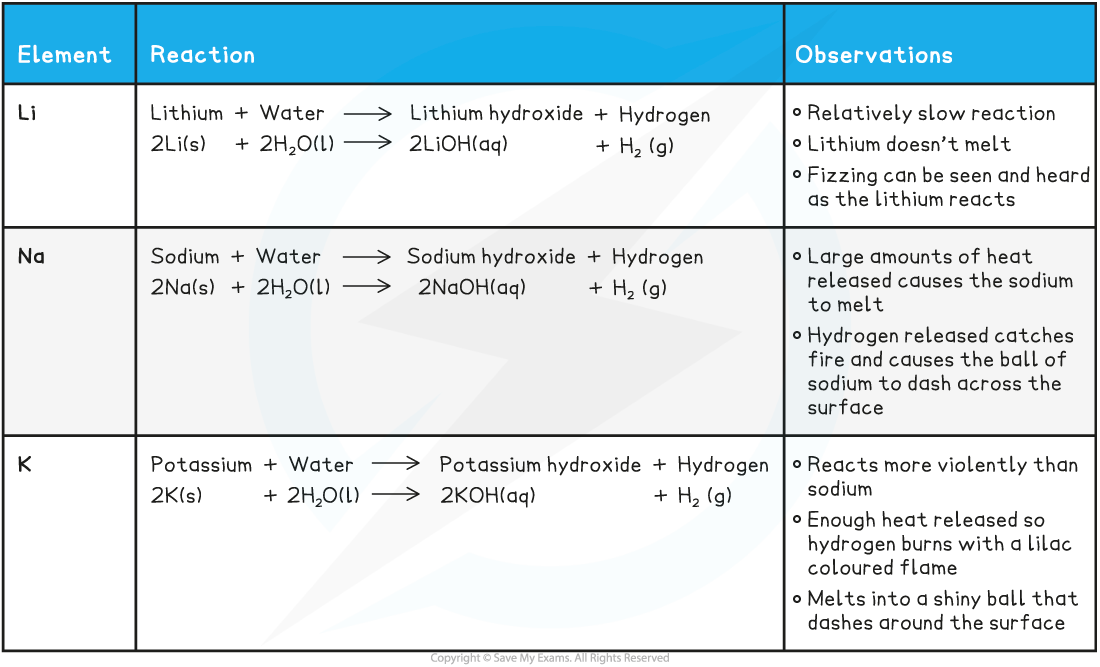
One Chemical Property Of Alkali Metals . Atoms of alkali metals have the largest atomic radii of elements in their periods. The chemistry of the alkali metals reflects their tendency to form +1 cations. Chemical properties of the alkali metals. Alkali metals are typically less dense than other metals. General physical properties of group 1 elements: The group 1 elements are all soft, reactive. Cation formation is favored by the relatively low. Alkali metals are grey solids with shiny silvery surfaces when. The lone outer shell electrons leads the alkali metal elements to share several common properties: The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). The alkali metals tend to form +1 cations. Table shows some properties of group 1 elements.
from www.linstitute.net
Cation formation is favored by the relatively low. Alkali metals are typically less dense than other metals. The lone outer shell electrons leads the alkali metal elements to share several common properties: The chemistry of the alkali metals reflects their tendency to form +1 cations. Atoms of alkali metals have the largest atomic radii of elements in their periods. General physical properties of group 1 elements: The group 1 elements are all soft, reactive. Table shows some properties of group 1 elements. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Chemical properties of the alkali metals.
Edexcel IGCSE Chemistry 复习笔记 2.1.1 Group 1 (Alkali Metals)翰林国际教育
One Chemical Property Of Alkali Metals Alkali metals are grey solids with shiny silvery surfaces when. The group 1 elements are all soft, reactive. Atoms of alkali metals have the largest atomic radii of elements in their periods. The chemistry of the alkali metals reflects their tendency to form +1 cations. Table shows some properties of group 1 elements. Cation formation is favored by the relatively low. General physical properties of group 1 elements: Alkali metals are typically less dense than other metals. The alkali metals tend to form +1 cations. Alkali metals are grey solids with shiny silvery surfaces when. The lone outer shell electrons leads the alkali metal elements to share several common properties: Chemical properties of the alkali metals. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +).
