Evaporation In Carbonated Water . Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. Crystallisation is a separation technique used to obtain crystals of a solid solute. We have answered what the evaporation rate is: When a solution is heated,. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. If the water is instead kept in a closed. 1.2 lb per hour (0.55 kg per hour). Carbon dioxide is weakly soluble in water,.
from www.dreamstime.com
1.2 lb per hour (0.55 kg per hour). Crystallisation is a separation technique used to obtain crystals of a solid solute. If the water is instead kept in a closed. Carbon dioxide is weakly soluble in water,. What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. We have answered what the evaporation rate is: Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. When a solution is heated,.
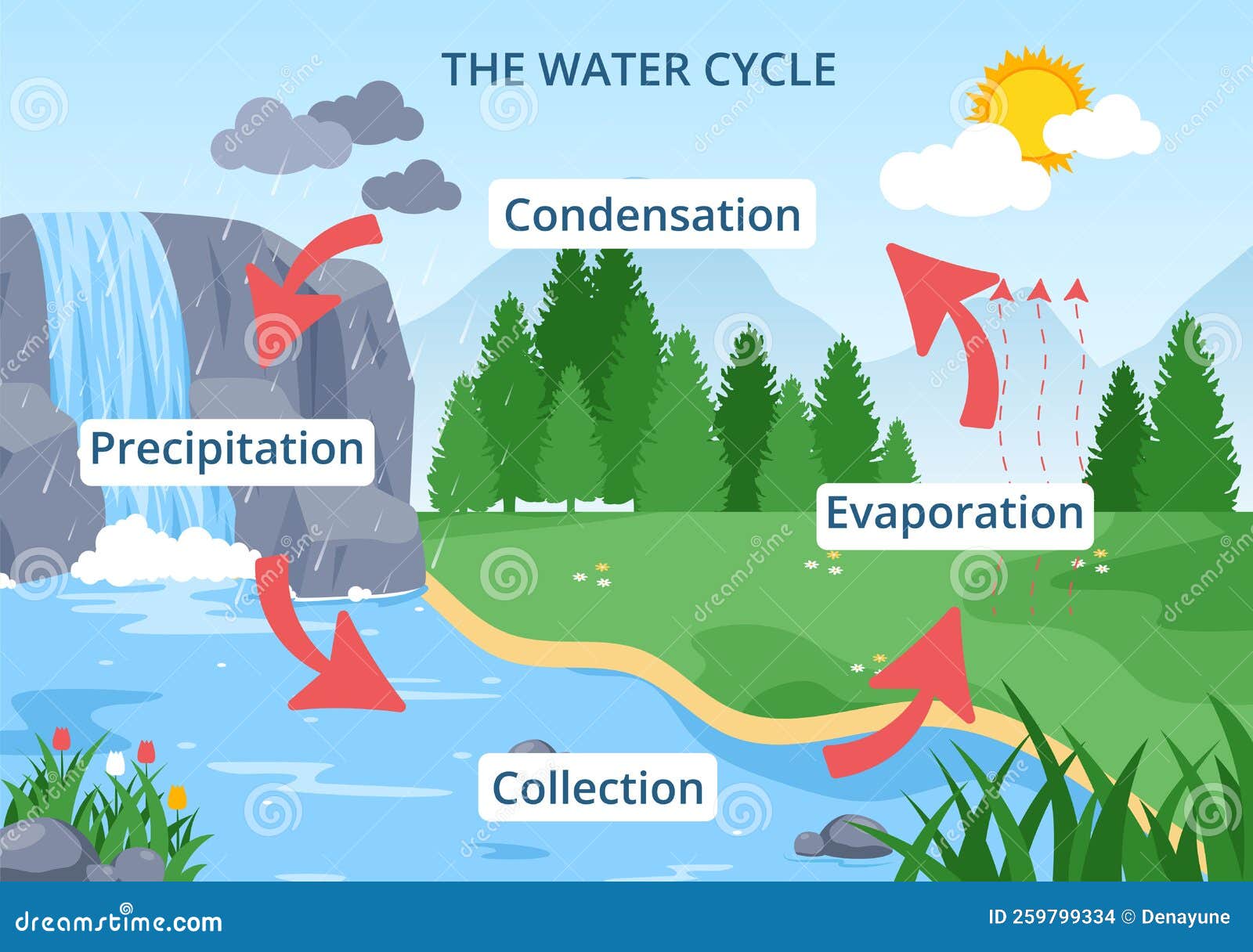
Water Cycle of Evaporation, Condensation, Precipitation To Collection
Evaporation In Carbonated Water Carbon dioxide is weakly soluble in water,. What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. We have answered what the evaporation rate is: If the water is instead kept in a closed. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. When a solution is heated,. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. 1.2 lb per hour (0.55 kg per hour). Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. Carbon dioxide is weakly soluble in water,. Crystallisation is a separation technique used to obtain crystals of a solid solute.
