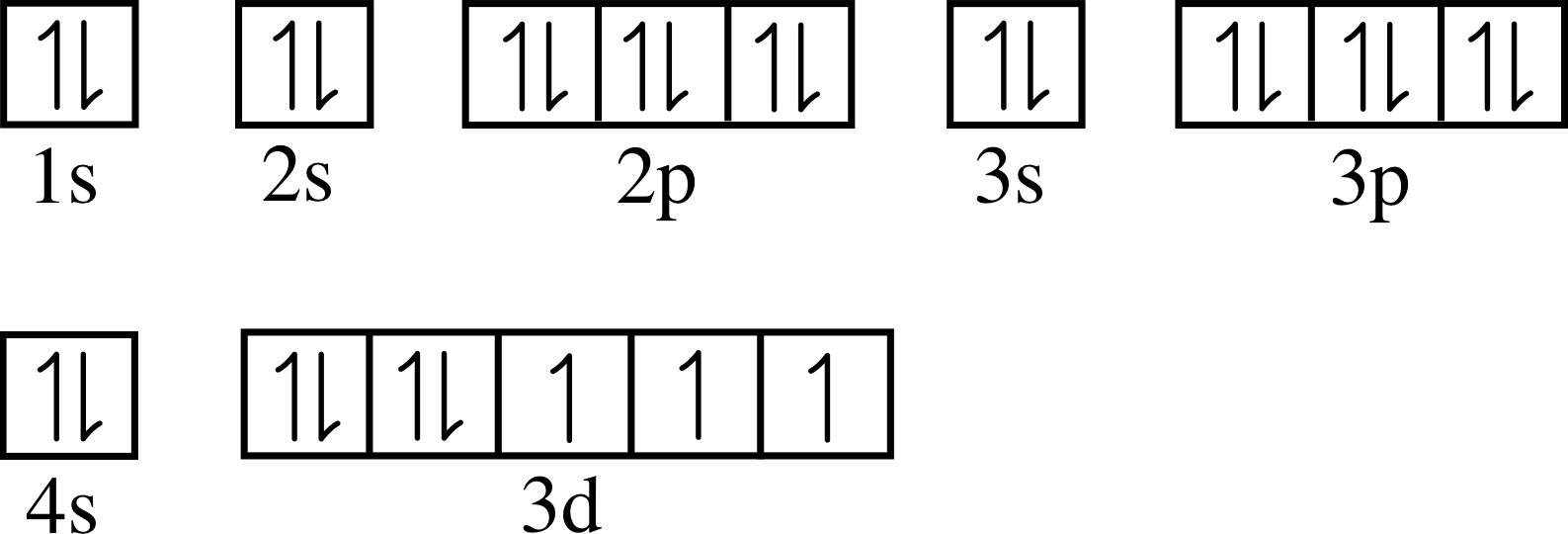
Bromine Number Of Valence Electrons . — valence electrons found in the s and p orbitals of the highest energy. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — bromine (br) is a halogen and has an atomic number of 35. — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. — there are two ways to find the number of valence electrons in bromine. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. 119 rows — — valence electrons: Now let’s see how you can easily find the valence electrons of bromine atom (br). — bromine has 7 valence electrons because there are 7 electrons present in the outermost shell of the bromine (br) atom. Bromine has an electron configuration of.
from dxooqbere.blob.core.windows.net
Now let’s see how you can easily find the valence electrons of bromine atom (br). It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — valence electrons found in the s and p orbitals of the highest energy. Bromine has an electron configuration of. 119 rows — — valence electrons: — bromine (br) is a halogen and has an atomic number of 35. — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. — there are two ways to find the number of valence electrons in bromine. — bromine has 7 valence electrons because there are 7 electrons present in the outermost shell of the bromine (br) atom. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons.
Bromine Configuration Of Electrons at Daniel Wright blog
Bromine Number Of Valence Electrons — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — bromine has 7 valence electrons because there are 7 electrons present in the outermost shell of the bromine (br) atom. 119 rows — — valence electrons: — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. Bromine has an electron configuration of. Now let’s see how you can easily find the valence electrons of bromine atom (br). — valence electrons found in the s and p orbitals of the highest energy. — there are two ways to find the number of valence electrons in bromine. — bromine (br) is a halogen and has an atomic number of 35.