From www.vedantu.com
Alkali Metals Chemical Elements, Properties Alkali Metals Periodic One Chemical Property Of Alkali Metals The alkali metals tend to form +1 cations. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Table shows some properties of group 1 elements. The chemistry of the alkali metals reflects their tendency to form +1 cations. The lone outer shell electrons leads the alkali metal elements to share several common properties: Atoms. One Chemical Property Of Alkali Metals.
From www.tes.com
Lesson Alkali Metals GCSE Edexcel 91 Teaching Resources One Chemical Property Of Alkali Metals The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). General physical properties of group 1 elements: The alkali metals tend to form +1 cations. Alkali metals are typically less dense than other metals. Table shows some properties of group 1 elements. The chemistry of the alkali metals reflects their tendency to form +1 cations.. One Chemical Property Of Alkali Metals.
From cabinet.matttroy.net
Alkali Metals Periodic Table Facts Matttroy One Chemical Property Of Alkali Metals Table shows some properties of group 1 elements. Cation formation is favored by the relatively low. Alkali metals are grey solids with shiny silvery surfaces when. Alkali metals are typically less dense than other metals. Chemical properties of the alkali metals. The alkali metals tend to form +1 cations. The chemistry of the alkali metals reflects their tendency to form. One Chemical Property Of Alkali Metals.
From www.slideserve.com
PPT Physical Properties of Metals PowerPoint Presentation, free One Chemical Property Of Alkali Metals The lone outer shell electrons leads the alkali metal elements to share several common properties: Atoms of alkali metals have the largest atomic radii of elements in their periods. The group 1 elements are all soft, reactive. General physical properties of group 1 elements: Table shows some properties of group 1 elements. The alkali metals tend to form +1 cations.. One Chemical Property Of Alkali Metals.
From www.pinterest.com
Image result for alkali metals One Chemical Property Of Alkali Metals The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Chemical properties of the alkali metals. Atoms of alkali metals have the largest atomic radii of elements in their periods. The alkali metals tend to form +1 cations. Alkali metals are typically less dense than other metals. The chemistry of the alkali metals reflects their. One Chemical Property Of Alkali Metals.
From www.slideserve.com
PPT Introduction to the Periodic Table of Elements PowerPoint One Chemical Property Of Alkali Metals Chemical properties of the alkali metals. Cation formation is favored by the relatively low. Alkali metals are grey solids with shiny silvery surfaces when. The group 1 elements are all soft, reactive. Alkali metals are typically less dense than other metals. General physical properties of group 1 elements: The lone outer shell electrons leads the alkali metal elements to share. One Chemical Property Of Alkali Metals.
From www.slideshare.net
Physical and chemical properties of alkali metals One Chemical Property Of Alkali Metals The chemistry of the alkali metals reflects their tendency to form +1 cations. General physical properties of group 1 elements: The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Table shows some properties of group 1 elements. The lone outer shell electrons leads the alkali metal elements to share several common properties: Atoms of. One Chemical Property Of Alkali Metals.
From www.slideserve.com
PPT ELEMENT CLASSES PowerPoint Presentation ID149914 One Chemical Property Of Alkali Metals The chemistry of the alkali metals reflects their tendency to form +1 cations. Alkali metals are grey solids with shiny silvery surfaces when. Cation formation is favored by the relatively low. General physical properties of group 1 elements: Table shows some properties of group 1 elements. The lone outer shell electrons leads the alkali metal elements to share several common. One Chemical Property Of Alkali Metals.