From www.nsta.org
Q What’s the difference between evaporation and boiling? NSTA Evaporation In Carbonated Water Crystallisation is a separation technique used to obtain crystals of a solid solute. Carbon dioxide is weakly soluble in water,. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. 1.2 lb. Evaporation In Carbonated Water.
From www.chemicals.co.uk
What is the Definition of Evaporation in Chemistry? Evaporation In Carbonated Water Carbon dioxide is weakly soluble in water,. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. Crystallisation is a separation technique used to obtain crystals of a solid solute. We have answered what the evaporation rate is: Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. If the water is instead. Evaporation In Carbonated Water.
From www.dreamstime.com
Water Cycle of Evaporation, Condensation, Precipitation To Collection Evaporation In Carbonated Water A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. If the water is instead kept in a closed. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. 1.2 lb per hour (0.55 kg. Evaporation In Carbonated Water.
From globalnews.ca
Is carbonated water as good for you as regular water? National Evaporation In Carbonated Water We have answered what the evaporation rate is: Crystallisation is a separation technique used to obtain crystals of a solid solute. When a solution is heated,. Carbon dioxide is weakly soluble in water,. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. What’s more, the alcohol and water in a drink evaporate at different rates, which. Evaporation In Carbonated Water.
From www.expii.com
Separating Mixtures — Overview & Common Methods Expii Evaporation In Carbonated Water What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. Carbon dioxide is weakly soluble in water,. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. Crystallisation is a separation technique used to obtain. Evaporation In Carbonated Water.
From www.aplustopper.com
How can we Separate a Mixture of a Solid and a Liquid using Evaporation Evaporation In Carbonated Water Crystallisation is a separation technique used to obtain crystals of a solid solute. Carbon dioxide is weakly soluble in water,. When a solution is heated,. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. If the water is instead kept in a closed. If there was the tiniest hole or crack for water to. Evaporation In Carbonated Water.
From learningschooltrkesp5v.z22.web.core.windows.net
Water Evaporation Rate By Temperature Evaporation In Carbonated Water If the water is instead kept in a closed. We have answered what the evaporation rate is: A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. When a solution is heated,. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. Since. Evaporation In Carbonated Water.
From curejoyinc.com
7 Side Effects Of Drinking Carbonated Water CureJoy Evaporation In Carbonated Water A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. What’s more, the alcohol and water in a drink evaporate at different rates, which produces. Evaporation In Carbonated Water.
From www.bbc.co.uk
Evaporation BBC Bitesize Evaporation In Carbonated Water Carbon dioxide is weakly soluble in water,. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. Evaporation occurs when a liquid slowly turns into. Evaporation In Carbonated Water.
From www.comsol.com
How to Model Sparging in COMSOL Multiphysics® COMSOL Blog Evaporation In Carbonated Water Carbon dioxide is weakly soluble in water,. We have answered what the evaporation rate is: If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. Evaporation is the conversion of a liquid to its. Evaporation In Carbonated Water.
