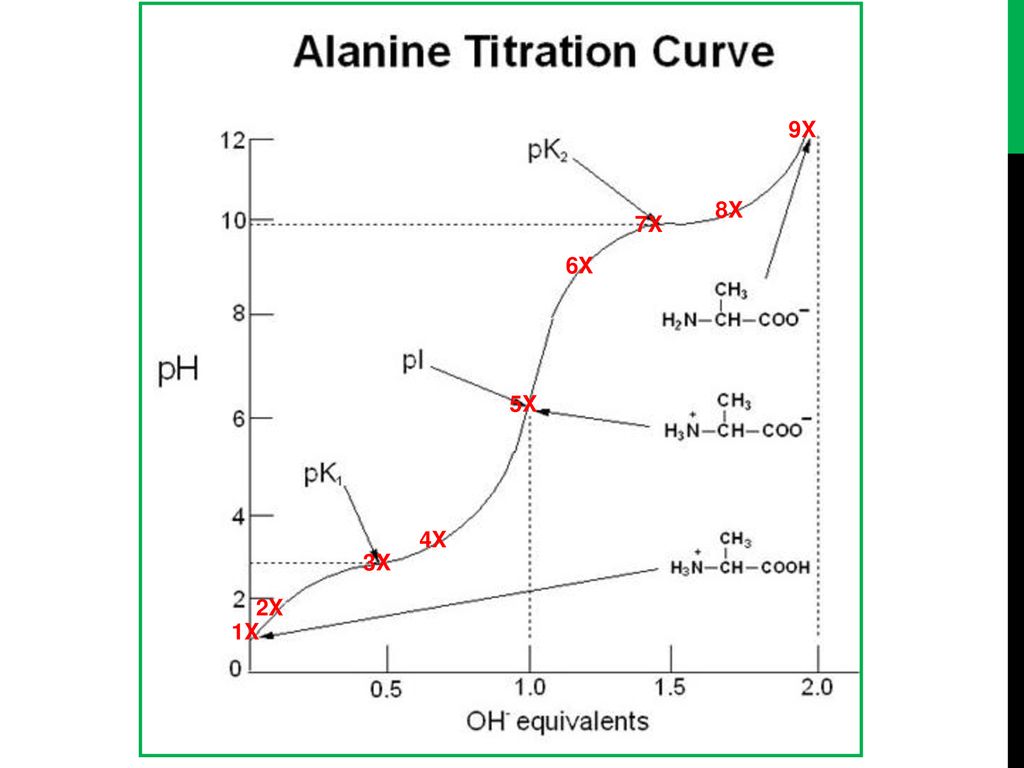
Titration Curve Of Amino Acid Glycine . But what happens when the amino acid contains an acidic side chain, such as in glutamic. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. The isoelectric points range from 5.5 to 6.2. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: Titration curve of amino acid (glycine) 1. Titration curves of amino acids are very useful for identification. As you can see in the example for glycine shown below, a simple amino acid has. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. These curves allow the prediction of protonation states, charges, and isoelectric points. Thus, you can use a titration. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. Each amino acid has a characteristic set of #pk# and #pi# values. Undergraduate biochemistry students should have great familiarity with titration curves.
from slideplayer.com
Obtaining pi through averaging the two pk a values of the amino acid is simple enough. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. Undergraduate biochemistry students should have great familiarity with titration curves. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. Each amino acid has a characteristic set of #pk# and #pi# values. But what happens when the amino acid contains an acidic side chain, such as in glutamic. Thus, you can use a titration. These curves allow the prediction of protonation states, charges, and isoelectric points. Titration curves of amino acids are very useful for identification. Titration curve of amino acid (glycine) 1.
Titration curve of amino acids ppt download
Titration Curve Of Amino Acid Glycine Thus, you can use a titration. Titration curves of amino acids are very useful for identification. Titration curve of amino acid (glycine) 1. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: Each amino acid has a characteristic set of #pk# and #pi# values. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. Thus, you can use a titration. But what happens when the amino acid contains an acidic side chain, such as in glutamic. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. These curves allow the prediction of protonation states, charges, and isoelectric points. The isoelectric points range from 5.5 to 6.2. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. As you can see in the example for glycine shown below, a simple amino acid has. Undergraduate biochemistry students should have great familiarity with titration curves.