From in.pinterest.com
What Are Alkali Metals On The Periodic Table? Alkali metal, Neon atom One Chemical Property Of Alkali Metals The chemistry of the alkali metals reflects their tendency to form +1 cations. The lone outer shell electrons leads the alkali metal elements to share several common properties: The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Chemical properties of the alkali metals. General physical properties of group 1 elements: Alkali metals are typically. One Chemical Property Of Alkali Metals.
From byjus.com
Alkali Metals Properties, Electronic Configuration, Periodic Trends One Chemical Property Of Alkali Metals Table shows some properties of group 1 elements. The alkali metals tend to form +1 cations. The lone outer shell electrons leads the alkali metal elements to share several common properties: General physical properties of group 1 elements: The chemistry of the alkali metals reflects their tendency to form +1 cations. The one outer electron is easily lost, forming the. One Chemical Property Of Alkali Metals.
From elchoroukhost.net
Properties Of Alkali Metals On The Periodic Table Elcho Table One Chemical Property Of Alkali Metals The lone outer shell electrons leads the alkali metal elements to share several common properties: Table shows some properties of group 1 elements. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). The chemistry of the alkali metals reflects their tendency to form +1 cations. The alkali metals tend to form +1 cations. Chemical. One Chemical Property Of Alkali Metals.
From www.slideserve.com
PPT Look at the following patterns. What are the patterns based on One Chemical Property Of Alkali Metals Chemical properties of the alkali metals. The group 1 elements are all soft, reactive. Atoms of alkali metals have the largest atomic radii of elements in their periods. Table shows some properties of group 1 elements. The alkali metals tend to form +1 cations. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Alkali. One Chemical Property Of Alkali Metals.
From www.chemistrysources.com
alkali metals (group 1 elements) مصادر الكيمياء One Chemical Property Of Alkali Metals The chemistry of the alkali metals reflects their tendency to form +1 cations. General physical properties of group 1 elements: Table shows some properties of group 1 elements. The alkali metals tend to form +1 cations. Alkali metals are typically less dense than other metals. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +).. One Chemical Property Of Alkali Metals.
From selfstudypoint.in
Group 1 Elements Alkali Metals One Chemical Property Of Alkali Metals The group 1 elements are all soft, reactive. Alkali metals are typically less dense than other metals. Atoms of alkali metals have the largest atomic radii of elements in their periods. Chemical properties of the alkali metals. The lone outer shell electrons leads the alkali metal elements to share several common properties: General physical properties of group 1 elements: Cation. One Chemical Property Of Alkali Metals.
From www.slideserve.com
PPT Atomic Theory PowerPoint Presentation, free download ID5435746 One Chemical Property Of Alkali Metals Atoms of alkali metals have the largest atomic radii of elements in their periods. The lone outer shell electrons leads the alkali metal elements to share several common properties: The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Alkali metals are typically less dense than other metals. The chemistry of the alkali metals reflects. One Chemical Property Of Alkali Metals.
From www.nagwa.com
Question Video Identifying the Property of Alkali Metals From a List One Chemical Property Of Alkali Metals The group 1 elements are all soft, reactive. Atoms of alkali metals have the largest atomic radii of elements in their periods. General physical properties of group 1 elements: The lone outer shell electrons leads the alkali metal elements to share several common properties: The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Table. One Chemical Property Of Alkali Metals.
From www.learnatnoon.com
What are the chemical properties of metals and nonmetals? Noon One Chemical Property Of Alkali Metals The alkali metals tend to form +1 cations. Atoms of alkali metals have the largest atomic radii of elements in their periods. Alkali metals are grey solids with shiny silvery surfaces when. The group 1 elements are all soft, reactive. The lone outer shell electrons leads the alkali metal elements to share several common properties: Cation formation is favored by. One Chemical Property Of Alkali Metals.
