What Is An Assay In Clinical Trials . once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. performance standards reporting requirements for essential assays in clinical trials.
from www.activemotif.com.cn
this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. performance standards reporting requirements for essential assays in clinical trials. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic.
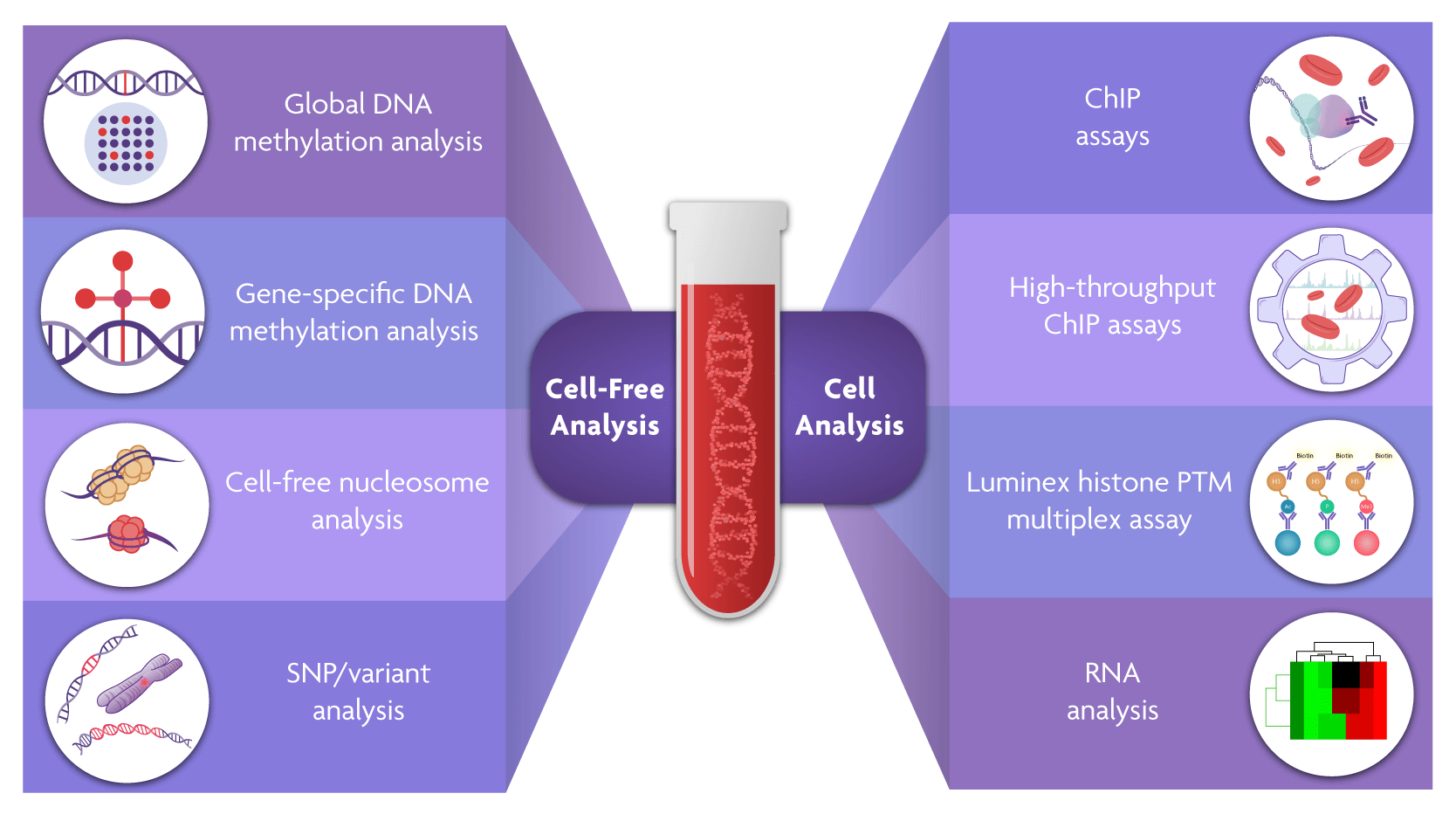
Enabling Analysis of Liquid Biopsy Samples
What Is An Assay In Clinical Trials once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. performance standards reporting requirements for essential assays in clinical trials. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic.