From www3.nd.edu
Amino Acid Titration Curves Titration Curve Of Amino Acid Glycine But what happens when the amino acid contains an acidic side chain, such as in glutamic. Each amino acid has a characteristic set of #pk# and #pi# values. Undergraduate biochemistry students should have great familiarity with titration curves. The isoelectric points range from 5.5 to 6.2. Thus, you can use a titration. Obtaining pi through averaging the two pk a. Titration Curve Of Amino Acid Glycine.
From www.numerade.com
SOLVED 10. The diagram to the right shows an OH titration curve with Titration Curve Of Amino Acid Glycine These curves allow the prediction of protonation states, charges, and isoelectric points. Titration curve of amino acid (glycine) 1. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. As you can see in the example for glycine shown below, a simple amino acid has. Titration curves of amino acids are very useful for identification. At a very. Titration Curve Of Amino Acid Glycine.
From www.slideserve.com
PPT Amino Acids PowerPoint Presentation, free download ID9502455 Titration Curve Of Amino Acid Glycine Each amino acid has a characteristic set of #pk# and #pi# values. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. Titration curves of amino acids are very useful for identification. Thus, you can use a titration. The isoelectric points range from 5.5 to 6.2. Titration curve of amino acid (glycine) 1. At. Titration Curve Of Amino Acid Glycine.
From www.researchgate.net
1 pI of Glycine with Titration curve Download Scientific Diagram Titration Curve Of Amino Acid Glycine Undergraduate biochemistry students should have great familiarity with titration curves. Each amino acid has a characteristic set of #pk# and #pi# values. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. Titration curve of amino acid (glycine) 1. These curves allow the prediction of protonation states, charges,. Titration Curve Of Amino Acid Glycine.
From learnbin.net
Physiochemical Properties Of Amino Acids Learnbin Titration Curve Of Amino Acid Glycine Each amino acid has a characteristic set of #pk# and #pi# values. But what happens when the amino acid contains an acidic side chain, such as in glutamic. Titration curve of amino acid (glycine) 1. Titration curves of amino acids are very useful for identification. Thus, you can use a titration. Undergraduate biochemistry students should have great familiarity with titration. Titration Curve Of Amino Acid Glycine.
From www.youtube.com
Titration Curve for glycine in a easiest way YouTube Titration Curve Of Amino Acid Glycine Undergraduate biochemistry students should have great familiarity with titration curves. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. As you can see in the example for glycine shown below, a simple amino acid has. The isoelectric points range from 5.5 to 6.2. Obtaining pi through averaging. Titration Curve Of Amino Acid Glycine.
From cwsimons.com
How to Draw Titration Curves of Amino Acids Food Science Toolbox Titration Curve Of Amino Acid Glycine Thus, you can use a titration. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. Titration curves of amino acids are very useful for identification. Titration curve of amino acid (glycine) 1. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. But what happens. Titration Curve Of Amino Acid Glycine.
From www.scribd.com
Titration Curves of Amino Acids For This Experiment We Have Used Amino Titration Curve Of Amino Acid Glycine Obtaining pi through averaging the two pk a values of the amino acid is simple enough. Thus, you can use a titration. Undergraduate biochemistry students should have great familiarity with titration curves. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: Titration curve of amino acid (glycine) 1. Each amino acid has a. Titration Curve Of Amino Acid Glycine.
From slidetodoc.com
Lab 8 Titration Curve Potentiometic Titration of amino Titration Curve Of Amino Acid Glycine Titration curve of amino acid (glycine) 1. Each amino acid has a characteristic set of #pk# and #pi# values. These curves allow the prediction of protonation states, charges, and isoelectric points. As you can see in the example for glycine shown below, a simple amino acid has. Undergraduate biochemistry students should have great familiarity with titration curves. Titration curves of. Titration Curve Of Amino Acid Glycine.
From www.youtube.com
Titration curve of Glycine YouTube Titration Curve Of Amino Acid Glycine The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. Undergraduate biochemistry students should have great familiarity with titration curves. But what happens when the amino acid contains an acidic side chain, such as in glutamic. As you can see in the example for glycine shown below, a. Titration Curve Of Amino Acid Glycine.
From www.aqion.de
Zwitterions and Amino Acids Titration Curve Of Amino Acid Glycine For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. Titration curves of amino acids are very useful for identification. These curves allow the prediction of protonation states, charges, and isoelectric points. Thus, you can use a titration. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. As you can see. Titration Curve Of Amino Acid Glycine.
From www.youtube.com
Zwitterion/Titration of amino acids/Titration curve of Glycine amino Titration Curve Of Amino Acid Glycine These curves allow the prediction of protonation states, charges, and isoelectric points. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. Undergraduate biochemistry students should have great familiarity with titration curves. Obtaining pi through averaging. Titration Curve Of Amino Acid Glycine.