From www.teachoo.com
Why is Evaporation called a surface phenomenon? Teachoo Evaporation In Carbonated Water Evaporation occurs when a liquid slowly turns into a gas below its boiling point. We have answered what the evaporation rate is: Carbon dioxide is weakly soluble in water,. If the water is instead kept in a closed. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. What’s more, the alcohol and. Evaporation In Carbonated Water.
From www.linkedin.com
Things to know about commercial carbonated water systems Evaporation In Carbonated Water When a solution is heated,. We have answered what the evaporation rate is: 1.2 lb per hour (0.55 kg per hour). Crystallisation is a separation technique used to obtain crystals of a solid solute. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. Evaporation occurs when a liquid slowly turns into a. Evaporation In Carbonated Water.
From www.researchgate.net
The evaporation rates, κ, of water/ethanol mixtures at the Evaporation In Carbonated Water Carbon dioxide is weakly soluble in water,. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. Crystallisation is a separation technique used to obtain crystals of a solid solute. What’s more, the alcohol and water in a drink evaporate. Evaporation In Carbonated Water.
From www.sciencephoto.com
Evaporation of water and salt solution Stock Image C036/3503 Evaporation In Carbonated Water Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. Crystallisation is a separation technique used to obtain crystals of a solid solute. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. A common example is the dissolving of carbon dioxide in water, resulting. Evaporation In Carbonated Water.
From www.researchgate.net
a Schematic illustration of water evaporation process and mechanism. b Evaporation In Carbonated Water Crystallisation is a separation technique used to obtain crystals of a solid solute. Carbon dioxide is weakly soluble in water,. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. 1.2 lb per hour. Evaporation In Carbonated Water.
From www.slideserve.com
PPT The Water Cycle and How Humans Impact It PowerPoint Presentation Evaporation In Carbonated Water Evaporation occurs when a liquid slowly turns into a gas below its boiling point. Carbon dioxide is weakly soluble in water,. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. If the water is instead kept in a closed. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),.. Evaporation In Carbonated Water.
From pubs.acs.org
Investigation of Water Evaporation Process at Air/Water Interface using Evaporation In Carbonated Water Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. We have answered what the evaporation rate is: Evaporation occurs when a liquid slowly turns into a gas below its boiling point. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long. Evaporation In Carbonated Water.
From www.pinterest.jp
Evaporation is the process by which a liquid turns into a gas. It is Evaporation In Carbonated Water 1.2 lb per hour (0.55 kg per hour). A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. Carbon dioxide is weakly soluble in water,. Crystallisation is a separation technique used to obtain crystals of a solid solute. We have answered. Evaporation In Carbonated Water.
From www.youtube.com
Carbonated Water evaporation YouTube Evaporation In Carbonated Water A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. If the water is instead kept in a closed. 1.2 lb per hour (0.55 kg per hour). When a solution is heated,. Carbon dioxide is weakly soluble in water,. Crystallisation is a separation technique used to obtain crystals of a solid solute. Evaporation occurs when. Evaporation In Carbonated Water.
From young6science3.weebly.com
5Th grade science The Water Cycle Evaporation In Carbonated Water Evaporation occurs when a liquid slowly turns into a gas below its boiling point. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. We have answered what the evaporation rate is: Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. Crystallisation is a separation technique used. Evaporation In Carbonated Water.
From vt.audubon.org
The Water Cycle Revisited! Audubon Vermont Evaporation In Carbonated Water We have answered what the evaporation rate is: 1.2 lb per hour (0.55 kg per hour). Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. When a solution is heated,. Crystallisation is a separation technique used to obtain. Evaporation In Carbonated Water.
From www.iqsdirectory.com
Wastewater Evaporator What Is It? How Does It Work? Types Evaporation In Carbonated Water What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. 1.2 lb per hour (0.55 kg per hour). Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. A common. Evaporation In Carbonated Water.
