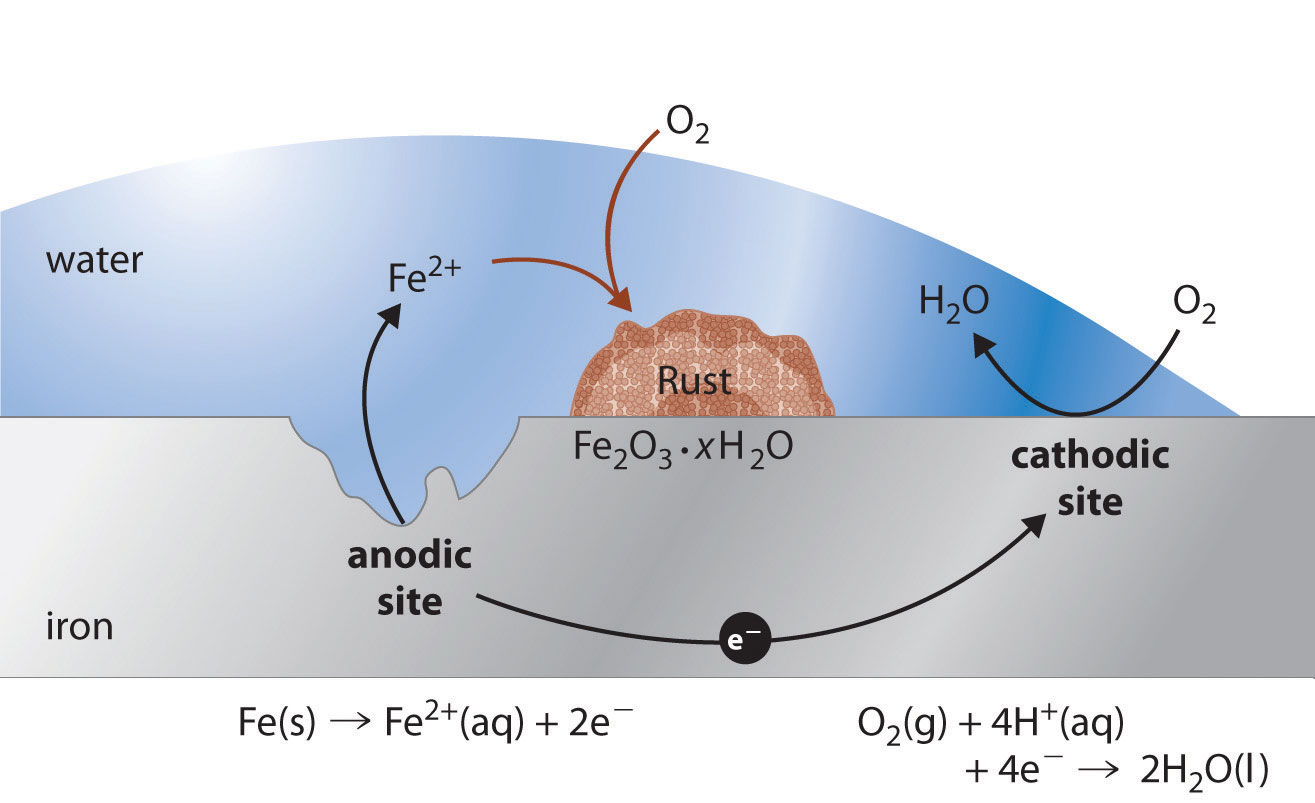
What Is Electrochemical Reaction In Corrosion . Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. It is also a chemical reaction which involves oxidation and reduction. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons.
from 2012books.lardbucket.org
It is also a chemical reaction which involves oxidation and reduction. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the.
Corrosion
What Is Electrochemical Reaction In Corrosion We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. It is also a chemical reaction which involves oxidation and reduction.
