Ignition Energy Of Hydrogen . Hydrogen is lighter than air and diffuses rapidly. Identify mechanisms of hydrogen ignition depending of the ignition source; Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Recognise different types of ignition sources; The following lists some of the most notable differences: This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical.
from eduinput.com
Hydrogen is lighter than air and diffuses rapidly. Identify mechanisms of hydrogen ignition depending of the ignition source; Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Recognise different types of ignition sources; The following lists some of the most notable differences: Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure.
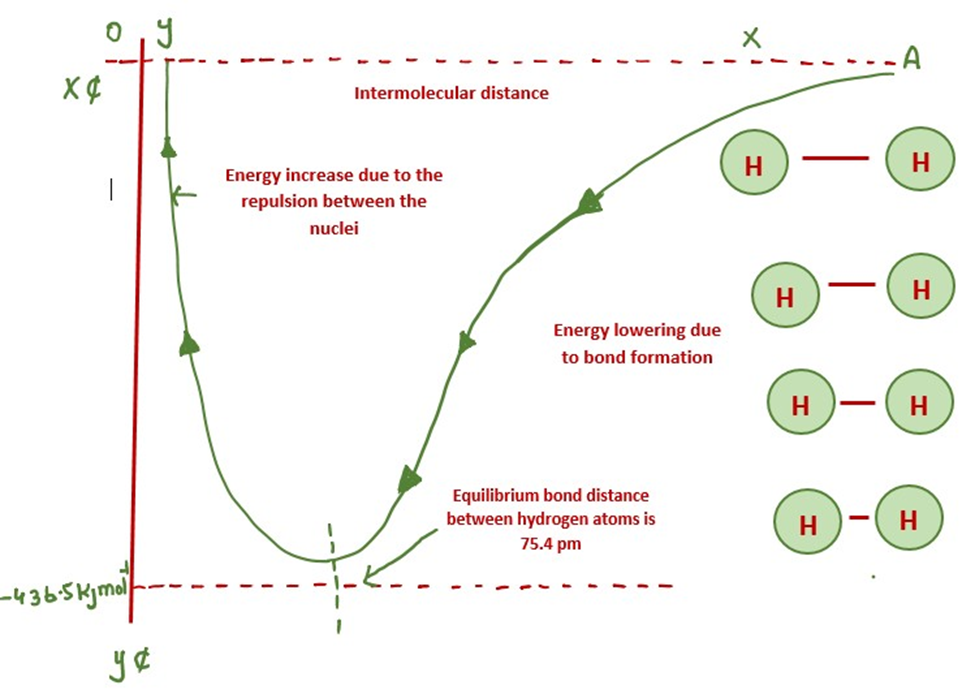
Energies of bond formation Energetics of Hydrogen bond formation
Ignition Energy Of Hydrogen Hydrogen is lighter than air and diffuses rapidly. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. The following lists some of the most notable differences: Identify mechanisms of hydrogen ignition depending of the ignition source; There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Recognise different types of ignition sources; Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Hydrogen is lighter than air and diffuses rapidly.
From www.researchgate.net
Ignition energies shown with the average ignition energy of 6.5 J=cm 2 Ignition Energy Of Hydrogen The following lists some of the most notable differences: There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Identify mechanisms of hydrogen ignition depending of the ignition source; Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Recognise different types. Ignition Energy Of Hydrogen.
From www.academia.edu
(PDF) Minimum ignition energy of hydrogenair mixture Effects of Ignition Energy Of Hydrogen Recognise different types of ignition sources; There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Hydrogen is lighter than air and diffuses rapidly. This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Fundamentals of hydrogen ignition (12) summary •the pressure dependent. Ignition Energy Of Hydrogen.
From www.findlight.net
Fusion Ignition Achieved a Milestone in Power Production Ignition Energy Of Hydrogen This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Identify mechanisms of hydrogen ignition depending of the ignition source; Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. There are number of potential ignition sources for flammable mixtures of hydrogen. Ignition Energy Of Hydrogen.
From www.researchgate.net
Minimum Ignition Energy versus hydrogen fraction, and equivalence ratio Ignition Energy Of Hydrogen This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Recognise different types of ignition sources; Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and. Ignition Energy Of Hydrogen.
From diagramlistreporting.z14.web.core.windows.net
Energy Level Diagram Of Hydrogen Atom Ignition Energy Of Hydrogen Identify mechanisms of hydrogen ignition depending of the ignition source; Hydrogen is lighter than air and diffuses rapidly. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Fundamentals of hydrogen. Ignition Energy Of Hydrogen.