From healthyliving.blog
5 Side Effects of Drinking Carbonated Water Healthy Living Evaporation In Carbonated Water Carbon dioxide is weakly soluble in water,. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. When a solution is heated,. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. Evaporation occurs when a liquid slowly turns into a gas below its boiling. Evaporation In Carbonated Water.
From www.pepsioman.com
OmanrefCo Evaporation In Carbonated Water Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. If the water is instead kept in a closed. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the. Evaporation In Carbonated Water.
From foodstruct.com
Water vs. Carbonated water — InDepth Nutrition Comparison Evaporation In Carbonated Water If the water is instead kept in a closed. When a solution is heated,. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. Crystallisation is a separation technique used to obtain crystals of a solid solute. We have answered what. Evaporation In Carbonated Water.
From www.healthbenefitstimes.com
Carbonated water Facts, Health Benefits & Nutritional Value Evaporation In Carbonated Water Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. 1.2 lb per hour (0.55 kg per hour). When a solution is heated,. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. A common example is the dissolving of carbon. Evaporation In Carbonated Water.
From www.scienceabc.com
Are Evaporation And Boiling The Same? » ScienceABC Evaporation In Carbonated Water What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. 1.2 lb per hour (0.55 kg per hour). We have answered what the evaporation rate is: A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. If the water is instead kept. Evaporation In Carbonated Water.
From saltworkconsultants.com
Physics of evaporation Evaporation In Carbonated Water Crystallisation is a separation technique used to obtain crystals of a solid solute. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. Carbon dioxide is weakly soluble in water,. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. When. Evaporation In Carbonated Water.
From www.vedantu.com
Evaporation Learn Important Terms and Concepts Evaporation In Carbonated Water If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. Carbon dioxide is weakly soluble in water,. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. We have answered what the evaporation rate is: Crystallisation is a separation technique used. Evaporation In Carbonated Water.
From www.animalia-life.club
Water Cycle Condensation Evaporation In Carbonated Water Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. We have answered what the. Evaporation In Carbonated Water.
From www.vectorstock.com
Changes of states evaporation water boiling Vector Image Evaporation In Carbonated Water Carbon dioxide is weakly soluble in water,. If there was the tiniest hole or crack for water to flow through, it would have done so and evaporated long ago. Since 1 liter of water weighs approximately 1 kilogram (see water density calculator),. If the water is instead kept in a closed. When a solution is heated,. 1.2 lb per hour. Evaporation In Carbonated Water.
From www.scienceabc.com
Why Does Water Evaporate At Room Temperature? Evaporation In Carbonated Water If the water is instead kept in a closed. What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. We have answered what the evaporation rate is: Carbon dioxide is weakly soluble in water,. Evaporation occurs when a liquid slowly turns into a gas below its boiling. Evaporation In Carbonated Water.
From aquadest.cz
Vacuum evaporation Aquadest Evaporation In Carbonated Water Crystallisation is a separation technique used to obtain crystals of a solid solute. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid. We have answered what the evaporation rate is: When a solution is heated,. What’s more, the. Evaporation In Carbonated Water.
From galliumstem.com
Water Cycle Gallium STEM Evaporation In Carbonated Water What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. Evaporation occurs when a liquid slowly turns into a gas below its boiling point. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. Carbon dioxide is weakly soluble in water,. If. Evaporation In Carbonated Water.
From netsolwater.com
What is the use of evaporators in waste water treatment Evaporation In Carbonated Water What’s more, the alcohol and water in a drink evaporate at different rates, which produces gradients in surface tension at the surface of. A common example is the dissolving of carbon dioxide in water, resulting in carbonated water. 1.2 lb per hour (0.55 kg per hour). Evaporation is the conversion of a liquid to its vapor below the boiling temperature. Evaporation In Carbonated Water.