From knowledge.electrochem.org
Electrochemistry Encyclopedia Electrochemistry of corrosion What Is Electrochemical Reaction In Corrosion Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. If an electrochemical reaction occurs on a metal surface,. What Is Electrochemical Reaction In Corrosion.
From courses.lumenlearning.com
Corrosion Chemistry What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. Electrochemical corrosion. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Proposed mechanism of electrochemical corrosion generating LTHC stress What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. We also examine the chemical basis. What Is Electrochemical Reaction In Corrosion.
From www.youtube.com
Electrochemical Nature of Aqueous Corrosion YouTube What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. It is also a chemical reaction which involves oxidation and reduction. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. If an electrochemical reaction occurs on a metal surface, then this leads to metal. What Is Electrochemical Reaction In Corrosion.
From www.slideserve.com
PPT Corrosion and Degradation of Materials PowerPoint Presentation What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. If an electrochemical reaction occurs on a metal surface, then this leads. What Is Electrochemical Reaction In Corrosion.
From www.youtube.com
Corrosion Electrochemical Cell or Corrosion Cell (Chapter 3 What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. It is also a chemical reaction which involves oxidation and reduction. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Schematic diagram of electrochemical corrosion mechanism Download What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. An electrochemical reaction. What Is Electrochemical Reaction In Corrosion.
From www.slideserve.com
PPT CORROSION PowerPoint Presentation, free download ID290411 What Is Electrochemical Reaction In Corrosion In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. It is also a chemical reaction which involves oxidation and reduction. Electrochemical. What Is Electrochemical Reaction In Corrosion.
From www.youtube.com
Part 1 Corrosion in refineries, Electrochemical & dry corrosion What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. It is also a. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Cathodic electrochemical reactions associated with CO2 corrosion for What Is Electrochemical Reaction In Corrosion We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. In this section, we describe some of the. What Is Electrochemical Reaction In Corrosion.
From www.youtube.com
Electrochemical theory of corrosion YouTube What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. If an electrochemical reaction occurs on a metal surface,. What Is Electrochemical Reaction In Corrosion.
From www.slideshare.net
Principles of corrosion What Is Electrochemical Reaction In Corrosion It is also a chemical reaction which involves oxidation and reduction. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Typical reaction during electrochemical corrosion. Electrons are What Is Electrochemical Reaction In Corrosion Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. If an electrochemical reaction occurs on a metal surface,. What Is Electrochemical Reaction In Corrosion.
From www.vrogue.co
Corrosion An Electrochemical Reaction vrogue.co What Is Electrochemical Reaction In Corrosion An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. It is also a chemical reaction which involves oxidation and reduction. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. Corrosion in an aqueous. What Is Electrochemical Reaction In Corrosion.
From www.slideserve.com
PPT Ch. 20 Electrochemistry PowerPoint Presentation, free download What Is Electrochemical Reaction In Corrosion In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. It is also a chemical reaction which involves oxidation and reduction. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. Electrochemical. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Corrosion as an electrochemical process.[1] Download Scientific Diagram What Is Electrochemical Reaction In Corrosion An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. It is also a chemical reaction which involves oxidation and reduction. We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Electrochemical corrosion mechanism of the... Download Scientific Diagram What Is Electrochemical Reaction In Corrosion Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. If an electrochemical reaction occurs on a metal surface,. What Is Electrochemical Reaction In Corrosion.
From www.youtube.com
Electrochemical Corrosion YouTube What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. It is also a chemical reaction which involves oxidation and reduction. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. An electrochemical reaction is defined as a chemical. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Figure The anode and cathode reactions in typical electrolytic What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. We also examine the chemical basis for some. What Is Electrochemical Reaction In Corrosion.
From www.hilti.com
Corrosion in Construction Hilti USA What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. It is also a chemical reaction which involves oxidation and reduction. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. If an electrochemical reaction occurs on a metal surface, then this. What Is Electrochemical Reaction In Corrosion.