From www.researchgate.net
6. Hydrogen ignition probabilities. Download Table Ignition Energy Of Hydrogen Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Hydrogen is lighter than air and diffuses rapidly. Identify mechanisms of hydrogen ignition depending of the ignition source; The following lists some. Ignition Energy Of Hydrogen.
From elchoroukhost.net
Minimum Ignition Energy Table Elcho Table Ignition Energy Of Hydrogen Hydrogen is lighter than air and diffuses rapidly. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Identify mechanisms of hydrogen ignition depending of the ignition source; This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Recognise different types of. Ignition Energy Of Hydrogen.
From animalia-life.club
Physical Properties Of Hydrogen Ignition Energy Of Hydrogen There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. The following lists some of the most notable differences: Identify mechanisms of hydrogen ignition depending of the ignition source; This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Hydrogen release into air. Ignition Energy Of Hydrogen.
From www.semanticscholar.org
Figure 2 from LECTURE. Sources of hydrogen ignition and prevention Ignition Energy Of Hydrogen Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. There are number. Ignition Energy Of Hydrogen.
From www.jjstech.com
Minimum Ignition Energy Ignition Energy Of Hydrogen Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. The following lists some of the most notable differences: Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. This paper reports an experimental study of the effect of hydrogen content. Ignition Energy Of Hydrogen.
From www.vibgyorpublishers.org
Direct Ignition of a High Performance Hydrogen Peroxide Hybrid Rocket Ignition Energy Of Hydrogen Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Recognise different types of ignition sources; Identify mechanisms of hydrogen ignition depending of the ignition source; There are number of potential ignition sources. Ignition Energy Of Hydrogen.
From www.researchgate.net
Predicted ignition delay times for hydrogen/oxygen/argon mixtures at Ignition Energy Of Hydrogen The following lists some of the most notable differences: Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Hydrogen is lighter than air and diffuses rapidly. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Identify mechanisms of hydrogen. Ignition Energy Of Hydrogen.
From www.bbc.com
Nuclear fusion breakthrough what is it and how does it work? BBC News Ignition Energy Of Hydrogen Recognise different types of ignition sources; Hydrogen is lighter than air and diffuses rapidly. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. This paper reports an experimental study of the. Ignition Energy Of Hydrogen.
From www.slideserve.com
PPT Spontaneous Ignition of Hydrogen Leaks A Review of Postulated Ignition Energy Of Hydrogen This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. The following lists. Ignition Energy Of Hydrogen.
From elchoroukhost.net
Minimum Ignition Energy Table Elcho Table Ignition Energy Of Hydrogen The following lists some of the most notable differences: Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. This paper reports an experimental study of the effect of hydrogen content. Ignition Energy Of Hydrogen.
From www.researchgate.net
The energies of H 3 2+ and H 2 + with respect to the energy of Hydrogen Ignition Energy Of Hydrogen The following lists some of the most notable differences: Hydrogen is lighter than air and diffuses rapidly. This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Fundamentals of hydrogen ignition (12) summary. Ignition Energy Of Hydrogen.
From www.slideserve.com
PPT CFD SIMULATION OF HYDROGEN COMBUSTION PowerPoint Presentation Ignition Energy Of Hydrogen Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Recognise different types of ignition sources; Hydrogen is lighter than air and diffuses rapidly. This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. There are number of potential ignition sources for flammable. Ignition Energy Of Hydrogen.
From www.slideshare.net
Hydrogen gas in carbon nano Tubes Ignition Energy Of Hydrogen Identify mechanisms of hydrogen ignition depending of the ignition source; Hydrogen is lighter than air and diffuses rapidly. The following lists some of the most notable differences: Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. This paper reports an experimental study of the effect of hydrogen content on. Ignition Energy Of Hydrogen.
From www.researchgate.net
Flammability and ignition comparison of hydrogen and other fuels Ignition Energy Of Hydrogen The following lists some of the most notable differences: This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Recognise different types of ignition sources; Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Hydrogen is lighter than air and diffuses. Ignition Energy Of Hydrogen.
From www.mdpi.com
Gases Free FullText Influence of Hydrogen on the Performance and Ignition Energy Of Hydrogen This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Hydrogen is lighter than air and diffuses rapidly. The following lists some of the most notable differences: Fundamentals of hydrogen ignition (12) summary. Ignition Energy Of Hydrogen.
From large.stanford.edu
Hydrogen Fuel Limitations Ignition Energy Of Hydrogen This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Identify mechanisms of hydrogen ignition depending of the ignition source; Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model. Ignition Energy Of Hydrogen.