From www.youtube.com
How many valence electrons does bromine have? YouTube Bromine Number Of Valence Electrons 119 rows — — valence electrons: — bromine (br) is a halogen and has an atomic number of 35. — bromine has 7 valence electrons because there are 7 electrons present in the outermost shell of the bromine (br) atom. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence. Bromine Number Of Valence Electrons.
From ar.inspiredpencil.com
Atomic Structure Of Bromine Bromine Number Of Valence Electrons — there are two ways to find the number of valence electrons in bromine. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — bromine (br) is a halogen and has an atomic number of 35. 119. Bromine Number Of Valence Electrons.
From www.youtube.com
Electron Configuration of Bromine Br Lesson YouTube Bromine Number Of Valence Electrons Now let’s see how you can easily find the valence electrons of bromine atom (br). — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. — bromine (br) is a halogen and has an atomic number of 35. — the total number of electrons present in the valence shell of. Bromine Number Of Valence Electrons.
From ar.inspiredpencil.com
Atomic Structure Of Bromine Bromine Number Of Valence Electrons — there are two ways to find the number of valence electrons in bromine. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. Bromine has an electron configuration of. Now let’s see how you can easily find the valence electrons of bromine atom (br). It has electronic configuration of 1s2. Bromine Number Of Valence Electrons.
From www.alamy.com
Bromine (Br). Diagram of the nuclear composition, electron Bromine Number Of Valence Electrons — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. Bromine has an electron configuration of. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. 119 rows — — valence electrons: Now let’s. Bromine Number Of Valence Electrons.
From valenceelectrons.com
How to Find the Valence Electrons for Bromine (Br)? Bromine Number Of Valence Electrons It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. Now let’s see how you can easily find the valence electrons of bromine atom (br). 119 rows — — valence electrons: Bromine has an electron configuration of. — valence electrons found in the s and p orbitals of the highest energy. — there are two ways. Bromine Number Of Valence Electrons.
From wou.edu
CH150 Chapter 2 Atoms and Periodic Table Chemistry Bromine Number Of Valence Electrons It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — there are two ways to find the number of valence electrons in bromine. Bromine has an electron configuration of. — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. . Bromine Number Of Valence Electrons.
From valenceelectrons.com
Complete Electron Configuration for Bromine (Br, Br ion) Bromine Number Of Valence Electrons 119 rows — — valence electrons: — bromine (br) is a halogen and has an atomic number of 35. — there are two ways to find the number of valence electrons in bromine. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — valence electrons found in the s and p orbitals of the. Bromine Number Of Valence Electrons.
From www.youtube.com
How to Find the Valence Electrons for Bromine (Br) YouTube Bromine Number Of Valence Electrons — valence electrons found in the s and p orbitals of the highest energy. 119 rows — — valence electrons: Now let’s see how you can easily find the valence electrons of bromine atom (br). — bromine has 7 valence electrons because there are 7 electrons present in the outermost shell of the bromine (br) atom. . Bromine Number Of Valence Electrons.
From sciencenotes.org
Bromine Facts Atomic Number 35 and Element Symbol Br Bromine Number Of Valence Electrons — valence electrons found in the s and p orbitals of the highest energy. 119 rows — — valence electrons: — bromine (br) is a halogen and has an atomic number of 35. — there are two ways to find the number of valence electrons in bromine. — bromine has 7 valence electrons because there. Bromine Number Of Valence Electrons.
From www.animalia-life.club
Electron Configuration For Bromine Bromine Number Of Valence Electrons — there are two ways to find the number of valence electrons in bromine. Now let’s see how you can easily find the valence electrons of bromine atom (br). It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — valence electrons found in the s and p orbitals of the highest energy. — bromine has. Bromine Number Of Valence Electrons.