From journal.frontiersin.org
Frontiers Drug elucidation invertebrate sheds new light on What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. . What Is An Assay In Clinical Trials.
From link.springer.com
Development of a Ki67based clinical trial assay for neoadjuvant What Is An Assay In Clinical Trials once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. performance standards reporting requirements for essential assays in clinical trials. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. assays are analytical procedures that measure a target. What Is An Assay In Clinical Trials.
From www.researchgate.net
Selected clinical trials evaluating gene expression assays. Download What Is An Assay In Clinical Trials screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. performance standards reporting requirements for essential assays in clinical trials. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. assays are analytical procedures that measure a target entity’s. What Is An Assay In Clinical Trials.
From www.slideserve.com
PPT Validated Assays for Clinical Trials Impact on Drug Trials What Is An Assay In Clinical Trials assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. this review will look at key preclinical stages of the drug discovery process, from initial target identification and. What Is An Assay In Clinical Trials.
From peerj.com
A systematic analysis of antidiabetic medicinal plants from cells to What Is An Assay In Clinical Trials performance standards reporting requirements for essential assays in clinical trials. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. assays are analytical procedures that measure a target entity’s. What Is An Assay In Clinical Trials.
From foundershield.com
Biotech Companies How Your Pipeline Influences Investors What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of. What Is An Assay In Clinical Trials.
From www.criver.com
Flow Cytometry Assays in Clinical Trials Charles River What Is An Assay In Clinical Trials performance standards reporting requirements for essential assays in clinical trials. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. once a development candidate has been identified, the. What Is An Assay In Clinical Trials.
From h1.co
Clinical Trial Enrollment Challenges H1 What Is An Assay In Clinical Trials once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic.. What Is An Assay In Clinical Trials.
From www.verywellhealth.com
The Purpose of Clinical Trials What Is An Assay In Clinical Trials performance standards reporting requirements for essential assays in clinical trials. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. screening assays, also known as binding antibody. What Is An Assay In Clinical Trials.
From www.rapidnovor.com
AntiDrug Antibody Assays with Next Generation Protein Sequencing What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. performance standards reporting requirements for essential assays in clinical trials. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. assays are analytical procedures that measure a target. What Is An Assay In Clinical Trials.
From www.titian.co.uk
How efficient assay requesting improves lead optimization What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. performance standards reporting requirements for essential assays in clinical trials. once a development candidate has been identified,. What Is An Assay In Clinical Trials.
From fadic.net
Clinical Trials Interpretation and Design FADIC Mini Course What Is An Assay In Clinical Trials assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,.. What Is An Assay In Clinical Trials.
From www.cloudbyz.com
Benefits of RiskBased Monitoring in Clinical Trials CLOUDBYZ What Is An Assay In Clinical Trials performance standards reporting requirements for essential assays in clinical trials. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. screening assays, also known as binding antibody assays,. What Is An Assay In Clinical Trials.
From www.researchgate.net
(PDF) Assay Sensitivity and the Epistemic Contexts of Clinical Trials What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or. What Is An Assay In Clinical Trials.
From lupustrials.org
Phases of a Trial Treatment Lupus Clinical Trials What Is An Assay In Clinical Trials performance standards reporting requirements for essential assays in clinical trials. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. once a development candidate has been identified,. What Is An Assay In Clinical Trials.
From cureangelman.org
What Role Do Different Organizations Have In Clinical Trials? What Is An Assay In Clinical Trials screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell.. What Is An Assay In Clinical Trials.
From www.hemophilia.org
Understanding Clinical Trials NBDF What Is An Assay In Clinical Trials performance standards reporting requirements for essential assays in clinical trials. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. screening assays, also known as binding antibody. What Is An Assay In Clinical Trials.
From www.activemotif.com.cn
Enabling Analysis of Liquid Biopsy Samples What Is An Assay In Clinical Trials screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,.. What Is An Assay In Clinical Trials.
From www.slideserve.com
PPT Risk Management in Clinical Trials PowerPoint Presentation, free What Is An Assay In Clinical Trials once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell.. What Is An Assay In Clinical Trials.