From elchoroukhost.net
Alkali Metals Periodic Table Properties Elcho Table One Chemical Property Of Alkali Metals The alkali metals tend to form +1 cations. General physical properties of group 1 elements: Cation formation is favored by the relatively low. Alkali metals are typically less dense than other metals. Alkali metals are grey solids with shiny silvery surfaces when. The chemistry of the alkali metals reflects their tendency to form +1 cations. The lone outer shell electrons. One Chemical Property Of Alkali Metals.
From quizizz.com
Alkali metals and Halogens Chemistry Quizizz One Chemical Property Of Alkali Metals Chemical properties of the alkali metals. Cation formation is favored by the relatively low. Table shows some properties of group 1 elements. The group 1 elements are all soft, reactive. Alkali metals are grey solids with shiny silvery surfaces when. The alkali metals tend to form +1 cations. The one outer electron is easily lost, forming the univalent (1+) cation. One Chemical Property Of Alkali Metals.
From ppt-online.org
Alkali metals презентация онлайн One Chemical Property Of Alkali Metals Cation formation is favored by the relatively low. Alkali metals are grey solids with shiny silvery surfaces when. Table shows some properties of group 1 elements. The group 1 elements are all soft, reactive. Chemical properties of the alkali metals. The chemistry of the alkali metals reflects their tendency to form +1 cations. The alkali metals tend to form +1. One Chemical Property Of Alkali Metals.
From studymind.co.uk
The Transition Metals (GCSE Chemistry) Study Mind One Chemical Property Of Alkali Metals The chemistry of the alkali metals reflects their tendency to form +1 cations. Table shows some properties of group 1 elements. Cation formation is favored by the relatively low. General physical properties of group 1 elements: The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Alkali metals are grey solids with shiny silvery surfaces. One Chemical Property Of Alkali Metals.
From www.youtube.com
Alkali Metals YouTube One Chemical Property Of Alkali Metals Chemical properties of the alkali metals. Atoms of alkali metals have the largest atomic radii of elements in their periods. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Alkali metals are typically less dense than other metals. The group 1 elements are all soft, reactive. Cation formation is favored by the relatively low.. One Chemical Property Of Alkali Metals.
From issuu.com
Chemistry Alkali Metals by AshfordSchool Issuu One Chemical Property Of Alkali Metals Table shows some properties of group 1 elements. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). General physical properties of group 1 elements: Alkali metals are grey solids with shiny silvery surfaces when. Cation formation is favored by the relatively low. The chemistry of the alkali metals reflects their tendency to form +1. One Chemical Property Of Alkali Metals.
From xlskoor.blogspot.com
Alkali Metals Chemistry One Chemical Property Of Alkali Metals The alkali metals tend to form +1 cations. Chemical properties of the alkali metals. General physical properties of group 1 elements: The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Table shows some properties of group 1 elements. Alkali metals are grey solids with shiny silvery surfaces when. The chemistry of the alkali metals. One Chemical Property Of Alkali Metals.
From awesomehome.co
Alkali Metals Periodic Table Awesome Home One Chemical Property Of Alkali Metals The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Alkali metals are grey solids with shiny silvery surfaces when. The alkali metals tend to form +1 cations. The chemistry of the alkali metals reflects their tendency to form +1 cations. Alkali metals are typically less dense than other metals. Cation formation is favored by. One Chemical Property Of Alkali Metals.
From slideplayer.com
Alkali Metals Electrostructure and reactivity Physical properties ppt One Chemical Property Of Alkali Metals Table shows some properties of group 1 elements. Alkali metals are typically less dense than other metals. Cation formation is favored by the relatively low. Chemical properties of the alkali metals. General physical properties of group 1 elements: The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Alkali metals are grey solids with shiny. One Chemical Property Of Alkali Metals.
From www.slideserve.com
PPT ELEMENTS CHEMICAL & PHYSICAL PROPERTIES PowerPoint Presentation One Chemical Property Of Alkali Metals Table shows some properties of group 1 elements. Cation formation is favored by the relatively low. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). The lone outer shell electrons leads the alkali metal elements to share several common properties: Alkali metals are grey solids with shiny silvery surfaces when. Chemical properties of the. One Chemical Property Of Alkali Metals.