From periodictable.me
How Do We Find The Electron Configuration For Bromine Dynamic Bromine Number Of Valence Electrons — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. — there are two ways to find the number of valence electrons in bromine. — bromine (br) is a halogen and has an atomic number of 35. — valence electrons found in the s and p orbitals of the. Bromine Number Of Valence Electrons.
From material-properties.org
Bromine Protons Neutrons Electrons Electron Configuration Bromine Number Of Valence Electrons — valence electrons found in the s and p orbitals of the highest energy. — there are two ways to find the number of valence electrons in bromine. Now let’s see how you can easily find the valence electrons of bromine atom (br). — learn how to draw the lewis structure of bromine, a diatomic molecule with. Bromine Number Of Valence Electrons.
From www.chegg.com
Solved on a Bromine 3. HBr Number of valence electrons Bromine Number Of Valence Electrons Bromine has an electron configuration of. — valence electrons found in the s and p orbitals of the highest energy. — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. . Bromine Number Of Valence Electrons.
From www.slideserve.com
PPT Atoms & Chemical Bonding PowerPoint Presentation, free download Bromine Number Of Valence Electrons It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. 119 rows — — valence electrons: — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. — bromine has 7 valence electrons because there are 7 electrons present in the. Bromine Number Of Valence Electrons.
From chromefity.weebly.com
Find valence electrons on periodic table chromefity Bromine Number Of Valence Electrons — bromine (br) is a halogen and has an atomic number of 35. — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — valence electrons found in the s. Bromine Number Of Valence Electrons.
From www.animalia-life.club
Electron Configuration For Bromine Bromine Number Of Valence Electrons — there are two ways to find the number of valence electrons in bromine. — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. 119 rows — — valence electrons:. Bromine Number Of Valence Electrons.
From brainly.in
bromine valence electrons, Brainly.in Bromine Number Of Valence Electrons — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. — bromine (br) is a halogen and has an atomic number of 35. 119 rows — — valence electrons: — the total number of electrons present in the valence shell of an atom are called valence electrons, and there. Bromine Number Of Valence Electrons.
From stock.adobe.com
Br Bromine Element Information Facts, Properties, Trends, Uses and Bromine Number Of Valence Electrons — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. — bromine (br) is a halogen and has an atomic number of 35. 119 rows — — valence electrons: — learn how to draw the lewis structure of bromine, a. Bromine Number Of Valence Electrons.
From www.numerade.com
SOLVED For the Bromine atom a) Determine the total number of unpaired Bromine Number Of Valence Electrons — valence electrons found in the s and p orbitals of the highest energy. Bromine has an electron configuration of. 119 rows — — valence electrons: — bromine (br) is a halogen and has an atomic number of 35. — the total number of electrons present in the valence shell of an atom are called valence. Bromine Number Of Valence Electrons.
From www.sciencecoverage.com
How Many Valence Electrons Does Bromine (Br) Have? [Valency of Bromine] Bromine Number Of Valence Electrons — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. Bromine has an electron configuration of. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. Now let’s see how you can easily find the valence electrons of bromine atom (br). — valence electrons found in the s and p. Bromine Number Of Valence Electrons.
From app.emaze.com
Bromine ) on emaze Bromine Number Of Valence Electrons Now let’s see how you can easily find the valence electrons of bromine atom (br). — bromine (br) is a halogen and has an atomic number of 35. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. 119 rows — — valence electrons: — bromine has 7 valence. Bromine Number Of Valence Electrons.
From www.alamy.com
Bromine (Br). Diagram of the nuclear composition, electron Bromine Number Of Valence Electrons — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. — there are two ways to find the number of valence electrons in bromine. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. 119 rows — — valence electrons: — the total number of electrons present in. Bromine Number Of Valence Electrons.
From dxooqbere.blob.core.windows.net
Bromine Configuration Of Electrons at Daniel Wright blog Bromine Number Of Valence Electrons — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. Bromine has an electron configuration of. — valence electrons found in the s and p orbitals of the highest energy. — learn how to draw the lewis structure of bromine, a. Bromine Number Of Valence Electrons.