From mavink.com
Electrochemical Theory Of Corrosion What Is Electrochemical Reaction In Corrosion We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. It is also a chemical reaction which involves oxidation and reduction. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are. What Is Electrochemical Reaction In Corrosion.
From www.youtube.com
Hydrogen evolution and Oxygen Absorption Mechanism of wet corrosion What Is Electrochemical Reaction In Corrosion An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. We also examine the chemical basis for some. What Is Electrochemical Reaction In Corrosion.
From www.vrogue.co
Corrosion An Electrochemical Reaction vrogue.co What Is Electrochemical Reaction In Corrosion It is also a chemical reaction which involves oxidation and reduction. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. Corrosion in an aqueous environment and in an atmospheric setting is. What Is Electrochemical Reaction In Corrosion.
From pharmacyscope.com
Theories of Corrosion Pharmacy Scope What Is Electrochemical Reaction In Corrosion An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. If an electrochemical reaction occurs on a metal surface, then. What Is Electrochemical Reaction In Corrosion.
From www.corrscience.com
Corr Science » Principles of Corrosion What Is Electrochemical Reaction In Corrosion Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. It is also a chemical reaction which involves oxidation and reduction. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Representation of electrochemical corrosion. Download Scientific Diagram What Is Electrochemical Reaction In Corrosion In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. Corrosion in an aqueous environment and in an atmospheric setting is an. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
3. Schematic representation of the electrochemical process of What Is Electrochemical Reaction In Corrosion We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation and the electrochemical process is. It is also a chemical reaction which involves oxidation and reduction. In this section, we describe some of the chemical and. What Is Electrochemical Reaction In Corrosion.
From www.slideserve.com
PPT Electrochemistry PowerPoint Presentation, free download ID7041672 What Is Electrochemical Reaction In Corrosion An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. Corrosion in an aqueous environment and in an atmospheric setting is. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Electrochemical process of corrosion... Download Scientific Diagram What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Scheme illustrating (a) the electrochemical corrosion process at a What Is Electrochemical Reaction In Corrosion An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. If an electrochemical reaction occurs on a metal surface, then this. What Is Electrochemical Reaction In Corrosion.
From chem.libretexts.org
Chapter 19.6 Corrosion Chemistry LibreTexts What Is Electrochemical Reaction In Corrosion It is also a chemical reaction which involves oxidation and reduction. In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons. What Is Electrochemical Reaction In Corrosion.
From www.slideserve.com
PPT Corrosion Mechanisms PowerPoint Presentation, free download ID What Is Electrochemical Reaction In Corrosion If an electrochemical reaction occurs on a metal surface, then this leads to metal deterioration or degradation, and the. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. In this section, we describe some. What Is Electrochemical Reaction In Corrosion.
From 2012books.lardbucket.org
Corrosion What Is Electrochemical Reaction In Corrosion In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. An electrochemical reaction is defined as a chemical reaction involving the transfer of electrons. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. It is also a chemical reaction which involves oxidation and reduction. If an electrochemical reaction. What Is Electrochemical Reaction In Corrosion.
From chem.libretexts.org
Chapter 19.6 Corrosion Chemistry LibreTexts What Is Electrochemical Reaction In Corrosion In this section, we describe some of the chemical and electrochemical processes responsible for corrosion. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte solution, resulting in the deterioration of the substrate. If an electrochemical reaction occurs on a metal surface, then. What Is Electrochemical Reaction In Corrosion.
From www.researchgate.net
Electrochemical accelerated corrosion method (a) Schematic diagram of What Is Electrochemical Reaction In Corrosion We also examine the chemical basis for some common methods for preventing corrosion and treating corroded metals. Electrochemical corrosion occurs in the large variety of electrolytes found in natural environments and industrial applications. Corrosion in an aqueous environment and in an atmospheric setting is an electrochemical process in which electrons are transferred between a metal surface and a liquid electrolyte. What Is Electrochemical Reaction In Corrosion.