From www.britannica.com
alkali metal Definition, Properties, & Facts Britannica One Chemical Property Of Alkali Metals Table shows some properties of group 1 elements. Atoms of alkali metals have the largest atomic radii of elements in their periods. Chemical properties of the alkali metals. The alkali metals tend to form +1 cations. Cation formation is favored by the relatively low. The chemistry of the alkali metals reflects their tendency to form +1 cations. General physical properties. One Chemical Property Of Alkali Metals.
From fyonvadia.blob.core.windows.net
Alkali Metals Chemical Property at Rolando Harry blog One Chemical Property Of Alkali Metals General physical properties of group 1 elements: The alkali metals tend to form +1 cations. Table shows some properties of group 1 elements. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Cation formation is favored by the relatively low. Chemical properties of the alkali metals. The lone outer shell electrons leads the alkali. One Chemical Property Of Alkali Metals.
From elchoroukhost.net
Alkali Metals Periodic Table Location Elcho Table One Chemical Property Of Alkali Metals Alkali metals are grey solids with shiny silvery surfaces when. Table shows some properties of group 1 elements. General physical properties of group 1 elements: Alkali metals are typically less dense than other metals. The lone outer shell electrons leads the alkali metal elements to share several common properties: The group 1 elements are all soft, reactive. The one outer. One Chemical Property Of Alkali Metals.
From www.differencebetween.com
Difference Between Group 1 Metals and Transition Metals Compare the One Chemical Property Of Alkali Metals The group 1 elements are all soft, reactive. The chemistry of the alkali metals reflects their tendency to form +1 cations. Alkali metals are grey solids with shiny silvery surfaces when. Chemical properties of the alkali metals. General physical properties of group 1 elements: Atoms of alkali metals have the largest atomic radii of elements in their periods. The one. One Chemical Property Of Alkali Metals.
From cabinet.matttroy.net
Alkali Metals Periodic Table Matttroy One Chemical Property Of Alkali Metals Alkali metals are grey solids with shiny silvery surfaces when. Cation formation is favored by the relatively low. Atoms of alkali metals have the largest atomic radii of elements in their periods. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). General physical properties of group 1 elements: The alkali metals tend to form. One Chemical Property Of Alkali Metals.
From spmchemistry.blog.onlinetuition.com.my
Physical Properties of Alkali Metals SPM Chemistry One Chemical Property Of Alkali Metals General physical properties of group 1 elements: The lone outer shell electrons leads the alkali metal elements to share several common properties: Table shows some properties of group 1 elements. Alkali metals are grey solids with shiny silvery surfaces when. The alkali metals tend to form +1 cations. Chemical properties of the alkali metals. Alkali metals are typically less dense. One Chemical Property Of Alkali Metals.
From www.slideserve.com
PPT Alkali Metals PowerPoint Presentation, free download ID2053861 One Chemical Property Of Alkali Metals Alkali metals are typically less dense than other metals. General physical properties of group 1 elements: The lone outer shell electrons leads the alkali metal elements to share several common properties: Table shows some properties of group 1 elements. Cation formation is favored by the relatively low. The alkali metals tend to form +1 cations. The group 1 elements are. One Chemical Property Of Alkali Metals.
From www.linstitute.net
Edexcel IGCSE Chemistry 复习笔记 2.1.1 Group 1 (Alkali Metals)翰林国际教育 One Chemical Property Of Alkali Metals The lone outer shell electrons leads the alkali metal elements to share several common properties: Cation formation is favored by the relatively low. The alkali metals tend to form +1 cations. The one outer electron is easily lost, forming the univalent (1+) cation (e.g., na +). Alkali metals are typically less dense than other metals. The chemistry of the alkali. One Chemical Property Of Alkali Metals.