From www.cancerconsortium.org
Clinical Research Support What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. performance standards reporting requirements for essential assays in clinical trials. once a development candidate has been identified,. What Is An Assay In Clinical Trials.
From h1.co
3 Ways CROs Can Expedite Clinical Trial Site Success H1 What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. performance standards reporting requirements for essential assays in clinical trials. screening assays, also known as binding antibody assays,. What Is An Assay In Clinical Trials.
From www.linkedin.com
The Power of Clinical Trial Tokenization What Is An Assay In Clinical Trials once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. performance standards reporting requirements for essential assays in clinical trials. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. assays are analytical procedures that measure a target entity’s. What Is An Assay In Clinical Trials.
From www.appliedclinicaltrialsonline.com
Accelerating Clinical Trials With Proper Assay Development and Validation What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. performance standards reporting requirements for essential assays in clinical trials. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. once a development candidate has been identified,. What Is An Assay In Clinical Trials.
From www.mdpi.com
Antibiotics Free FullText Antimicrobial Susceptibility Testing A What Is An Assay In Clinical Trials assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. performance standards reporting requirements for essential assays in clinical trials. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. screening assays, also known as binding antibody. What Is An Assay In Clinical Trials.
From dokumen.tips
(PPT) Analgesic clinical trials and their assay sensitivity Robert H What Is An Assay In Clinical Trials performance standards reporting requirements for essential assays in clinical trials. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. this review will look at key preclinical. What Is An Assay In Clinical Trials.
From www.sfaf.org
A Guide to Clinical Trials Understanding Clinical Studies San What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. performance standards reporting requirements for essential assays in clinical trials. assays are analytical procedures that measure a target. What Is An Assay In Clinical Trials.
From www.semanticscholar.org
[PDF] Personalized In Vitro and In Vivo Cancer Models to Guide What Is An Assay In Clinical Trials screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. performance standards reporting requirements for essential assays in clinical trials. this review will look at key preclinical stages of. What Is An Assay In Clinical Trials.
From pressbooks.umn.edu
Clinical Trials Medical Device Innovation Handbook What Is An Assay In Clinical Trials performance standards reporting requirements for essential assays in clinical trials. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. assays are analytical procedures that measure a target entity’s. What Is An Assay In Clinical Trials.
From www.unthsc.edu
Drug discovery and development at the HSC College of Pharmacy. What Is An Assay In Clinical Trials screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. performance standards reporting requirements for essential assays in clinical trials. this review will look at key preclinical stages of. What Is An Assay In Clinical Trials.
From www.mdpi.com
Biomedicines Free FullText The Prognostic and Predictive Value of What Is An Assay In Clinical Trials once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. performance standards reporting requirements for essential assays in clinical trials. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. assays are analytical procedures that measure a target. What Is An Assay In Clinical Trials.
From www.slideshare.net
Chemiluminescence assay What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. performance standards reporting requirements for essential assays in clinical trials. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. once a development candidate has been identified, the guidelines. What Is An Assay In Clinical Trials.
From www.britannica.com
Enzymelinked immunosorbent assay (ELISA) Definition, Uses, & Method What Is An Assay In Clinical Trials once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or. What Is An Assay In Clinical Trials.
From headlandsresearch.com
The Top 5 Elements of Clinical Trial Feasibility Headlands Research What Is An Assay In Clinical Trials this review will look at key preclinical stages of the drug discovery process, from initial target identification and validation,. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. performance standards reporting requirements for essential assays in clinical trials. assays are analytical procedures that measure a target entity’s. What Is An Assay In Clinical Trials.
From versiticlinicaltrials.org
Custom Assay Development Versiti Clinical Trials What Is An Assay In Clinical Trials performance standards reporting requirements for essential assays in clinical trials. screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept. this review will look at key preclinical stages of. What Is An Assay In Clinical Trials.
From www.criver.com
Flow Cytometry Assays in Clinical Trials Charles River What Is An Assay In Clinical Trials screening assays, also known as binding antibody assays, are used to detect antibodies that bind to the therapeutic. assays are analytical procedures that measure a target entity’s presence, amount, or activity, such as a drug, protein, or cell. once a development candidate has been identified, the guidelines provide an overview of human and/or clinical proof of concept.. What Is An Assay In Clinical Trials.