From www.numerade.com
What is the difference between (a) a bromine atom, (b) a bromine Bromine Number Of Valence Electrons Now let’s see how you can easily find the valence electrons of bromine atom (br). Bromine has an electron configuration of. 119 rows — — valence electrons: — bromine has 7 valence electrons because there are 7 electrons present in the outermost shell of the bromine (br) atom. — there are two ways to find the number. Bromine Number Of Valence Electrons.
From material-properties.org
Bromine Periodic Table and Atomic Properties Bromine Number Of Valence Electrons — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. — bromine has 7 valence electrons because there are 7 electrons present in the outermost shell of the bromine (br) atom. Bromine has an electron configuration of. — there are two. Bromine Number Of Valence Electrons.
From valenceelectrons.com
Complete Electron Configuration for Bromine (Br, Br ion) Bromine Number Of Valence Electrons — bromine (br) is a halogen and has an atomic number of 35. — valence electrons found in the s and p orbitals of the highest energy. Now let’s see how you can easily find the valence electrons of bromine atom (br). 119 rows — — valence electrons: It has electronic configuration of 1s2 2s2 2p6 3s2. Bromine Number Of Valence Electrons.
From proper-cooking.info
Bromine Valence Electrons Bromine Number Of Valence Electrons — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. Now let’s see how you can easily find the valence electrons of bromine atom (br). 119 rows — — valence electrons: — learn how to draw the lewis structure of bromine,. Bromine Number Of Valence Electrons.
From periodictable.me
2000pxElectron_configuration_bromine.svg Dynamic Periodic Table of Bromine Number Of Valence Electrons It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. — bromine has 7 valence electrons because there are 7 electrons present in the outermost shell of the bromine (br) atom. — there are two ways to find the. Bromine Number Of Valence Electrons.
From kdi-ppi.com
How to Create a Lewis Dot Diagram for Bromine StepbyStep Guide Bromine Number Of Valence Electrons — valence electrons found in the s and p orbitals of the highest energy. — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a total of seven electrons. Now let’s see how you can easily find the valence electrons of bromine atom (br). — there. Bromine Number Of Valence Electrons.
From srkrkzbhxtdlh.blogspot.com
How Many Valence Electrons Does Bromine Have Bromine is a group viia Bromine Number Of Valence Electrons — valence electrons found in the s and p orbitals of the highest energy. 119 rows — — valence electrons: — there are two ways to find the number of valence electrons in bromine. — the total number of electrons present in the valence shell of an atom are called valence electrons, and there are a. Bromine Number Of Valence Electrons.
From mavink.com
Lewis Dot Diagram For Bromine Bromine Number Of Valence Electrons — bromine (br) is a halogen and has an atomic number of 35. — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. — bromine has 7 valence electrons because there are 7 electrons present in the outermost shell of the bromine (br) atom. — the total number of. Bromine Number Of Valence Electrons.
From www.nuclear-power.com
Bromine Atomic Number Atomic Mass Density of Bromine nuclear Bromine Number Of Valence Electrons — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. — there are two ways to find the number of valence electrons in bromine. It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — bromine has 7 valence electrons because there are 7 electrons present in the outermost. Bromine Number Of Valence Electrons.
From www.animalia-life.club
Electron Configuration For Bromine Bromine Number Of Valence Electrons Now let’s see how you can easily find the valence electrons of bromine atom (br). — learn how to draw the lewis structure of bromine, a diatomic molecule with seven valence electrons. — valence electrons found in the s and p orbitals of the highest energy. — there are two ways to find the number of valence. Bromine Number Of Valence Electrons.
From periodictable.me
Bromine Electron Configuration (Br) with Orbital Diagram Bromine Number Of Valence Electrons — bromine (br) is a halogen and has an atomic number of 35. Bromine has an electron configuration of. 119 rows — — valence electrons: Now let’s see how you can easily find the valence electrons of bromine atom (br). It has electronic configuration of 1s2 2s2 2p6 3s2 3p6 4s2. — bromine has 7 valence electrons. Bromine Number Of Valence Electrons.