From slideplayer.com
Read the following slides by “Day 7” ppt download Titration Curve Of Amino Acid Glycine But what happens when the amino acid contains an acidic side chain, such as in glutamic. Titration curves of amino acids are very useful for identification. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. Thus, you can use a titration. The isoelectric points range from 5.5 to 6.2. The titration curve of. Titration Curve Of Amino Acid Glycine.
From www.studypool.com
SOLUTION Titration curve of amino acids Studypool Titration Curve Of Amino Acid Glycine At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: Titration curve of amino acid (glycine) 1. Thus, you can use a titration. These curves allow the prediction of protonation states, charges, and isoelectric points. Each amino acid has a characteristic set of #pk# and #pi# values. Titration curves of amino acids are very. Titration Curve Of Amino Acid Glycine.
From www3.nd.edu
Titration curve of glycine Titration Curve Of Amino Acid Glycine Titration curve of amino acid (glycine) 1. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. Titration curves of amino acids are very useful for identification. The isoelectric points range from 5.5 to 6.2. Obtaining. Titration Curve Of Amino Acid Glycine.
From ar.inspiredpencil.com
Glycine Titration Curve Titration Curve Of Amino Acid Glycine Each amino acid has a characteristic set of #pk# and #pi# values. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. Titration curve of amino acid (glycine) 1. The isoelectric points range from 5.5 to 6.2. But what happens when the amino acid contains an acidic side chain, such as in glutamic. These. Titration Curve Of Amino Acid Glycine.
From ar.inspiredpencil.com
Glycine Titration Curve Titration Curve Of Amino Acid Glycine These curves allow the prediction of protonation states, charges, and isoelectric points. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. As you can see in the example for glycine shown below, a simple amino acid has. Titration curves of amino acids are very useful for identification. For glycine, #pk_a1 = 2.34#, #pk_a2. Titration Curve Of Amino Acid Glycine.
From www.slideserve.com
PPT Chapter 3 PowerPoint Presentation, free download ID1205383 Titration Curve Of Amino Acid Glycine The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. As you can see in the example for glycine shown below, a simple amino acid has. Undergraduate biochemistry students should have great familiarity with titration curves. But what happens when the amino acid contains an acidic side chain,. Titration Curve Of Amino Acid Glycine.
From www.youtube.com
Titration curve of glycine Amino acids as buffers Glycine titration Titration Curve Of Amino Acid Glycine Thus, you can use a titration. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: Titration curves of amino acids are very useful for identification. The isoelectric points range from 5.5 to 6.2. Undergraduate biochemistry students should have great familiarity with. Titration Curve Of Amino Acid Glycine.
From www.numerade.com
SOLVED The diagram below represents a titration curve of glycine Titration Curve Of Amino Acid Glycine The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. Titration curve of amino acid (glycine) 1. Each amino acid has a characteristic set of #pk# and #pi# values. But what happens when the amino acid contains an acidic side chain, such as in glutamic. Titration curves of. Titration Curve Of Amino Acid Glycine.
From www.studypool.com
SOLUTION Amino acid titration curve Studypool Titration Curve Of Amino Acid Glycine Each amino acid has a characteristic set of #pk# and #pi# values. Undergraduate biochemistry students should have great familiarity with titration curves. Titration curve of amino acid (glycine) 1. The isoelectric points range from 5.5 to 6.2. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: Thus, you can use a titration. These. Titration Curve Of Amino Acid Glycine.
From www.animalia-life.club
Titration Curve Amino Acid Titration Curve Of Amino Acid Glycine The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. But what happens when the amino acid contains an acidic side chain, such as in glutamic. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: Undergraduate biochemistry students should have great. Titration Curve Of Amino Acid Glycine.
From www.chegg.com
Solved The following is a titration curve for which amino Titration Curve Of Amino Acid Glycine Titration curves of amino acids are very useful for identification. Thus, you can use a titration. As you can see in the example for glycine shown below, a simple amino acid has. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: Undergraduate biochemistry students should have great familiarity with titration curves. But what. Titration Curve Of Amino Acid Glycine.
From www.slideserve.com
PPT Lecture 3 Amino Acids PowerPoint Presentation, free download Titration Curve Of Amino Acid Glycine The isoelectric points range from 5.5 to 6.2. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. But what happens when the amino acid contains an acidic side chain, such as in glutamic. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. The titration curve of glycine tells the tale. Titration Curve Of Amino Acid Glycine.