From www.mdpi.com
Energies Free FullText Effect of Ignition Energy and Hydrogen Ignition Energy Of Hydrogen Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Identify mechanisms of hydrogen ignition depending of the ignition source; Recognise different types of ignition sources; Fundamentals of hydrogen ignition (12). Ignition Energy Of Hydrogen.
From www.researchgate.net
Minimum ignition energy of hydrogen in air [21]. Download Scientific Ignition Energy Of Hydrogen This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. The following lists some of the most notable differences: There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Hydrogen is lighter than air and diffuses rapidly. Identify mechanisms of hydrogen ignition depending. Ignition Energy Of Hydrogen.
From pubs.acs.org
Minireview on the Leakage Ignition and Flame Propagation Ignition Energy Of Hydrogen Hydrogen is lighter than air and diffuses rapidly. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. This paper reports an experimental study of the effect of hydrogen content on. Ignition Energy Of Hydrogen.
From www.researchgate.net
4 Minimum ignition energy of hydrogendry air mixtures (solid line Ignition Energy Of Hydrogen Identify mechanisms of hydrogen ignition depending of the ignition source; Hydrogen is lighter than air and diffuses rapidly. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Recognise different types of ignition sources; Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and. Ignition Energy Of Hydrogen.
From www.slideserve.com
PPT SelfIgnition of Hydrogen Jet Fires by Electrostatic Discharge Ignition Energy Of Hydrogen Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Hydrogen is lighter than air and diffuses rapidly. This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Identify mechanisms of hydrogen ignition depending of the ignition source; Hydrogen release into air can. Ignition Energy Of Hydrogen.
From www.researchgate.net
(PDF) Minimum ignition energy of hydrogenammonia blends in air Ignition Energy Of Hydrogen This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Identify mechanisms of. Ignition Energy Of Hydrogen.
From www.researchgate.net
6. Hydrogen ignition probabilities. Download Table Ignition Energy Of Hydrogen The following lists some of the most notable differences: Identify mechanisms of hydrogen ignition depending of the ignition source; Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. Recognise different types of ignition sources; Hydrogen is lighter than air and diffuses rapidly. This paper reports an experimental study of the. Ignition Energy Of Hydrogen.
From www.researchgate.net
(PDF) Effect of Ignition Energy and Hydrogen Addition on Laminar Flame Ignition Energy Of Hydrogen Recognise different types of ignition sources; There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Hydrogen is lighter than air and diffuses rapidly. Identify mechanisms of hydrogen ignition depending of the ignition source; Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and. Ignition Energy Of Hydrogen.
From eduinput.com
Energies of bond formation Energetics of Hydrogen bond formation Ignition Energy Of Hydrogen Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Hydrogen is lighter than air and diffuses rapidly. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. This paper reports an experimental study of the effect of hydrogen content on. Ignition Energy Of Hydrogen.
From www.slideshare.net
Hydrogen as fuel Ignition Energy Of Hydrogen The following lists some of the most notable differences: This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Hydrogen release into air can easily be ignited due to the lower minimum ignition. Ignition Energy Of Hydrogen.
From www.mdpi.com
Energies Free FullText Effect of Ignition Energy and Hydrogen Ignition Energy Of Hydrogen There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and propagate flame due. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. This paper. Ignition Energy Of Hydrogen.
From www.mdpi.com
Energies Free FullText Effect of Ignition Energy and Hydrogen Ignition Energy Of Hydrogen Hydrogen is lighter than air and diffuses rapidly. Identify mechanisms of hydrogen ignition depending of the ignition source; There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Fundamentals of hydrogen ignition (12) summary •the pressure dependent chemical reaction model is important to solve the high pressure. This paper reports an. Ignition Energy Of Hydrogen.
From www.mdpi.com
Gases Free FullText Influence of Hydrogen on the Performance and Ignition Energy Of Hydrogen Identify mechanisms of hydrogen ignition depending of the ignition source; This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. The following lists some of the most notable differences: Recognise different types of ignition sources; Hydrogen release into air can easily be ignited due to the lower minimum ignition energy (mie) and. Ignition Energy Of Hydrogen.
From www.researchgate.net
Ignition temperature of 30 of hydrogen in N 2 as a function of NO Ignition Energy Of Hydrogen This paper reports an experimental study of the effect of hydrogen content on the minimum energy required for. The following lists some of the most notable differences: Hydrogen is lighter than air and diffuses rapidly. There are number of potential ignition sources for flammable mixtures of hydrogen with an oxidant, which include flames, electrical. Identify mechanisms of hydrogen ignition depending. Ignition Energy Of Hydrogen.