrations, and is another. an indicator is usually some weak organic acid or base dye that changes colors at. What Is Acid-Base Indicator Phenolphthalein.
From www.alamy.com
Phenolphthalein is used as a single indicator in acidbase titrations What Is Acid-Base Indicator Phenolphthalein substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. In this case, the weak acid is colourless. For this application, it turns colorless in acidic solutions and. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. The weak acid form (hin) will have. What Is Acid-Base Indicator Phenolphthalein.
From www.vrogue.co
Acid Base Titration Setup Phenolphthalein Indicator V vrogue.co What Is Acid-Base Indicator Phenolphthalein substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. The weak acid form (hin) will have one. For this application, it turns colorless in acidic solutions and. Phenolphthalein is another commonly used indicator for. What Is Acid-Base Indicator Phenolphthalein.
From www.vrogue.co
Why Is Phenolphthalein A Good Indicator For Acid Base vrogue.co What Is Acid-Base Indicator Phenolphthalein In this case, the weak acid is colourless. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. substances such as phenolphthalein, which can be used. What Is Acid-Base Indicator Phenolphthalein.
From courses.lumenlearning.com
AcidBase Indicators Introduction to Chemistry What Is Acid-Base Indicator Phenolphthalein Phenolphthalein is another commonly used indicator for titrations, and is another. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. The weak acid form (hin) will have one. For this application, it turns colorless in acidic solutions and. Phenolphthalein is another commonly used indicator for titrations, and is another weak. What Is Acid-Base Indicator Phenolphthalein.
From www.dreamstime.com
Phenolphthalein Indicator Molecule. Used in Acid Base Titrations and As What Is Acid-Base Indicator Phenolphthalein Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. In this case, the weak acid is colourless. Phenolphthalein is another commonly used indicator for titrations, and is another. in acids, the phenolphthalein indicator remains colorless but starts. What Is Acid-Base Indicator Phenolphthalein.
From smallbusinessron.web.fc2.com
what color does phenolphthalein turn in a base What Is Acid-Base Indicator Phenolphthalein substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. In this case, the weak acid is colourless. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. an indicator is usually some weak organic acid or base dye that changes colors at definite ph values. in. What Is Acid-Base Indicator Phenolphthalein.
From sciencenotes.org
Phenolphthalein Indicator What Is Acid-Base Indicator Phenolphthalein phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. In this case, the weak acid is colourless. Phenolphthalein is another commonly used indicator for titrations, and is another. substances such as phenolphthalein, which can be used to determine. What Is Acid-Base Indicator Phenolphthalein.
From www.sciencephoto.com
Phenolphthalein Indicator Stock Image C039/1218 Science Photo Library What Is Acid-Base Indicator Phenolphthalein phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. For this application, it turns colorless in acidic solutions and. In this case, the weak acid is colourless. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. an indicator is usually some weak. What Is Acid-Base Indicator Phenolphthalein.
From exouyldhc.blob.core.windows.net
Name Of Indicator Used In Acid Base Titration at Harold Petersen blog What Is Acid-Base Indicator Phenolphthalein Phenolphthalein is another commonly used indicator for titrations, and is another. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at a ph of 8.2, becoming bright purple in strong bases. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. In this case, the weak acid is colourless.. What Is Acid-Base Indicator Phenolphthalein.
From slideplayer.com
Indicators. ppt download What Is Acid-Base Indicator Phenolphthalein In this case, the weak acid is colourless. Phenolphthalein is another commonly used indicator for titrations, and is another. For this application, it turns colorless in acidic solutions and. The weak acid form (hin) will have one. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. in acids, the phenolphthalein indicator. What Is Acid-Base Indicator Phenolphthalein.
From www.sciencephoto.com
Phenolphthalein Indicator Stock Image C039/1215 Science Photo Library What Is Acid-Base Indicator Phenolphthalein The weak acid form (hin) will have one. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. Phenolphthalein is another commonly. What Is Acid-Base Indicator Phenolphthalein.
From anyboli.com
How does phenolphthalein indicator reacts with acidic and basic solution? What Is Acid-Base Indicator Phenolphthalein The weak acid form (hin) will have one. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. Phenolphthalein is another commonly used indicator for titrations, and is another. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. In this case, the weak acid is colourless. in. What Is Acid-Base Indicator Phenolphthalein.
From fphoto.photoshelter.com
science chemistry titration phenolphthalein Fundamental Photographs What Is Acid-Base Indicator Phenolphthalein For this application, it turns colorless in acidic solutions and. In this case, the weak acid is colourless. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. The weak acid form (hin) will have one. Phenolphthalein is another commonly used indicator for titrations, and is another weak acid. phenolphthalein, (c20h14o4),. What Is Acid-Base Indicator Phenolphthalein.
From www.britannica.com
Phenolphthalein pH indicator, acidbase titration, indicator dye What Is Acid-Base Indicator Phenolphthalein phenolphthalein, (c20h14o4), an organic compound of the phthalein family that is widely employed as an acid. substances such as phenolphthalein, which can be used to determine the ph of a solution, are called. Phenolphthalein is another commonly used indicator for titrations, and is another. in acids, the phenolphthalein indicator remains colorless but starts to turn pink at. What Is Acid-Base Indicator Phenolphthalein.