From www3.nd.edu
Amino Acid Titration Curves Titration Curve Of Amino Acid Glycine Thus, you can use a titration. Titration curves of amino acids are very useful for identification. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: Undergraduate biochemistry students should have great. Titration Curve Of Amino Acid Glycine.
From mavink.com
Titration Curve Of Amino Acids Titration Curve Of Amino Acid Glycine Thus, you can use a titration. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. Undergraduate biochemistry students should have great familiarity with titration curves. Each amino acid has a characteristic set of #pk# and #pi# values. Titration curves of amino acids are very useful for identification.. Titration Curve Of Amino Acid Glycine.
From www.slideserve.com
PPT Amino acids PowerPoint Presentation, free download ID543480 Titration Curve Of Amino Acid Glycine As you can see in the example for glycine shown below, a simple amino acid has. Undergraduate biochemistry students should have great familiarity with titration curves. The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. These curves allow the prediction of protonation states, charges, and isoelectric points.. Titration Curve Of Amino Acid Glycine.
From slideplayer.com
Titration curve of amino acids ppt download Titration Curve Of Amino Acid Glycine The isoelectric points range from 5.5 to 6.2. But what happens when the amino acid contains an acidic side chain, such as in glutamic. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. Each amino acid has a characteristic set of #pk# and #pi# values. These curves allow the prediction of protonation states,. Titration Curve Of Amino Acid Glycine.
From chemistryguru.com.sg
Titration Curve of Amino Acid Titration Curve Of Amino Acid Glycine The titration curve of glycine tells the tale of how this amino acid responds and transforms when exposed to varying ph levels. Obtaining pi through averaging the two pk a values of the amino acid is simple enough. Undergraduate biochemistry students should have great familiarity with titration curves. Titration curve of amino acid (glycine) 1. These curves allow the prediction. Titration Curve Of Amino Acid Glycine.
From www.vedantu.com
What is the titration curve of glycine? Titration Curve Of Amino Acid Glycine Titration curves of amino acids are very useful for identification. But what happens when the amino acid contains an acidic side chain, such as in glutamic. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: As you can see in the example for glycine shown below, a simple amino acid has. Obtaining pi. Titration Curve Of Amino Acid Glycine.
From www.slideserve.com
PPT Chapter 3 AMINO ACIDS, PEPTIDES, AND PROTEINS PowerPoint Titration Curve Of Amino Acid Glycine For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. Thus, you can use a titration. As you can see in the example for glycine shown below, a simple amino acid has. Each amino acid has a characteristic set of #pk# and #pi# values. At a very low ph (acidic) both groups are fully protonated where the solution. Titration Curve Of Amino Acid Glycine.
From www.numerade.com
SOLVED 47. point) The graph below represents the titration curve for Titration Curve Of Amino Acid Glycine Titration curve of amino acid (glycine) 1. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. Thus, you can use a titration. The isoelectric points range from 5.5 to 6.2. As you can see in the example for glycine shown below, a simple amino acid has. Titration curves of amino acids are very useful for identification. The. Titration Curve Of Amino Acid Glycine.
From www.chegg.com
1)According to the titration curve of glycine shown Titration Curve Of Amino Acid Glycine Thus, you can use a titration. The isoelectric points range from 5.5 to 6.2. Titration curve of amino acid (glycine) 1. Undergraduate biochemistry students should have great familiarity with titration curves. Titration curves of amino acids are very useful for identification. But what happens when the amino acid contains an acidic side chain, such as in glutamic. At a very. Titration Curve Of Amino Acid Glycine.
From www.transtutors.com
(Solved) Shown below is the titration curve of glycine (Gly), using Titration Curve Of Amino Acid Glycine For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: But what happens when the amino acid contains an acidic side chain, such as in glutamic. As you can see in the example for glycine shown below, a simple amino acid has.. Titration Curve Of Amino Acid Glycine.
From biochemden.com
Titration Curve of Glycine The zwitter ionic changes Titration Curve Of Amino Acid Glycine These curves allow the prediction of protonation states, charges, and isoelectric points. For glycine, #pk_a1 = 2.34#, #pk_a2 = 9.60#, and #pi = 5.97#. As you can see in the example for glycine shown below, a simple amino acid has. At a very low ph (acidic) both groups are fully protonated where the solution predominantly contains: The titration curve of. Titration Curve Of Amino Acid Glycine.